You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Acetaminophen is one of the most universal over-the-counter (OTC) medications used to treat pain and fever, and the most common drug ingredient in the United States.1-5 Fifty-two million consumers (approximately 23% of all adults in the United States) use an acetaminophen-containing product to manage fever and mild to moderate acute or chronic pain of headaches, migraine headaches, toothaches, menstrual cramps, and osteoarthritis.6-10 Acetaminophen is also the drug of choice for pregnant and lactating women to treat mild pain and for children with fevers.11-17
More than 600 different products contain acetaminophen, and it is available in a plethora of dosage forms: capsules, chewable tablets, effervescent tablets, elixirs, extended-release tablets, suppositories, and regular oral tablets. In many countries purchases of acetaminophen-containing products are not restricted to pharmacies, and sales are increasing worldwide.18 For the treatment of more severe pain, acetaminophen is often formulated with non-opioid and opioid agents such as ibuprofen, codeine, hydrocodone, oxycodone, and tramadol; acetaminophen and opioid-containing products are available by prescription only. Given the accessibility of acetaminophen and its widespread use, should there be any significant safety concerns within dentistry?
History
Some of the earliest remedies to treat fevers (antipyretic compounds) were known to be contained in white willow and cinchona bark.19 These chemicals were known as salicins (which were the precursors of aspirin) and quinine (an anti-malarial drug, which also has anti-fever activity). The burgeoning science of pharmacognosy, however, led to the over-harvesting of many of these natural sources, and scientists began synthesizing new antipyretic agents in the laboratory, most notably acetanilide in 1886 and phenacetin the following year.20
Harmon Morse is credited with the production of acetaminophen in 1887, when he accidently prepared the drug by combining paranitrophenol, instead of analine, with glacial acetic acid and tin.21 The discovery was made after the incorrect medicine, N-acetyl-para-aminophenol (acetaminophen or paracetamol) and not acetanilide, was given to a patient, yet his fever was still reduced. This new antipyretic, however, received very little interest until the 1940s when scientists rediscovered acetaminophen as the major metabolite of phenacetin and acetanilide.15-17 These findings were significant as phenacetin was associated with nephrotoxicity and acetanilide was found to cause methemoglobinemia, yet the analgesic and antipyretic effects of both of these medications were due to their active metabolite acetaminophen, which did not have either of these toxic effects.16
While the US Food and Drug Administration (FDA) first issued a patent for acetaminophen in 1951, McNeil Laboratories did not begin actively marketing the drug under the brand name Tylenol Children's Elixir® until 1955 as a fever and pain reliever for children.6 A year later it was introduced in the United Kingdom as Panadol® where it is more frequently referred to as paracetamol. Sales of aspirin began to drop in the 1970s and 1980s due to safety concerns in children and certain adult populations, while phenacetin was withdrawn from the US market in 1983 due to unacceptable levels of interstitial nephritis in patients and potential risks of tumorigenicity. Both of these events led to the increased popularity and use of acetaminophen, and with expiration of the FDA patent in 1984 many generic versions and formulations of Tylenol® were produced and currently exist.
Pharmacology
Acetaminophen is thought to act within the central nervous system inhibiting central cyclooxygenase (COX) activity, an enzyme involved in prostaglandin synthesis, resulting in an increased pain threshold. Prostaglandins facilitate the inflammatory response responsible for inflammation, pain, and fever. Specifically, acetaminophen inhibits both central isoforms of the COX enzyme (COX-1 and COX-2) but does not inhibit prostaglandin synthesis in peripheral tissues. This explains acetaminophen's lack of peripheral anti-inflammatory effects.22,23 However, while acetaminophen's exact mechanism of action is still unknown, investigators believe it may involve a specific interaction with a unique COX isozyme, or it may be by inhibition of nitric oxide synthesis or serotonergic pathways, or through an active metabolite influencing cannabinoid receptors.22,24 The mechanism of action may also involve a combination of all of these routes.
The usual adult dose of acetaminophen is 325 mg to 650 mg every 4 to 6 hours, as needed for the treatment of mild pain. While 1000 mg up to four times per day can be administered, it is important not to exceed 1000 mg per dose or 4000 mg in a 24-hour period.6,22 A statement from the American Dental Association (ADA) House of Delegates states that, "Dentists should consider nonsteroidal anti-inflammatory drugs (NSAIDs) as first-line therapy for acute pain management, and for situations in which NSAIDs alone are not effective, the combination of a NSAID with acetaminophen is recommended."25,26 This closely aligns with the prescribing mnemonic "2-4-24" first published in 2010 to help prescribers remember the prescription of choice for acute postoperative orofacial pain.27 This mnemonic refers to a combination of acetaminophen 650 mg to 1000 mg and ibuprofen 600 mg to be administered every 6 hours for 24 hours: "two drugs, in four doses, for 24 hours." A more recent publication further suggests that the anticipated pain level for the patient can also help determine the best postoperative analgesic prescribing regimen.28
While Advil Dual Action® may seem like an ideal postoperative analgesia strategy to improve patient compliance by minimizing the potential pill burden to the patient, there may be shortcomings related to this combination product to consider. Most importantly, the target doses of the "2-4-24" strategy (acetaminophen 650 mg and ibuprofen 600 mg per dose) are impossible to achieve when using the fixed dose tablets of Advil Dual Action (acetaminophen 250 mg and ibuprofen 125 mg per dose). Secondly, if patients attempt to match the optimal analgesic dose of ibuprofen 600 mg using this OTC pain medication, they will greatly exceed the recommended maximum dose of acetaminophen, which is known to have serious health implications, including potential liver toxicity and mortality.
Metabolism and Toxicity
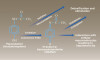
Acetaminophen has an elimination half-life of 2 to 3 hours in healthy adult patients and is renally excreted as the sulfate metabolite (25% to 35%) and the glucuronide conjugate (40% to 65%); less than 5% is excreted as unchanged drug.6,22 Acetaminophen is metabolized in the liver via three separate pathways: glucuronidation, sulfation, and cytochrome P450 (CYP450) oxidation (Figure 1). Glucuronidation and sulfation are the primary routes of metabolism in detoxifying and eliminating the drug from the body, while a minor amount of acetaminophen undergoes oxidative metabolism by isoenzyme CYP2E1 creating the hepatotoxic metabolite, N-acetyl-p-benzoquinoneimine (NAPQI). NAPQI is rapidly conjugated with glutathione to inert metabolites when acetaminophen is administered at therapeutic doses.6,22,24,29,30 Repeated therapeutic doses or supratherapeutic doses of acetaminophen, fasting, and alcoholism can deplete glutathione stores, however, resulting in hepatotoxicity due to increased concentrations of NAPQI in the blood.22,30
Regulations
The hepatotoxic effects of NAPQIs have been well-documented and are the most common cause of acetaminophen toxicity leading to acute liver failure in both the United States and the United Kingdom.31,32 In a combined 5-year study from 22 specialty medical centers in the United States, acetaminophen-induced liver injury led all causes of acute hepatic failure.31 Almost half of the reported cases were due to unintentional overdoses where acetaminophen toxicity occurred as the result of acute overdose or chronic excessive dosing.31 Early nonspecific symptoms of acetaminophen-induced hepatotoxicity include abdominal pain, malaise, nausea/vomiting, and anorexia, which may progress to more severe signs such as jaundice, hepatic encephalopathy, and hepatic necrosis.7,33
Between 1998 and 2003, acetaminophen-related acute liver failure cases increased from 28% of all acute liver failure cases in the United States to 51%.31 From 2000 to 2004, 86% of the 1,600 cases of acute liver failure in the United States were due to unintentional or intentional acetaminophen overdoses.34 The mounting evidence linking acute liver failure with acetaminophen prompted the FDA to assemble its Analgesic Advisory Committee in 2009 to vote on seven specific questions (Table 1).35 This meeting resulted in proposed strategies for acetaminophen dose reduction in order to decrease morbidity and mortality: decrease the maximum single dose from 1000 mg to 650 mg, reduce the maximum daily dose from 4000 mg to 3250 mg, reclassify 500 mg strength tablets to prescription status, and eliminate prescription and nonprescription acetaminophen-combination drugs.35
So far the FDA has instituted only one of the proposed strategies to help address some of the safety concerns with acetaminophen: limit the amount of acetaminophen in combination products to 325 mg per capsule, tablet, or other dosage units (Table 2).35-38 This is particularly interesting as Johnson & Johnson, without being regulated by the FDA to do so, modified the labeling of Tylenol®in 2012 to recommend a maximum adult daily dose of just 3000 mg.39 A timeline of all FDA acetaminophen actions is shown in Figure 2.
In 2021, a group of experts from Australia, Brazil, Canada, Europe, Israel, Scotland, United Kingdom, and United States published a consensus statement in Nature Reviews Endocrinology highlighting the potential risks of acetaminophen use during pregnancy.40 Based on the results of 29 studies investigating the use of acetaminophen, 26 found causative evidence of birth defects, including increased risks of some reproductive, urogenital, and neurodevelopmental disorders. These effects are dependent on the timing of exposure in relation to specific developmental processes and duration as well as dose. Interestingly, the article does not call for a ban on using acetaminophen during pregnancy, but the experts urge both patients and healthcare providers to regard acetaminophen with caution. Oral healthcare providers should be reminded that NSAIDs such as ibuprofen as well as aspirin are not suitable alternatives as they carry an even higher risk of fetotoxicity compared to acetaminophen when administered during pregnancy. The consensus statement also included recommendations for US and European regulatory agencies (ie, the FDA and the European Medicines Agency) to conduct new safety reviews of acetaminophen and update recommendations accordingly. Additional recommendations that providers can give to patients to help keep the use of acetaminophen safe are as follows:
Patients should know the milligrams of acetaminophen in their tablets/capsules. In OTC acetaminophen products, each pill may contain 325 mg or 500 mg of the drug. Extra caution needs to be used when taking 500 mg or 650 mg pills.
Patients should beware of "shotgun" preparations: medications which contain multiple ingredients that include acetaminophen. They should review the labeled ingredients of all OTC as well as prescription products (including cough, cold, and flu remedies and analgesics).
Alcohol intake should be minimal, if done at all. Studies show that people who drink more than three alcohol-containing drinks a day and take acetaminophen are at increased risk of developing hepatotoxicity.41,42 Drinking alcohol causes the liver to convert more of the ingested acetaminophen into toxic byproducts (NAPQI).
Pregnant or breastfeeding patients should avoid ingesting any medications without first consulting their physician. Increasing experimental and epidemiological research suggests that prenatal exposure to acetaminophen might alter fetal development.40
Patients should ask their doctor or pharmacist if any of their medications could interact with acetaminophen, although this is rare. (Alcohol is most noteworthy.)
Patients need to adhere to recommended doses (at present this should be less than 4000 mg a day of acetaminophen from all sources, and less for smaller individuals). When taking acetaminophen, overcome any urge to exceed recommended doses, as most cases of acetaminophen toxicity are unintentional.
Conclusions
Last year, 2021, marked the 70th anniversary of the approval of acetaminophen for use in the United States. It continues to be a popular choice for treating pain and fever, including for special populations such as pregnant and lactating women experiencing mild pain and children with fevers. Acetaminophen is also an important adjunct, endorsed by the ADA, to manage postoperative dental pain in combination with a NSAID such as ibuprofen. Despite the widespread availability and general safe use, acetaminophen toxicity continues to be the most common cause of acute liver failure as the result of chronic excessive dosing or acute overdose, which in most cases is unintentional. Also, new epidemiological and experimental research suggests that prenatal exposure to the drug can alter fetal development, increasing the risks of some reproductive, urogenital, and neurodevelopmental disorders. The general public as well as all healthcare professionals need to remember that sometimes too much of a good thing is too much.
About the Authors
Jason H. Goodchild, DMD
Vice President of Clinical Affairs, Premier Dental Products Co., Plymouth Meeting, Pennsylvania; Associate Clinical Professor, Department of Oral and Maxillofacial Surgery, Creighton University School of Dentistry, Omaha, Nebraska; Adjunct Assistant Professor, Division of Oral Diagnosis, Department of Diagnostic Sciences, Rutgers School of Dental Medicine, New Brunswick, New Jersey
Mark Donaldson, BSP, ACPR, PharmD
Associate Principal, Vizient Pharmacy Advisory Solutions, Irving, Texas; Clinical Professor, School of Pharmacy, University of Montana, Missoula, Montana; Clinical Assistant Professor, School of Dentistry, Oregon Health & Sciences University, Portland, Oregon; Adjunct Professor, Faculty of Dentistry, University of British Columbia, Vancouver, British Columbia; Fellow, American Society of Health-System Pharmacists; Fellow, American College of Healthcare Executives®
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
https://www.chpa.org/our-issues/otc-medicines/acetaminophenReferences
1. Acetaminophen. Consumer Healthcare Products Association website. https://www.chpa.org/our-issues/otc-medicines/acetaminophen. Accessed March 22, 2022.
2. Freo U, Ruocco C, Valerio A, et al. Paracetamol: a review of guideline recommendations. J Clin Med. 2021;10(15):3420.
3. Wongrakpanich S, Wongrakpanich A, Melhado K, Rangaswami J. A comprehensive review of non-steroidal anti-inflammatory drug use in the Elderly. Aging Dis. 2018;9(1):143-150.
4. Blyth FM, Briggs AM, Schneider CH, et al. The global burden of musculoskeletal pain - where to from here? Am J Public Health. 2019;109(1):35-40.
5. Qaseem A, McLean RM, O'Gurek D, et al. Nonpharmacologic and pharmacologic management of acute pain from non-low back, musculoskeletal injuries in adults: a clinical guideline from the American College of Physicians and American Academy of Family Physicians. Ann Intern Med. 2020;173(9):739-748.
6. Tylenol Children's Elixir (acetaminophen) [prescribing information]. October 2015. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/204767s000lbl.pdf. Accessed March 22, 2022.
7. Hashimoto R, Suto M, Tsuji M, et al. Use of antipyretics for preventing febrile seizure recurrence in children: a systematic review and meta-analysis. Eur J Pediatr. 2021;180(4):987-997.
8. Moore RA, Moore N. Paracetamol and pain: the kiloton problem. Eur J Hosp Pharm. 2016;23(4):187-188.
9. Abalos E, Sguassero Y, Gyte GM. Paracetamol/acetaminophen (single administration) for perineal pain in the early postpartum period. Cochrane Database Syst Rev. 2021;1(1):CD008407.
10. Marhofer D, Jaksch W, Aigmüller T, et al. Pain management during pregnancy: an expert-based interdisciplinary consensus recommendation [in German]. Schmerz. 2021;35(6):382-390.
11. Tan E, Braithwaite I, McKinlay CJD, Dalziel SR. Comparison of acetaminophen (paracetamol) with ibuprofen for treatment of fever or pain in children younger than 2 years: a systematic review and meta-analysis. JAMA Netw Open. 2020;3(10):e2022398.
12. Zafeiri A, Mitchell RT, Hay DC, Fowler PA. Over-the-counter analgesics during pregnancy: a comprehensive review of global prevalence and offspring safety. Hum Reprod Update. 2021;27(1):67-95.
13. Donaldson M, Goodchild JH. Pregnancy, breast-feeding and drugs used in dentistry. J Am Dent Assoc. 2012;143(8):858-871.
14. Amundsen S, Nordeng H, Nezvalová-Henriksen K, et al. Pharmacological treatment of migraine during pregnancy and breastfeeding. Nat Rev Neurol. 2015;11(4):209-219.
15. Smith JN, Williams RT. Studies in detoxication; the metabolism of acetanilide in the rabbit. Biochem J. 1948;42(4):538-544.
16. Brodie BB, Axelrod J. The fate of acetanilide in man. J Pharmacol Exp Ther. 1948;94(1):29-38.
17. Smith JN, Williams RT. Studies in detoxication, the metabolism of phenacetin (p-ethoxyacetanilide) in the rabbit and a further observation on acetanilide metabolism. Biochem J. 1949;44(2):239-242.
18. Global Acetaminophen Market 2020 by Manufacturers, Regions, Type and Application, Forecast to 2026. Markets and Research.Biz website. April 2020. https://www.marketsandresearch.biz/report/43101/global-acetaminophen-market-2020-by-manufacturers-regions-type-and-application-forecast-to-2026. Accessed March 22, 2022.
19. Mueller RL, Scheidt S. History of drugs for thrombotic disease. Discovery, development, and directions for the future. Circulation. 1994;89(1):432-449.
20. Brune K, Renner B, Tiegs G. Acetaminophen/paracetamol: a history of errors, failures, and false decisions. Eur J Pain. 2015;19(7):953-965.
21. Morse HN. Ueber eine neue Darstellungsmethode der Acetylamidophenole. Berichte Dtsch Chem Gesell. 1878;11:232-233.
22. McNeil Consumer Healthcare. Tylenol® Professional Product Information. Tylenol® website. https://www.tylenolprofessional.com/resource-library. Accessed March 22, 2022.
23. Anderson BJ. Paracetamol (acetaminophen): mechanisms of action. Paediatr Anaesth. 2008;18(10):915-921.
24. Hersh EV, Dionne, RA. Nonopioid analgesics. In: Dowd FJ, Johnson BS, Mariotti AJ, eds. Pharmacology and Therapeutics for Dentistry. 7th ed. St. Louis, MO: Mosby Elsevier; 2011:257-276.
25. Aminoshariae A, Kulild JC, Donaldson M, Hersh EV. Evidence-based recommendations for analgesic efficacy to treat pain of endodontic origin: a systematic review of randomized controlled trials. J Am Dent Assoc. 2016;147(10):826-839.
26. American Dental Association. Oral analgesics for acute dental pain. ADA website. September 15, 2020. https://www.ada.org/en/member-center/oral-health-topics/oral-analgesics-for-acute-dental-pain. Accessed March 22, 2022.
27. Donaldson M, Goodchild JH. Appropriate analgesic prescribing for the general dentist. Gen Dent. 2010;58(4):291-297.
28. Donaldson M, Goodchild JH. Tramadol: what have we learned in the last 25 years? Gen Dent. 2021;69(4):14-18.
29. Tolman KG. Hepatotoxicity of non-narcotic analgesics. Am J Med. 1998;105(1B):13S-19S.
30. McGill MR, Hinson JA. The development and hepatotoxicity of acetaminophen: reviewing over a century of progress. Drug Metab Rev. 2020;52(4):472-500.
31. Ryder SD, Beckingham IJ. ABC of diseases of liver, pancreas, and biliary system. Other causes of parenchymal liver disease. BMJ. 2001;322(7281):290-292.
32. Larson AM, Polson J, Fontana RJ, et al. Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology. 2005;42(6):1364-1372.
33. Hodgman MJ, Garrard AR. A review of acetaminophen poisoning. Crit Care Clin. 2012;28(4):499-516.
34. Bower WA, Johns M, Margolis HS, et al. Population-based surveillance for acute liver failure [published erratum appears in Am J Gastroenterol. 2008;103(1):255]. Am J Gastroenterol. 2007;102(11):2459-2463.
35. Krenzelok EP. The FDA Acetaminophen Advisory Committee Meeting - what is the future of acetaminophen in the United States? The perspective of a committee member. Clin Toxicol (Phila). 2009;47(8):784-789.
36. Lee WM. Acetaminophen (APAP) hepatotoxicity - isn't it time for APAP to go away? J Hepatol. 2017;67(6):1324-1331.
37. US Food and Drug Administration. FDA drug safety communication: prescription acetaminophen products to be limited to 325 mg per dosage unit; boxed warning will highlight potential for severe liver failure. FDA website. January 13, 2011. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-prescription-acetaminophen-products-be-limited-325-mg-dosage-unit. Accessed March 22, 2022.
38. Dept of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research. Organ-specific warnings: internal analgesic, antipyretic, and antirheumatic drug products for over-the-counter human use: final monograph, final rule. https://www.govinfo.gov/content/pkg/FR-2009-11-25/pdf/E9-28296.pdf. Accessed March 22, 2022.
39. NEJM Journal Watch. Maximum daily dose of Tylenol lowered by manufacturer. NEJM Journal Watch website. August 1, 2011. https://www.jwatch.org/fw201108010000001/2011/08/01/maximum-daily-dose-tylenol-lowered-manufacturer. Accessed March 22, 2022.
40. Bauer AZ, Swan SH, Kriebel D, et al. Paracetamol use during pregnancy - a call for precautionary action. Nat Rev Endocrinol. 2021;17(12):757-766.
41. Thummel KE, Slattery JT, Ro H, et al. Ethanol and production of the hepatotoxic metabolite of acetaminophen in healthy adults. Clin Pharmacol Ther. 2000;67(6):591-599.
42. Florén CH, Thesleff P, Nilsson A. Severe liver damage caused by therapeutic doses of acetaminophen. Acta Med Scand. 1987;222(3):285-288.