You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Autologous blood concentrates (ABCs) are created by centrifugation of a patient's blood for use in various soft- and hard-tissue procedures to enhance healing and tissue regeneration. Platelet-rich plasma (PRP) was the first ABC reported in the literature, dating back to the 1950s.1-4 Decades later, a better understanding of the benefits of ABCs, specifically PRP, would begin to influence treatment and wound healing.
In the 1980s, several reports identified the pivotal role of oxygen in wound healing, recognizing growth factors to promote healing related to the macrophage response to oxygen gradients that secrete wound healing growth factors.5-9 Platelet-derived wound healing factor was introduced clinically first, with development continuing to PRP, which has been utilized for more than two decades to initiate wound healing. Thrombin-released platelets were identified to stimulate collagen synthesis with fibrin eliciting a cellular exudate, which is then followed by angiogenesis. In the early 1990s, Marx and Garg began work on patient-derived blood as a fibrin source for use with osseous grafts to improve product handling, and this fibrin adhesive was first published in 1994.10
Platelets secrete growth factors (platelet-derived growth factors PDGF-AA, PDGF-BB, and PDGF-AB), which stimulate mesenchymal stem cells to replicate hard and soft tissues, osteoblasts to replicate and produce osteoid, endothelial cells to replicate secreting basal lamina for new blood vessels, and fibroblasts to replicate producing collagen. Additionally, transforming growth factors (TGF-β1 and TGF-β2) and bone morphogenic protein are present that stimulate matrix production and guide cell differentiation into bone. Other factors present in autologous blood concentrates include vascular endothelial growth factor, which supports new blood vessel development, and epithelial growth factor, which stimulates migration of the surrounding soft tissue to cover the wounded area and form a basement membrane.
Incorporation of ABCs, such as PRP, into a particulate graft material provides growth factors to accelerate healing of the graft, stimulating the host cells adjacent to the graft to convert into native bone. Accelerated wound closure of the soft tissue when using the patient's own factors as a stimulating mechanism can be expected with PRP-derived membranes.
ABC Fabrication
Platelet-rich plasma is a fraction of the plasma-containing platelets and leukocytes obtained by centrifuging a portion of whole blood at 200 RCF (relative centrifugation force) for 10 minutes. After centrifugation, the upper portion of red blood cells in the test tube is collected, including the buffy coat zone, which adds the leukocyte fraction of the sample to the PRP.11-14 To facilitate and ensure that growth factors present therein can be released at the desired site, a gel is obtained through the addition of autologous thrombin or simply 10% calcium gluconate to the PRP in the tube as it is collected during phlebotomy. This protocol, which is diffused and used internationally, has shown its efficiency in several areas of medicine and dentistry.15-20
In 2001, Choukroun et al developed a simple protocol for obtaining a platelet aggregate similar to PRP using only whole blood, without the addition of anticoagulants.21 The resulting fibrin gel was called platelet-rich fibrin (PRF). Referred to as a second-generation protocol, the Choukroun protocol was very simple and provided for the centrifugation of a whole blood sample, without anticoagulants, immediately following its collection in a serological centrifuge for 10 minutes at 400 RCF. At this centrifugation rate, the platelets rupture at the same time they are stratified by releasing thromboplastin, and at the end of the centrifugation form a fractured clot within the tube, which leaves the heavier portions of the blood at the bottom of the tube and a portion containing plasma, platelets, and leukocytes at the top of the tube. Using this protocol, a clot is obtained without the addition of thrombin or calcium gluconate. This can be utilized to obtain consistent membranes containing fibrin, growth factors, and leukocytes. The membranes thus obtained may be sutured or fractionated into small pieces or added to osseous graft material.22-24 PRP, however, has an advantage of being able to be mixed with bone graft particles or biomaterials to be grafted to form a putty-like consistency, which is not possible with PRF membranes.
From Choukroun's publications and others, several protocols were presented with varying speeds and centrifugation times and which proposed several uses for the obtained platelet aggregate.21-25 Those articles, for the most part, did not explain that g-force was used by limiting RPM (revolutions per minute) and time. It is known that g-force (which is obtained in centrifugation) is dependent on the RPM, amount of time centrifuged, and radius of the centrifuge. Thus, the formula for this calculation is: RCF = 1.12 x radius x (RPM/1000)2.
Various laboratory centrifuges are available on the market. Some consist of a rotational center portion that houses the tubes to be centrifuged, locking them into a fixed angle position (fixed angle centrifuge). In other centrifuges, the tube is placed in a fixed vertical position (for the purpose of reducing the size of the system) and does not allow tube movement during rotation (vertical tube centrifuge). The third type of centrifuge allows the tubes to position in a horizontal direction during centrifugation using a swing bucket with tubes loaded vertically and returning to the vertical position after horizontal centrifugation is completed (swing bucket centrifuge or horizontal centrifuge).
Tube direction during centrifugation also determines how the ABC will be separated within the tube and ultimately the quality of layer separation. A fixed angle centrifuge will result in angled layers within the tube; the desired separation of the layers for extraction from the tube for clinical use in surgical and cosmetic applications will be less ideal. Vertical centrifuge results in vertical layers, making it more difficult to remove the desired layers than those in tubes centrifuged in a fixed angle centrifuge. Horizontal centrifugation results in tubes with horizontal layers, hence allowing for easier extraction from the tube and layers that are more easily discerned. Horizontal centrifugation also yields the thickest and most stable gel layer. Consider that in a fixed angle centrifugation the gel barrier layer is formed at a 45-degree angle to the tube, so the same volume of gel must seal 40% more surface area when compared to a horizontal centrifuge.26
The radius (distance between the central screw of the centrifuge cradle and the tube) can be measured in several ways.It can be measured from the central screw to either the bottom of the tube (maximum radius, Rmax), the middle of the tube (or the second third), or the tube cap (minimum radius, Rmin). This allows three different g-force values to be obtained in the same system, depending on the measured distance and the type of centrifuge being used. If these values are applied to the formula described above, the result will be three different RPMs. These variations were never considered (or reported) in the literature, which can cause confusion for professionals trying to reproduce in their own equipment the protocols proposed by Choukroun et al.21 Finally, another consideration is the amount of blood in each tube; although tubes are uniform in size (volumetric capacity), vacuum tubes do not always draw the same volume of blood. Such data should be further explained in published studies, especially those comparing different protocols, to homogenize the results.25
Novel Protocols
Since an initial publication on the use of bone grafts in 1999,27 endeavors have been made to create protocols that could be reproduced without the need for special equipment with particular characteristics and inputs. Essentially, such protocols would be able to utilize conventional centrifuges with conventional inputs to provide the desired results to the centrifuged ABCs. The protocols presented here can be used with conventional centrifuges, as the instructions meet the technical specifications thereof. These protocols measure the radius of the centrifuge from the center screw to the bottom of the tube. A third-generation protocol, published in 2017, allows for obtaining PRP in liquid form (ie, injectable PRP [I-PRP]), which can be mixed with biomaterials or bone particles to form a compact, malleable, and moldable graft that is referred to as "gummy bone" or bone block.28 These protocols can also be applied to achieve more consistent fabrication of PRF or concentrated growth factor (CGF).
PRP Third-Generation Protocol
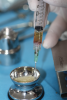
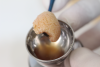
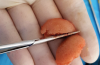
In this third-generation protocol, blood is collected in four 10-ml vacuum-loaded tubes (eg, BD Vacutainer® Red Cap, Becton, Dickinson and Company; Streck BCT® tubes, Streck; AlphaLab tubes, AlphaProMed) without any additives in the tube. The tubes are immediately placed into a conventional hematological centrifuge and rotated at 600 RCF for 3 minutes. After centrifugation, the tubes are removed from the centrifuge, and the blood has now been separated within the tubes, leaving the red blood cells in the lower portion of the tube, a white intermediate portion containing leukocytes and CD34 (cluster of differentiation protocol 34) stem cells, and a yellow upper portion containing platelets and plasma (Figure 1). Two tubes are opened and the entire upper yellow portion along with a small part of the white portion, which remains in a liquid state (I-PRP), is immediately collected with a 5-ml syringe (eg, BD Luer-Lok™, Becton, Dickinson and Company; Hamilton® syringe, Sigma-Aldrich, Inc.; SOL-CARE™, Sol-Millennium Medical Group), and the content is immediately added to the vessel containing the bone graft material to be used (Figure 2). The I-PRP and bone graft particles are set aside for 10 minutes to coalesce (Figure 3), and the result is a congealed mass referred to as "gummy bone" that can be cut to appropriate-sized pieces for the intended application (Figure 4).
The white portion of the centrifuged blood (ie, buffy coat) contains monocytes that are mesenchymal stem cells present in the blood (CD34) and growth factors that are released by platelets that will differentiate osteoblasts. The addition of this white portion was proposed in the authors' initial 1999 protocol27 as was utilization of low-speed centrifugation and a lower g-force.
The other two tubes are placed in a tube rack and allowed to stand for about 10 minutes to allow formation of the PRP membranes. Upon removal of the coagulated portion of the membrane from the tubes that have rested (Figure 5), the layer composed of red cells remains a liquid (non-coagulated) (Figure 6), demonstrating that the platelets were only in the upper portion of the tube where they degranulated and formed the PRP membrane (Figure 7). This result differentiates the end product from liquid-PRF where the platelets are in both the top and bottom of the tube, resulting in a total coagulation. Thus, with this protocol a larger number of trapped platelets can be obtained, forming a higher-volume, better-quality membrane.
The use of this protocol to obtain I-PRP, gummy bone, and PRP membranes that are suturable is an option that is easily clinically obtainable. The protocol produces a moldable bone graft that allows greater stability of the biomaterial used and biologic membranes that enhance soft-tissue and osseous healing related to the patient's own growth factors contained in the resulting materials. The contained growth factors as well as other patient-derived components enhance wound closure and soft-tissue healing.29,30 The literature supports the assertion that, compared with grafts hydrated with saline solution or blood, the resulting PRP-enriched gummy bone is a superior grafting material possessing both osteoconductive and osteoinductive properties while eliciting the least immunological response and improved clinical handling.31,32
PRP Fifth-Generation Protocol
While the third-generation protocol is used to obtain PRP liquid and a subsequent fourth-generation protocol may be utilized to acquire only membranes, a fifth-generation (5G) protocol is aimed at obtaining high-platelet-concentration PRP and autogenous thrombin. This 5G protocol allows PRP to be obtained in which there is a platelet concentration six times higher than that present in the original clot. In protocols where only one whole blood tube is utilized and is centrifuged only once, the result is always a clot that contains approximately 90% of the platelets in the collected blood volume.33 The platelets rupture during centrifugation resulting in a clot at the end or within a few minutes of the end of centrifugation, depending on the g-force used. With the 5G protocol, six tubes of blood are collected during phlebotomy in tubes with an anticoagulant, and platelets are separated in the first centrifugation. This is followed by collection of the platelets from the six tubes along with the plasma, which is then placed in two dry tubes and centrifuged a second time. This results in the platelets being in the bottom of the tube, leaving all the plasma in the upper portion of the tube. Plasma (comprising 50% to 70% of the volume) is removed, and the platelets are resuspended in the remaining plasma, thus creating a higher concentrate containing platelets than that which was in the initial six tubes.
The 5G protocol is described specifically as follows. Blood is collected in six 10-ml vacuum-loaded tubes (eg, BD Vacutainer® Blue Cap, Streck BCT tubes, AlphaLab tubes) with an anticoagulant present in the tubes (10% sodium citrate) and two vacuum-loaded tubes (eg, BD Vacutainer Red Cap, Streck BCT tubes, AlphaLab tubes) without any substance added (Figure 8, A). The tubes are immediately placed in a conventional hematological centrifuge and rotated at a centrifugal force of 150 RCF for 15 minutes. When centrifugation is completed, the blue cap tubes are removed; separation of the blood leaves the red blood cells in the lower portion of the tube, a yellow portion consisting of plasma and platelets, and a white intermediate layer that contains the leukocytes and stem cells (CD34) (Figure 8, B). The tube contents are collected and put into two dry tubes (red cap) in equal volumes to balance the centrifuge and are centrifuged again at 300 RCF for 10 minutes. This procedure moves the platelets, leukocytes, and CD34 stem cells to the bottom of the tube with the plasma remaining in the upper portion of the tube (Figure 8, C).
Thus, 50% to 80% of the plasma has now been collected, with the remainder left in the tube (Figure 8, D). This plasma can be used to obtain a fibrin albumin gel if heated at 75°C for 15 minutes. The remainder in the tube is then gently shaken by hand to resuspend the platelets and obtain concentrated PRP (Figure 8, E).
When removed from the centrifuge the red cap tubes will be coagulated. The yellow portion, which has separated, is removed from these tubes and placed in a sterile dish and allowed to slowly release for 5 to 10 minutes a liquid portion containing a large volume of autogenous thrombin. This liquid is collected in a syringe and will be used to form the PRP gel at the desired time. At the time of gel formation, this thrombin is added to the PRP (10% to 15% in proportion), which may be mixed with bone or biomaterials as previously discussed to form gummy bone.
Discussion
Autologous blood concentrates, specifically the first identified version, PRP, were originally mentioned in the literature more than 60 years ago. Since then, the protocol to fabricate PRP from patient-derived blood has steadily evolved to yield better products with more effective clinical results. As the protocol has advanced, the key has been the development of ABCs with higher concentrates of platelets and other blood components that enhance soft- and hard-tissue healing and, hence, regeneration.
Debate has been ongoing as to whether changes to the fabrication protocol have any effect on or improve the ABC products derived. Unfortunately, preparation procedures for PRP currently are non-standardized. Various protocols, based on distinct centrifugation patterns (rotation speed and time), yield PRPs with different characteristics regarding platelet and growth factor concentrations as well as platelet activation. Multiple PRP studieshave demonstrated that when applied to surgical sites and wounds PRPs are a safe and effective method to promote soft-tissue healing and bone growth.34-36 Numerous protocols have been developed for preparing PRP, with subtle but important differences between them.37
Reports have demonstrated that the double-centrifugation protocol (5G) results in higher platelet concentration,38,39 although leukocytes are not concentrated by this protocol. When compared with the single-centrifugation protocol, this protocol achieves increased platelet activation and aggregation, and TGF-β1 concentration also was reported to be higher.39 A significant, positive correlation between platelet counts and TGF-β1 concentration regarding Pearson's correlation coefficients has been reported.40 Although premature platelet aggregation occurs with the double-centrifugation protocol, it still appears to be an effective method for preparation of PRP resulting in high platelet and TGF-β1 concentrations. While TGF-β1 and PDGF levels are higher in preparations that contain leukocytes compared to leukocyte-poor PRP, platelet concentration may be the most reliable criterion for comparing different preparations.41 This 5G protocol can recover 80% of platelets from whole blood, with an increased platelet concentration of sixfold compared to single centrifugation, and it produced the highest concentration of PDGF-BB in activated fractions.42
The double-centrifugation PRP protocol is a relatively simple and inexpensive method for obtaining platelet-rich plasma. It does not require expensive equipment or a high technical ability by the practitioner or staff and can readily be carried out in a private dental clinical setting.42,43
Conclusion
Numerous studies in the literature support improved soft- and hard-tissue regeneration with the use of autologous blood concentrates. Regeneration can be accelerated with the incorporation of growth factors derived from the patient's blood, and wound closure with advanced healing related to platelet concentrations within PRP-applied products can be achieved at the surgical site. The fifth-generation protocols presented increase the concentration of platelets and growth factors compared to prior centrifugation protocols, thus providing a higher concentration of the desired factors to the site to improve regeneration results.
As the graft material is dehydrated, a sufficient volume of liquid PRP is used to rehydrate the graft and leave some volume of liquid to surround the graft particles. Because the ideal amount of growth factors for a given volume of bone is still unknown, further research is needed to determine what amount is best for each type of graft application. The information and principles discussed in this article may also be applied to other ABCs (ie, PRF and CGF) that clinicians use in their practice and are not confined only to fabrication of PRP. Understanding the types of centrifuges available and their effect on ABC products produced can help the practitioner with decisions regarding centrifuge selection.
About the Authors
Renato Rossi Jr., DMD, MSc, PhD
Director, Oral and Maxillofacial Surgery Residency Program, and Dean, School of Dentistry, University of São Caetano do Sul, São Paulo, Brazil; Private Practice limited to Oral and Maxillofacial Surgery and Oral Pathology, São Paulo, Brazil
Arun K. Garg, DMD
Former Professor of Surgery, Department of Oral and Maxillofacial Surgery, and former Director of Residency Training, University of Miami Leonard M. Miller School of Medicine, Miami, Florida; Private Practice, Miami, Florida
Gregori M. Kurtzman, DDS, MAGD
Former Assistant Clinical Professor, Department of Restorative Dentistry and Endodontics, University of Maryland School of Dentistry; Diplomate, International Congress of Oral Implantologists; Private Practice, Silver Spring, Maryland
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Kingsley CS. Blood coagulation; evidence of an antagonist to factor VI in platelet-rich human plasma. Nature. 1954;173(4407):723-724.
2. Born GV. Evidence for the formation of a labile phospholipoprotein during the clotting of platelet-rich plasma. Nature. 1957;180(4585):546-547.
3. McKay DG, Shapiro SS, Shanberge JN. Alterations in the blood coagulation system induced by bacterial endotoxins. II. In vitro. J Exp Med. 1958;107(3):369-376.
4. Born GV. Changes in the distribution of phosphorus in platelet-rich plasma during clotting. Biochem J. 1958;68(4):695-704.
5. Knighton DR, Silver IA, Hunt TK. Regulation of wound-healing angiogenesis-effect of oxygen gradients and inspired oxygen concentration. Surgery. 1981;90(2):262-270.
6. Knighton DR, Hunt TK, Scheuenstuhl H, et al. Oxygen tension regulates the expression of angiogenesis factor by macrophages. Science. 1983;221(4617):1283-1285.
7. Marx RE, Johnson RP. Studies in the radiobiology of osteoradionecrosis and their clinical significance. Oral Surg Oral Med Oral Pathol. 1987;64(4):379-390.
8. Marx RE, Ehler WJ, Tayapongsak P, Pierce LW. Relationship of oxygen dose to angiogenesis induction in irradiated tissue. Am J Surg. 1990;160(5):519-524.
9. Knighton DR, Hunt TK, Thakral KK, Goodson WH 3rd. Role of platelets and fibrin in the healing sequence: an in vivo study of angiogenesis and collagen synthesis. Ann Surg. 1982;196(4):379-388.
10. Tayapongsak P, O'Brien DA, Monteiro CB, Arceo-Diaz LY. Autologous fibrin adhesive in mandibular reconstruction with particulate cancellous bone and marrow. J Oral Maxillofac Surg. 1994;52(2):161-166.
11. Marx RE, Carlson ER, Eichstaedt RM, et al. Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1998;85(6):638-646.
12. Whitman DH, Berry RL, Green DM. Platelet gel: an autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. J Oral Maxillofac Surg. 1997;55(11):1294-1299.
13. Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14(4):529-535.
14. Rossi R Jr, Leme JJ, Pispico R. Utilização de plasma rico em plaquetas em enxertos ósseos: proposta de um protocolo de obtenção simplificado. Rev Assoc Paul Cir Dent-SBC. 2001;33:5-7.
15. Pretungaro PS. Using platelet-rich plasma to accelerate soft tissue maturation in esthetic periodontal surgery. Compend Contin Educ Dent. 2001;22(9):729-736.
16. Weibrich G, Kleis WK, Hafner G. Growth factor levels in the platelet-rich plasma produced by 2 different methods: curasan-type PRP kit versus PCCS PRP system. Int J Oral Maxillofac Implants. 2002;17(2):184-190.
17. Rossi R Jr, Souza Filho MAP. Uso de trombina autógena em plasma rico em plaquetas - proposta de um protocolo simplificado e de fácil reprodução clínica. ver Pau Odontol. 2004;26(5):4-9.
18. Rossi R Jr, Negreiros RM, Elias FM, Jorge WA. Utilização de plasma rico em plaquetas em enxertos ósseos para reparação de defeitos ósseos. Revista de Odontologia da Universidade Cidade de São Paulo. 2008;20(3):265-300.
19. Yamamiya K, Okuda K, Kawase T, et al. Tissue-engineered cultured periosteum used with platelet-rich plasma and hydroxyapatite in treating human osseous defects. J Periodontol. 2008;79(5):811-818.
20. Chen TL, Lu HJ, Liu GQ, et al. Effect of autologous platelet-rich plasma in combination with bovine porous bone mineral and bio-guide membrane on bone regeneration in mandible bicortical bony defects. J Craniofac Surg. 2014;25(1):215-223.
21. Choukroun J, Adda F, Schoeffler C, Vervelle A. Une opportunité en paro-implantologie: Le PRF. Implantodontie. 2001;42:55-62.
22. Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;
101(3):e37-e44.
23. Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologic features. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101
(3):e45-e50.
24. Su CY, Kuo YP, Tseng YH, et al. In vitro release of growth factors from platelet-rich fibrin (PRF): a proposal to optimize the clinical applications of PRF. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;
108(1):56-61.
25. Miron R, Choukroun J, Ghanaati S. Controversies related to scientific report describing g-forces from studies on platelet-rich fibrin: necessity for standardization of relative centrifugal force values. Int J Growth Factors Stem Cells Dent. 2018;1(3):80-89.
26. Lourenço ES, Alves GG, de Lima Barbosa R, et al. Effects of rotor angle and time after centrifugation on the biological in vitro properties of platelet rich fibrin membranes. J Biomed Mater Res B Appl Biomater. 2021;109(1):60-68.
27. Rossi R Jr, Leme JJ, Pispico R. Uso de plasma rico em plaquetas em enxertos ósseos. Revista da Associação Paulista de Cirurgiões Dentistas de Sbc. 1999;33:5-7.
28. Rossi R Jr, Garg AK, de Souza BR, Rossi AL. Reconstruction of bone defects with platelets rich plasma (PRP) obtained by the bone block technique (third generation) - clinical case report. J Biodentistry and Biomaterials. 2017;7(1).
29. Verma R, Negi G, Kandwal A, et al. Effect of autologous PRP on wound healing in dental regenerative surgeries and its correlation with PDGF levels. Asian J Transfus Sci. 2019;13(1):47-53.
30. Middleton KK, Barro V, Muller B, et al. Evaluation of the effects of platelet-rich plasma (PRP) therapy involved in the healing of sports-related soft tissue injuries. Iowa Orthop J. 2012;32:150-163.
31. Sethi AK, Kar IB, Mohanty T, et al. Use of plasma-enriched demineralized freeze-dried bone matrix in postsurgical jaw defects. Natl J Maxillofac Surg. 2018;9(2):174-183.
32. Baeyens W, Glineur R, Evrard L. The use of platelet concentrates: platelet-rich plasma (PRP) and platelet-rich fibrin (PRF) in bone reconstruction prior to dental implant surgery [in French]. Rev Med Brux. 2010;31(6):521-527.
33. Wagner SJ, Hapip CA, Turgeon A, et al. Influence of apheresis collection device and container on the storage properties of platelets in 90% PAS-5/10% plasma. Blood Transfus. 2019;17(3):210-216.
34. Huang Y, Bornstein MM, Lambrichts I, et al. Platelet-rich plasma for regeneration of neural feedback pathways around dental implants: a concise review and outlook on future possibilities. Int J Oral Sci. 2017;9(1):1-9.
35. Sánchez-Avila RM, Merayo-Lloves J, Fernández ML, et al. Plasma rich in growth factors eye drops to treat secondary ocular surface disorders in patients with glaucoma. Int Med Case Rep J. 2018;11:97-103.
36. Shimizu S, Tsuchiya S, Hirakawa A, et al. Design of a randomized controlled clinical study of tissue-engineered osteogenic materials using bone marrow-derived mesenchymal cells for maxillomandibular bone defects in Japan: the TEOM study protocol. BMC Oral Health. 2019;19(1):69.
37. Davis VL, Abukabda AB, Radio NM, et al. Platelet-rich preparations to improve healing. Part I: workable options for every size practice. J Oral Implantol. 2014;40(4):500-510.
38. El-Husseiny RM, Saleh HM, Moustafa AA, Salem SA. Comparison between single- versus double-spin prepared platelet-rich plasma injection in treatment of female pattern hair loss: clinical effect and relation to vascular endothelial growth factor. Arch Dermatol Res. 2021;313(7):557-566.
39. Seidel SRT, Vendruscolo CP, Moreira JJ, et al. Does double centrifugation lead to premature platelet aggregation and decreased TGF-β1 concentrations in equine platelet-rich plasma? Vet Sci. 2019;6(3):68.
40. Gutiérrez CM, López C, Giraldo CE, Carmona JU. Study of a two-step centrifugation protocol for concentrating cells and growth factors in bovine platelet-rich plasma. Vet Med Int. 2017;2017:1950401.
41. Davis VL, Abukabda AB, Radio NM, et al. Platelet-rich preparations to improve healing. Part II: platelet activation and enrichment, leukocyte inclusion, and other selection criteria. J Oral Implantol. 2014;40(4):511-521.
42. Shin HS, Woo HM, Kang BJ. Optimisation of a double-centrifugation method for preparation of canine platelet-rich plasma. BMC Vet Res. 2017;13(1):198.
43. Perazzi A, Busetto R, Martinello T, et al. Description of a double centrifugation tube method for concentrating canine platelets. BMC Vet Res. 2013;9:146.