You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Adrenal insufficiency is a relatively rare pathologic condition characterized by a physiologically reduced level of glucocorticoid, mineralocorticoid, and androgen production and/or effectiveness by the malfunctioning hypothalamic-pituitary-adrenal (HPA) axis.1-3 Disease forms may be classified as primary, secondary, or tertiary depending on the origin of the disorder, resulting from injury or dysfunction in the adrenal cortex, anterior pituitary, or hypothalamus, respectively.1,2 Clinical symptoms of adrenal insufficiency most often manifest mildly as weakness, anorexia, fatigue, weight loss, abdominal pain, orthostatic hypotension, salt craving, and, in cases of primary insufficiency, hyperpigmentation of the skin.3-5 A more comprehensive list of symptoms is provided in Table 1.6 When properly treated, patients can expect to live a normal lifespan; however, if left untreated, the disease may lead to a life-threatening shock condition requiring immediate medical attention.1
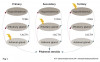
The prevalence of adrenal insufficiencies has been estimated to be between 82 and 144 cases per million for primary insufficiencies and 150 to 280 cases per million for secondary insufficiencies.1,7 Tertiary insufficiencies are much rarer. Since the underlying causes of adrenal insufficiencies are so diverse, no distinct population of individuals has been identified as having an increased risk of developing the disorder.1 Primary insufficiencies have been shown to present at any age, including in children, yet clinical manifestation is most often seen between the fourth and fifth decades of life.2,8 Clinical symptoms of secondary insufficiencies have been shown to present most commonly in the sixth decade of life.1,8 Both primary and secondary disorders affect male and female genders with a slightly increased frequency among women.2 A schematic of the pathological origin of each of the three types of adrenal insufficiency is shown in Figure 1.2
Anatomy and Physiology of the Adrenal Gland
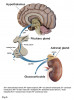
The human adrenal glands are small, paired, endocrine glands located in the posterior abdomen above the left and right kidney, respectively.9 They are responsible for producing and releasing critical catecholamine hormones, including glucocorticoid, mineralocorticoid, and androgen hormones into the bloodstream. The anatomical structure consists of a connective tissue capsule, an outer cortex, and an inner medulla as depicted in Figure 2.10 The cortex is divided functionally into three layers, each responsible for producing and secreting a different hormone.3,9,10 The most superficial layer (zona glomerulosa) produces mineralocorticoids such as aldosterone, the middle layer (zona fasciculata) produces corticosteroids such as cortisol, and the deepest layer (zona reticularis) produces androgen hormones. The medulla, which lies at the heart of the gland, contains chromaffin cells and is responsible for producing and secreting catecholamines, such as adrenaline, in periods of stress.3,9,10 Blood enters and exits the glands through the hilum; blood is supplied by the superior, middle, and inferior arteries and drained by the right and left adrenal veins.9
Hypothalamic-Pituitary-Adrenal Axis and Hormone Regulation
Release of adrenal hormones is tightly controlled by both the hypothalamus and anterior pituitary through the HPA axis and is heavily integrated into the human stress response.11,12 Hypophysiotropic neurons found within the paraventricular nucleus of the hypothalamus are responsible for synthesizing and releasing corticotropin-releasing factor/hormone (CRH), which serves as the principal regulator of the HPA axis.11,12 A system of hypophysial portal vessels exists, connecting the hypothalamus to the anterior pituitary, which allows CRH to be secreted directly into the anterior pituitary in times of stress.11 Binding of CRH within the anterior pituitary initiates the release of adrenocorticotropic hormone (ACTH), which is secreted into systemic circulation.11 ACTH travels to the adrenal cortex where the zona fasciculata is its principal target, stimulating synthesis and release of glucocorticoids into the body.11,13,14
A schematic of the HPA axis and its regulatory mechanisms is provided in Figure 3.11 The body relies on tight regulation of the HPA axis through numerous pathways of both endocrine and neuronal stimulation.11,15 Endocrine regulation includes feedback inhibition by circulating glucocorticoids and CRH binding proteins.11,16 Neuronal regulation is much more complex, involving stimulation from the brain stem, hypothalamus, and limbic system.11,17
Adrenal Hormones
As previously stated, the adrenal cortex is responsible for producing and secreting critical hormones throughout the human body. Each hormone plays a role in essential functions of the body, including but not limited to metabolism, stress response, immune function, and blood pressure regulation.1,11,18 Over- or underproduction of any of the major hormones often results in pathology.
Mineralocorticoids-The adrenal cortex produces aldosterone, a mineralocorticoid heavily involved in sodium balance, blood volume, and blood pressure regulation.18,19 It plays a primary role in the renin angiotensin aldosterone system resulting in action on the kidney's distal convoluted tubule to reabsorb sodium and excrete potassium.18,19
Glucocorticoids-The most notable glucocorticoid found in the human body is cortisol, which is produced in the adrenal cortex.1,2,18 Cortisol's importance within the body cannot be overstated as it functions in metabolism, immune response, and inflammation among other areas.18 Its effects on metabolism are numerous, but it primarily serves to increase circulating glucose levels by mobilizing amino acids and stimulating gluconeogenesis in the liver.20,21 It has also been shown to mobilize free fatty acids, providing an alternate energy source.20,21 Besides blood glucose regulation, cortisol acts to suppress the human immune system and has been found to be a potent anti-inflammatory.21
Adrenal androgens-Androgens produced in the zona reticularis are weak in nature and primarily dehydroepiandrosterone (DHEA), a steroid precursor that serves as a metabolic intermediate to produce more potent male and female sex hormones, including testosterone and estrogen, respectively.20,22
Catecholamines-The adrenal medullae are responsible for producing most of the adrenaline found within the human body yet only a small amount of noradrenaline.23 Effects of said catecholamines include increasing heart rate and blood pressure in response to stress.23
Causes and Pathophysiology of Primary Adrenal Insufficiency
Primary adrenal insufficiencies are those characterized by dysfunction of the adrenal gland itself. Causes may be autoimmune, genetic, traumatic, infectious, or drug-induced in origin.2
Autoimmune adrenalitis (Addison's disease)-The most common form of adrenal insufficiency affecting roughly 80% of adrenal insufficiency patients is of autoimmune origin and is often referred to as Addison's disease since it was first described by Thomas Addison in the mid-19th century.1,2 This condition affects all age groups and genders. It may manifest acutely or chronically and is characterized by cell-mediated destruction of the adrenal cortex through immune antibodies targeting 21-hydroxylase, a steroid involved in the biosynthesis of aldosterone and cortisol.1,2,24-27 Various genetic factors have been identified as playing a role in this disease process, including DR3-DQ2 and DR4-DQ8, a group of major histocompatibility complex haplotypes.24-27
Adrenoleukodystrophy-This is a rare X-linked recessive disorder resulting in mutation of a gene responsible for beta-oxidation of very long chain fatty acids. Buildup of said fatty acids in organs of the body, including the adrenal cortex, leads to clinical pathology.2,28
Hemorrhagic infarction-Hemorrhage and adrenal vein thrombosis may cause serious damage to the adrenal glands leading to adrenal insufficiency.2 Thrombocytopenia, among many other coagulopathies, have been found to cause hemorrhage and thrombosis of the adrenal vessels.29,30 Hemorrhagic infarction has been associated with meningococcemia and Pseudomonas aeruginosa infections.31
Infectious adrenalitis-The adrenal glands are susceptible to attack from a number of infectious agents.2 Tuberculosis, human immunodeficiency virus (HIV)-associated infections, and disseminated fungal infections have all been cited to cause infectious adrenalitis.32-34
Drug-induced insufficiency-There are several pharmacodynamic mechanisms through which drugs may cause adrenal insufficiency. Those that inhibit cortisol biosynthesis include aminoglutethimide, etomidate, ketoconazole, and metyrapone.35-37 Drugs that increase cortisol and synthetic glucocorticoid metabolism include phenytoin, barbiturates, and rifampicin and are particularly dangerous in patients with limited adrenal reserve or those on glucocorticoid replacement therapies.38
Drug withdrawal has also shown to be a common cause of adrenal insufficiencies. Using high-dose corticosteroids for a prolonged period of time may ultimately suppress the adrenal glands by downregulating the production and release of CRH and ACTH.39 In times of prolonged suppression, the adrenal glands may shrink in size, and while recovering, the patient is in a state of increased susceptibility to adrenal insufficiencies, especially in times of stress and illness.39,40
Pathophysiology of primary adrenal insufficiencies is due to loss of glandular tissue and function itself. That being said, clinical manifestations of diseased states are not noticeable until roughly 90% of adrenocortical tissue is lost.2,41 The destruction of the adrenal glands occurs gradually through distinctly complex yet complementary molecular mechanisms, including cytotoxic effects of lymphocytes, interferons, lymphotoxins, autoantibodies, the complement system, inflammatory cytokines, and free radicals.42 Initially, adrenal reserve is enough to maintain basal steroid levels while forfeiting the ability to react adequately to stress.41 Any stressor may have the ability to cause acute adrenal insufficiency due to the gland's inability to adapt and secrete adequate steroid levels. As time and tissue loss progress, basal levels are diminished and negative feedback inhibition results in the overproduction of ACTH, which is responsible for the aforementioned hyperpigmentation seen in primary adrenal insufficiency patients.41
Causes and Pathophysiology of Secondary and Tertiary Adrenal Insufficiencies
Secondary adrenal insufficiencies are those characterized by dysfunction of the anterior pituitary gland and altered ACTH production and release. Dysfunction can be a result of space-occupying lesions or genetic disorders.43 Most commonly, secondary insufficiency is a result of pituitary tumors or trauma.44 Pituitary surgery, irradiation, infections, pituitary apoplexy and underlying diseases, and genetically mutated transcription factors responsible for pituitary development have also been shown to cause pituitary dysfunction leading to adrenal insufficiency.2,45 Tertiary adrenal insufficiencies are those characterized by dysfunction within the hypothalamus.1,2 The same causes apply to tertiary pathologies with tumors and trauma being most common. Surgery, irradiation, and infections may also cause dysfunction. Glucocorticoid therapy or hypersecretion of endogenous glucocorticoids in response to adrenal hyperactivity (Cushing's disease) may also cause insufficiency by altering the HPA axis.45 Pathophysiology of secondary and tertiary insufficiencies are a result of deficient ACTH release.41 This leads to a reduction of cortisol and adrenal androgen release, while production and release of aldosterone remains unchanged.
Early stages of the disease are characterized by maintained basal secretion levels and reduced secretion in response to stress. Over time, as basal secretion becomes increasingly impaired, atrophy of the gland occurs within the cortex in the areas of the zona fasciculata and reticularis, leaving basal aldosterone levels unaffected.41
Medical Management of Adrenal Insufficiency
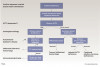
Management of adrenal insufficiency patients begins with diagnosis. Diagnosing adrenal insufficiency is usually a four-step process beginning with a series of tests that includes the measurement of blood cortisol levels.20,46 Once insufficiency is confirmed, the defect type must be determined as either primary, secondary, or tertiary; this is most often confirmed with a corticotropin (ACTH) stimulation test.20,46,47 If ACTH levels are high, primary insufficiency is suspected, whereas low ACTH levels would indicate secondary/tertiary insufficiency.
Once the level of the defect is known, further testing is performed to better understand the cause of the defect and any associated pathologies that may need to be treated. Such determinations are made in consideration with the patient's age, gender, and comorbidities while further blood testing and imaging tests, including chest x-rays and abdominal CT scans, may also be undertaken.20,46 Figure 4 outlines a flow chart that may be followed for diagnosis of adrenal insufficiency.46
The medical management of adrenal insufficiency varies depending on the type of insufficiency and whether conditions are chronic or acute; however, hormone replacement therapy underlies all treatment plans. The hormone replacement therapies discussed in the following sections are highlighted in greater detail in Table 2. The goal of glucocorticoid replacement therapy is to eliminate symptoms of glucocorticoid insufficiency and prevent the occurrence of an adrenal crisis.
Management of Chronic Primary Adrenal Insufficiency
Treatment of primary insufficiencies requires daily doses of both glucocorticoid and mineralocorticoid hormone pills.2,20,46,48 There is no cure for this disease and the treatment is required for life to stabilize functional and physiological hormone levels and relieve any presenting symptoms.2,20 Androgen replacement therapy is sometimes recommended as well depending on the patient. There are several synthetic glucocorticoids for clinicians to prescribe patients that mimic the action of cortisol within the body. The prescribing clinician should work closely with the patient to determine which replacement therapy is both the most effective treatment and the one with the least accompanying side effects.2,20 Synthetic glucocorticoids vary in duration of action and potency.49
The most commonly prescribed drug is hydrocortisone, a short-acting synthetic glucocorticoid given in two to three doses per day with the largest dose in the morning to mimic the circadian rhythm that physiologically regulates cortisol release within the body.2,20,46 The variety of doses in which hydrocortisone is prescribed is also an advantage over longer-acting drugs such as prednisolone, which is offered in a more limited dosing range as it allows for ease of dose adjustment if the patient is being under- or overtreated.20,46,50,51 Longer-acting drugs may also be avoided to minimize the risk of glucocorticoid excess, which manifests as loss of bone density and lean body mass.2 However, longer-acting options can be beneficial for the less-compliant patient as typically only one pill per day is required.20,46
Drug monitoring is very important in treating these patients as symptoms such as weight gain and/or development of a puffy face are signs of overtreatment and indicative of the need for a lower dose.20 Dosing these patients is related to body weight and age, with younger and smaller individuals requiring a lower dose and vice versa.20,46 Dose adjustments are relatively common among this patient population; however, the nausea and fatigue commonly seen at the onset of treatment is usually overcome within days and is unrelated to dose.20,46,50,51 Mineralocorticoid replacement therapy is much less variable and most often consists of the use of the oral synthetic mineralocorticoid, fludrocortisone.2,20,46 Dosing for mineralocorticoid replacement is tailored to manage the patient's blood pressure and fluid imbalance.20,46,50,51 The patient should not experience dizziness and/or light-headedness when standing up, and clinicians should observe closely for any signs and symptoms that may point to overtreatment, such as swelling in the ankles.20 If such signs are noticed, the clinician should expect hypertension and low blood potassium levels and dose reduction is indicated.20,50,51
Androgen replacement is most often indicated for female primary insufficiency patients in order to improve overall sense of well-being and libido.2,20 A daily dose of DHEA is most commonly prescribed for these patients and has shown to enhance mood and general well-being among patients; however, potential side effects of acne, deepened voice, and facial hair may occur.2,20,52 Use of this drug must be cautioned due to the limited research associated with it and regulation concerns.
Management of Chronic Secondary/Tertiary Adrenal Insufficiency
Management of secondary and tertiary insufficiencies differ from primary treatment in that only glucocorticoid replacement therapy is warranted, because the adrenal gland is still able to produce functional levels of aldosterone and androgens.2 The same glucocorticoids listed in Table 2 (compendiumce.com/go/2115) are available and prescribed for this patient population. Frequently, dosing is adjusted at the onset of treatment.2 In some cases, therapy may include the replacement of other deficient pituitary or hypothalamic hormones.2
Management of Acute Adrenal Crisis
Acute adrenal crisis is a life-threatening medical emergency that must be addressed immediately.1,2,20,46,53 The most common cause of such crises is corticosteroid withdrawal.20,53 Other causes include acute exacerbation of chronic insufficiencies, often precipitated by surgical stressors and sepsis, as well as acute adrenal hemorrhage (potentially as an adverse consequence of anticoagulant treatment).1,2,53
Signs and symptoms of adrenal crisis include nausea and vomiting, hypotension, hypoglycemia, hyponatremia, hyperkalemia, loss of consciousness, cardiovascular collapse, and death.20,53 Treatment must be initiated immediately with an emergency injection of hydrocortisone into the thigh following any emergency precaution such as a major injury with substantial blood loss or visible signs of adrenal crisis.2,20,46 Once medical personnel are on the scene, several liters of saline solution are given to correct fluid and electrolyte imbalances in conjunction with an intravenous injection of glucocorticoid as a continuous infusion or a bolus roughly every 6 to 8 hours.2,20,39,53 Once the situation is under control, a maintenance glucocorticoid dose and, if necessary, mineralocorticoid dose is determined.2,20,53
Patient Education
As with all diseases, patient education and understanding are key elements in treating adrenal insufficiencies. Patients must be equipped to assume responsibility of their own medical management in the absence of health professionals to avoid acute crises and other medical emergencies. Patients should always have a steroid card on hand as well as identification jewelry to assist medical personnel in cases of lost consciousness.2,20,46,53 Oral hydrocortisone tablets, injectable hydrocortisone, and a sterile needle and syringe should always be available, and parenteral administration training should be undertaken by all partners and family members.20,46,53 Patients should always be cognizant of the need to consult their doctor regarding necessary dosing adjustment instructions in cases of intercurrent illnesses to avoid adrenal crises.46,53
Oral Manifestations and Dental Management of Adrenal Insufficiencies
Dentists have a responsibility to understand and identify any signs and symptoms, both internal and external to the oral cavity, that may suggest their patient has an underlying adrenal insufficiency. The dentist is one of a select group of medical professionals that patients see on a consistent basis and may be the first to suspect an undiagnosed medical condition in a patient. If the dentist suspects any of the aforementioned symptoms listed in Table 1, immediate referral to a doctor is advised.
Oral manifestations of adrenal insufficiency are limited and selective to the type of presenting insufficiency. In primary adrenal insufficiency, diffuse or focal brown macular pigmentation of the oral mucosa often precedes the extraoral pigmentation commonly seen in these patients.54 The most common area of pigmentation is the buccal mucosa, however it may also manifest on the tongue, lips, gingiva, and palate.54 Mouth sores, including but not limited to chronic mucocutaneous oral candidiasis and oral hairy leukoplakia, may also be found in primary insufficiency patients and in those taking prolonged inhaled corticosteroid therapy.54,55 In secondary insufficiency patients, dentists may observe painless, blood-filled blisters in the oral cavity called angina bullosa hemorrhagica.54,55 These are only found, however, in those patients undergoing chronic use of inhaled corticosteroid therapy for intercurrent diseases, including asthma and chronic obstructive pulmonary disease.54,55
For dentists, the biggest implication on oral management of patients with adrenal insufficiency is the patient's compromised response to stressful stimuli.48,54,56 Stress in the dental office may precipitate during either non-invasive or invasive procedures, ranging from standard hygiene appointments to complex surgical procedures, depending on the patient's dental anxiety level.48,54,56,57 Before treatment, medical consults are advised for all suspected or known adrenal insufficiency patients.54,57 Medical clearance is required to deem both the patient's tolerance level at any specific time as well as the need for corticosteroid replacement therapy prior to treatment.54 Antibiotic prophylaxis typically is not required for these patients; however, it should be considered for patients who are deemed to be at risk for immunosuppression, often due to prolonged corticosteroid use. If the patient has been deemed to have been undiagnosed or inadequately treated for their insufficiency, is medically unstable, or has not complied with the pre-medication indicated by their physician, postponing treatment is recommended.54
The primary goal in treating these patients is preventing an acute adrenal crisis.48,54,56,57 Patients should present to the office with their identification jewelry and supplementary hydrocortisone tablets and solution as well as a sterile needle and syringe ready for use if needed.2,20,46,53 As previously mentioned, advisory from the patient's physician as to the need for medical supplementation prior to treatment is required and is affected by the degree of adrenal suppression, the patient's anxiety as it relates to dental treatment, and the type of treatment involved.54 Appointments generally should be scheduled in the morning when the patient is less tired and susceptible to stress, which coincides with the time of greatest endogenous cortisol release and dose of glucocorticoid replacement.48,54,57
Stress reduction protocols should be in place and the dentist should make a concerted effort to reduce the patient's stress as much as possible throughout the appointment.54,58 Altering chair position is only warranted for those patients who have experienced chronic adrenal insufficiency for long time periods and suffer from postural hypotension.54 Dental procedures should be supplemented with the use of a long-acting dental anesthetic, and plans to properly manage postoperative pain should be made.48,54 Special consideration should be given to the use of epinephrine in these patients due to the hormone's potential to stimulate the sympathetic nervous system, inducing physiologic and psychogenic responses such as increasing heart rate, blood pressure, perspiration, nervousness, and irritability.59-61 Even minor, transient epinephrine-related reactions have the capacity to induce nervousness and stress, especially given the increase in circulating catecholamines in patients taking hormone supplementation and those anxiously awaiting forthcoming anesthetic injections.59,62 Managing patient expectation prior to the use of epinephrine is essential in maintaining lower levels of circulating catecholamines and dampening the stress-induced sympathetic response.59 The clinician should prepare the patient in advance of the injection and outline any potential minor adverse reactions the patient may experience after anesthetic delivery. Should an epinephrine-related medical emergency arise following injection, the practitioner and staff should be equipped to manage the situation effectively.
Because prolonged glucocorticoid therapy has shown to thin bones, extra caution should be taken during surgery to avoid iatrogenic fracture.48,54,58 Patients undergoing surgical dental treatment should have their heart rate and blood pressure monitored at frequent, regular intervals.54,56,58 Synthetic glucocorticoids have shown to interact with the drugs and foods listed in Table 3, and a thorough medical and pharmacological history should be taken prior to treating these patients.54,63 Finally, it should be noted that patients with adrenal insufficiency may experience impaired/delayed wound healing and increased susceptibility to infections.54,56,57 A comprehensive follow-up with these patients following treatment is recommended.
Conclusion
Adrenal insufficiencies are a relatively uncommon group of diseases. However, should patients with these conditions present for dental care, understanding how to identify symptoms and treat the patient appropriately in the dental setting is critically important. Routine dental procedures could be enough to precipitate an adrenal crisis; therefore, minimizing stress and being prepared to respond to a medical emergency is an imperative responsibility of the dentist.
About the Authors
David Carmona, BSc (Hons)
Fourth-Year Dental Student, Faculty of Dentistry, University of Toronto, Toronto, Ontario, Canada
Aviv Ouanounou, BSc, MSc, DDS
Associate Professor, Department of Clinical Sciences (Pharmacology and Preventive Dentistry), Faculty of Dentistry, University of Toronto, Toronto, Ontario, Canada; Fellow, International College of Dentists; Fellow, American College of Dentists; Fellow, International Congress of Oral Implantologists
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Huecker MR, Bhutta BS, Dominique E. Adrenal insufficiency. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2021. http://www.ncbi.nlm.nih.gov/books/NBK441832/. Accessed June 22, 2021.
2. Nicolaides NC, Chrousos GP, Charmandari E. Adrenal insufficiency. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc.; 2000. http://www.ncbi.nlm.nih.gov/books/NBK279083/. Accessed June 22, 2021.
3. Martin-Grace J, Dineen R, Sherlock M, Thompson CJ. Adrenal insufficiency: physiology, clinical presentation and diagnostic challenges. Clin Chim Acta. 2020;505:78-91.
4. Addison's disease - Symptoms and causes. Mayo Clinic website. https://www.mayoclinic.org/diseases-conditions/addisons-disease/symptoms-causes/syc-20350293. Accessed June 22, 2021.
5. Definition and Facts of Adrenal Insufficiency & Addison's Disease. National Institute of Diabetes and Digestive and Kidney Diseases website. https://www.niddk.nih.gov/health-information/endocrine-diseases/adrenal-insufficiency-addisons-disease/definition-facts. Accessed June 22, 2021.
6. Adrenal Insufficiency (Addison's Disease). Johns Hopkins Medicine website. https://www.hopkinsmedicine.org/health/conditions-and-diseases/underactive-adrenal-glands--addisons-disease. Accessed June 22, 2021.
7. Chabre O, Goichot B, Zenaty D, Bertherat J. Group 1. Epidemiology of primary and secondary adrenal insufficiency: prevalence and incidence, acute adrenal insufficiency, long-term morbidity and mortality. Ann Endocrinol (Paris). 2017;78(6):490-494.
8. Neary N, Nieman L. Adrenal insufficiency: etiology, diagnosis and treatment. Curr Opin Endocrinol Diabetes Obes. 2010;17(3):217-223.
9. Avisse C, Marcus C, Patey M, et al. Surgical anatomy and embryology of the adrenal glands. Surg Clin North Am. 2000;80(1):403-415.
10. The Adrenal Glands - Anatomical Location and Relations, Anatomical Structure. TeachMeAnatomy website. https://teachmeanatomy.info/abdomen/viscera/adrenal-glands/. Accessed June 22, 2021.
11. Smith SM, Vale WW. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci. 2006;
8(4):383-395.
12. Rivier C, Vale W. Modulation of stress-induced ACTH release by corticotropin-releasing factor, catecholamines and vasopressin. Nature. 1983;305(5932):325-327.
13. Bamberger CM, Schulte HM, Chrousos GP. Molecular determinants of glucocorticoid receptor function and tissue sensitivity to glucocorticoids. Endocr Rev. 1996;17(3):245-261.
14. Munck A, Guyre PM, Holbrook NJ. Physiological functions of glucocorticoids in stress and their relation to pharmacological actions. Endocr Rev. 1984;5(1):25-44.
15. Carrasco GA, Van de Kar LD. Neuroendocrine pharmacology of stress. Eur J Pharmacol. 2003;463(1-3):235-272.
16. Keller-Wood ME, Dallman MF. Corticosteroid inhibition of ACTH secretion. Endocr Rev. 1984;5(1):1-24.
17. McEwen BS. The neurobiology of stress: from serendipity to clinical relevance. Brain Res. 2000;886(1-2):172-189.
18. Kendall EC. Hormones of the adrenal cortex in health and disease. Proc Am Philos Soc. 1953;97(1):8-11.
19. Adrenal gland hormones. Canadian Cancer Society website. https://www.cancer.ca:443/en/cancer-information/cancer-type/adrenal-gland/adrenal-gland-cancer/adrenal-gland-hormones/?region=on. Accessed June 22, 2021.
20. Patient education: Adrenal insufficiency (Addison's disease) (Beyond the Basics). UpToDate® website. https://www.uptodate.com/contents/adrenal-insufficiency-addisons-disease-beyond-the-basics#H25. Accessed June 22, 2021.
21. Boron WF, Boulpaep EL. Medical Physiology, 2e Updated Edition E-Book, 2nd ed. Saunders; 2012. https://www.mea.elsevierhealth.com/medical-physiology-2e-updated-edition-e-book-9781455711819.html#description. Accessed June 22, 2021.
22. Hall JE. Guyton and Hall Textbook of Medical Physiology. 12th ed. Saunders; 2011. https://evolve.elsevier.com/cs/product/9780323265140?role=student. Accessed June 22, 2021.
23. Colledge NR, Walker BR, Ralston S. Davidson's Principles and Practice of Medicine. 21st ed. Churchill Livingstone/Elsevier; 2010.
24. Erichsen MM, Løvås K, Skinningsrud B, Wolff AB, et al. Clinical, immunological, and genetic features of autoimmune primary adrenal insufficiency: observations from a Norwegian registry. J Clin Endocrinol Metab. 2009;94(12):4882-4890.
25. Mitchell AL, Pearce SHS. Autoimmune Addison disease: pathophysiology and genetic complexity. Nat Rev Endocrinol. 2012;8(5):306-316.
26. Husebye E, Løvås K. Pathogenesis of primary adrenal insufficiency. Best Pract Res Clin Endocrinol Metab. 2009;23(2):147-157.
27. Napier C, Pearce SHS. Autoimmune Addison's disease. Presse Med. 2012;
41(12 P 2):e626-e635.
28. Bornstein SR. Predisposing factors for adrenal insufficiency. N Engl J Med. 2009;360(22):2328-2339.
29. Rao RH, Vagnucci AH, Amico JA. Bilateral massive adrenal hemorrhage: early recognition and treatment. Ann Intern Med. 1989;110(3):227-235.
30. Rao RH. Bilateral massive adrenal hemorrhage. Med Clin North Am. 1995;79(1):107-129.
31. Caron P, Chabannier MH, Cambus JP, et al. Definitive adrenal insufficiency due to bilateral adrenal hemorrhage and primary antiphospholipid syndrome. J Clin Endocrinol Metab. 1998;83(5):1437-1439.
32. Bhatia E, Jain SK, Gupta RK, Pandey R. Tuberculous Addison's disease: lack of normalization of adrenocortical function after anti-tuberculous chemotherapy. Clin Endocrinol (Oxf). 1998;48(3):355-359.
33. Walker BF, Gunthel CJ, Bryan JA, et al. Disseminated cryptococcosis in an apparently normal host presenting as primary adrenal insufficiency: diagnosis by fine needle aspiration. Am J Med. 1989;86(6 Pt 1):715-717.
34. Norbiato G, Galli M, Righini V, Moroni M. The syndrome of acquired glucocorticoid resistance in HIV infection. Baillieres Clin Endocrinol Metab. 1994;8(4):777-787.
35. Wagner RL, White PF, Kan PB, et al. Inhibition of adrenal steroidogenesis by the anesthetic etomidate. N Engl J Med. 1984;310(22):1415-1421.
36. Sonino N. The use of ketoconazole as an inhibitor of steroid production. N Engl J Med. 1987;317(13):812-818.
37. Schöneshöfer M, Claus M. Multiple-sites of inhibition by intravenous metyrapone of human adrenal steroidogenesis. Acta Endocrinol (Copenh). 1985;109(3):378-385.
38. Elias AN, Gwinup G. Effects of some clinically encountered drugs on steroid synthesis and degradation. Metabolism. 1980;29(6):582-595.
39. Nicolaides NC, Pavlaki AN, Alexandra MA, Chrousos GP. Glucocorticoid therapy and adrenal suppression. In: Feingold KR, Anawalt B, Boyce A, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc.; 2000. http://www.ncbi.nlm.nih.gov/books/NBK279156/. Accessed June 22, 2021.
40. Davis CP, Shiel WC Jr. Steroid Drug Withdrawal Symptoms, Treatment, Cure, and Prevention. MedicineNet website. October 22, 2019. https://www.medicinenet.com/steroid_withdrawal/article.htm. Accessed June 22, 2021.
41. Carroll TB, Aron DC, Findling JW, Tyrrell JB. Glucocorticoids and adrenal androgens. In: Gardner DG, Shoback D, eds. Greenspan's Basic & Clinical Endocrinology. 10th ed. New York, NY: McGraw-Hill Education; 2017:chap 9.
42. Bratland E, Husebye ES. Cellular immunity and immunopathology in autoimmune Addison's disease. Mol Cell Endocrinol. 2011;336(1-2):180-190.
43. Melmed S, Polonsky K, Reed Larsen P, Kronenberg H. Williams Textbook of Endocrinology. 12th ed. Saunders; 2011. https://www.elsevier.com/books/williams-textbook-of-endocrinology/melmed/978-1-4377-0324-5. Accessed June 22, 2021.
44. Raff H, Sharma ST, Nieman LK. Physiological basis for the etiology, diagnosis, and treatment of adrenal disorders: Cushing's syndrome, adrenal insufficiency, and congenital adrenal hyperplasia. Compr Physiol. 2014;4(2):739-769.
45. Charmandari E, Nicolaides NC, Chrousos GP. Adrenal insufficiency. Lancet. 2014;383(9935):2152-2167.
46. Pazderska A, Pearce SH. Adrenal insufficiency - recognition and management. Clin Med (Lond). 2017;17(3):258-262.
47. Dickstein G, Shechner C, Nicholson WE, et al. Adrenocorticotropin stimulation test: effects of basal cortisol level, time of day, and suggested new sensitive low dose test. J Clin Endocrinol Metab. 1991;72(4):773-778.
48. Calabria JL, Grau D, Silvestre J, Hernández-Mijares A. Manejo del paciente con insuficiencia suprarrenal en la clínica odontológica. Med Oral. 2003;8:207-214.
49. Katzung BG. Basic & Clinical Pharmacology. 14th ed. McGraw-Hill Medical; 2018. https://accessmedicine.mhmedical.com/book.aspx?bookID=2249. Accessed June 22, 2021.
50. Arlt W. The approach to the adult with newly diagnosed adrenal insufficiency. J Clin Endocrinol Metab. 2009;94(4):1059-1067.
51. Løvås K, Husebye ES. Replacement therapy for Addison's disease: recent developments. Expert Opin Investig Drugs. 2008;17(4):497-509.
52. Arlt W, Callies F, van Vlijmen JC, et al. Dehydroepiandrosterone replacement in women with adrenal insufficiency. N Engl J Med. 1999;341(14):1013-1020.
53. Dineen R, Thompson CJ, Sherlock M. Adrenal crisis: prevention and management in adult patients. Ther Adv Endocrinol Metab. 2019;10:
2042018819848218.
54. Adrenal Insufficiency. Toronto, Ontario, Canada: College of Dental Hygienists of Ontario; June 20, 2019. https://www.cdho.org/Advisories/CDHO_Factsheet_Adrenal_Insufficiency.pdf. Accessed June 22, 2021.
55. Cox DP, Ferreira L. The oral effects of inhalation corticosteroid therapy: an update. J Calif Dent Assoc. 2017;45(5):227-233.
56. Khalaf MW, Khader R, Cobetto G, et al. Risk of adrenal crisis in dental patients: results of a systematic search of the literature. J Am Dent Assoc. 2013;144(2):152-160.
57. Carlos Fabue L, Jimenez Soriano Y, Sarrion Perez MG. Dental management of patients with endocrine disorders. J Clin Exp Dent. 2010;
2(4):e196-e203.
58. Hahner S, Allolio B. Management of adrenal insufficiency in different clinical settings. Expert Opin Pharmacother. 2005;6(14):2407-2417.
59. Finder RL, Moore PA. Adverse drug reactions to local anesthesia. Dent Clin North Am. 2002;46(4):747-757.
60. Ouanounou A, Ng K, Chaban P. Adverse drug reactions in dentistry. Int Dent J. 2020;70(2):79-84.
61. Decloux D, Ouanounou A. Local anaesthesia in dentistry: a review. Int Dent J. 2021;71(2):87-95.
62. Takahashi Y, Nakano M, Sano K, Kanri T. The effects of epinephrine in local anesthetics on plasma catecholamine and hemodynamic responses. Odontology. 2005;93(1):72-79.
63. Husebye ES, Allolio B, Arlt W, et al. Consensus statement on the diagnosis, treatment and follow-up of patients with primary adrenal insufficiency. J Intern Med. 2014;275(2):104-115.