You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Sinus pneumatization is a known sequela of tooth extraction that results in loss of the alveolar ridge height and width. Maxillary sinus augmentation (MSA) has been demonstrated to be a surgical procedure with low morbidity that increases the alveolar bone height in preparation for implant placement. MSA is a widely accepted and highly predictable procedure with favorable outcomes.1 The key determinant of high success rates of this procedure is the microvascularization of the bone graft material. Neoangiogenesis is a vital component to promote the migration of osteoblasts into the scaffolding graft material.2 Although the postoperative complication rate in MSA is low, wound infections, graft or barrier membrane exposure, graft infection, flap dehiscence, and sinusitis occasionally may occur.
It is well documented that tobacco smoking affects the microvascularization of tissues and, therefore, compromises the vascularization of bone-grafted sites.3 Nicotine, which is present in cigarettes, increases the incidence of postoperative complications due to vasoconstriction-induced wound healing issues. The number of cigarettes consumed daily has also been shown to be directly proportional to the extent of postoperative complications observed.4
Currently, cannabis sativa and electronic cigarettes (e-cigs) are popular among young adults. Cannabis, also known as marijuana, is the most commonly used recreational drug in the world, particularly in young individuals.5 Routine use of cannabis leads to a decrease of heart rate, an increase in blood pressure, enhancement of parasympathetic activity, and disruption of microcirculation.6,7 Although it has been shown that peripheral vascularity is decreased predominantly in skeletal muscle vessels, cannabinoids may also disrupt the physiological activity of bone-grafted sites, deteriorating osteoblast and osteoclast activity.8
E-cigs, introduced in recent years, have gained immediate acceptance worldwide. These electronic devices heat propylene glycol and glycerol and transform these liquids into vapor. E-cigs may be used with or without nicotine and, therefore, may compromise wound healing.9
Early failure of a MSA or grafted bone site associated with the use of cannabis or e-cigs has not been reported in the literature thus far. The objectives of this study are to: report an unexpected early failure of a MSA in an otherwise healthy 36-year-old patient with a history of regular cannabis and e-cig use with subsequent loss of the sinus graft; draw meaningful, clinical conclusions in order to assess this new pathological risk factor; and provide recommendations on whether the habit of using these substances might be considered as a contraindication for sinus grafting.
Case Presentation
The authors obtained approval from the ethics committee of New York University College of Dentistry (NYUCD). The procedure in this study adheres to the tenets of the Declaration of Helsinki.
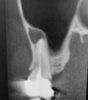
A 36-year-old male patient presented to the Oral and Maxillofacial Surgery department at NYUCD with the chief complaint of pain in the posterior right maxilla. Clinical and radiological evaluation revealed a nonrestorable maxillary second molar with a vertical root fracture and a periapical pathology (Figure 1). The patient had no significant medical history and no known drug allergies. He denied tobacco smoking and alcohol consumption, but admitted using recreational drugs, specifically five marijuana cigarettes per day and e-cigarettes. Thus, the patient was classified as American Society of Anesthesiologists (ASA) II.10
An informed consent was received before the surgical procedure. The maxillary tooth was extracted with minimal trauma with preservation of the alveolar and gingival architecture.
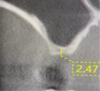
Three months later, a cone-beam computed tomography (CBCT) scan revealed the indication of a MSA due to a pneumatized right maxillary sinus with residual bone height of 2.5 mm and two Underwood septa (Figure 2). Prior to the MSA procedure, based on scientific protocols that suggest smoking cessation for at least 10 days before and after surgery,6,11 the patient was strongly advised to cease his smoking habits due to the risk of bone graft failure.
The MSA surgery was performed under local anesthesia. After a crestal incision, a full-thickness mucoperiosteal flap was reflected. A MSA lateral window technique was performed with a piezoelectric device. The bony window was elevated along with the Schneiderian membrane, and a cancellous particulate allograft was placed in the surgical site and covered with a pericardium membrane. Then, the buccal flap was advanced and secured with a tension-free primary closure. No intraoperative complications were reported, and the patient was dismissed with antibiotic (Augmentin 875/125 mg twice per day for 7 days) and analgesic coverage along with chlorhexidine mouthrinse.
The first follow-up visit, which occurred 1 week after MSA, was uneventful with no signs of infection or membrane exposure. However, at the 2-week postoperative follow-up visit, the patient reported localized soreness; delayed healing, erythema, and edema were observed clinically, and sutures were removed.
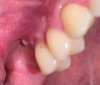
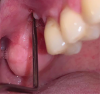
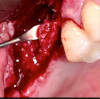
The patient returned asymptomatic at the 4-week follow-up. Nevertheless, the clinical evaluation revealed a fistula of approximately 2 mm to 3 mm in diameter in the buccal vestibule and a wound dehiscence of 3 mm (Figure 3 and Figure 4). Sinus percussion and Valsalva maneuver were negative. Panoramic x-rays revealed no suspicious radiographic changes in the residual sinus cavity or within the grafted site itself compared to the baseline radiograph (Figure 5).
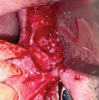
A re-entry to the maxillary sinus was performed. Once the sinus tract was excised, it was visibly evident that the bone graft had been invaded by granulation tissue. The graft was then removed and sent for histopathological evaluation (Figure 6). Intraoperatively, no perforation, defect, or communication to the maxillary sinus was detected; even a distinct layer of solidified grafting material covered the Schneiderian membrane (Figure 7). The histopathological examination reported an acute and chronic inflammation of bone consistent with avascular bone grafting material. Different sections revealed a well-vascularized fibrous connective tissue containing viable and nonviable bone trabeculae and an inflammatory infiltrate of lymphocytes and plasma cells. Focally, the presence of aggregate of necrotic debris, fibrin, and white blood cells was observed (Figure 8).
Discussion
The authors performed a literature search in MEDLINE/PubMed, Cochrane, Scopus, and Google Scholar with no restrictions in terms of time years or types of works. Keywords were used independently and combined with medical subject headings (MeSh) (AND, OR) "cannabis," "marijuana," "e-cigarettes," "maxillary sinus augmentation," "maxillary sinus elevation," "maxillary sinus lift," "bone graft," "infection," "vape smoking," "vaping," and "titanium implant." All of the included studies aimed to analyze the effect of cannabis or e-cigs on bone graft or bone metabolism or their possible association with peri-implant disease. Eight articles qualified to be included in the review.8,12-18 The search process is presented in Figure 9,19 and an overview of the general characteristics of the study is provided in Table 1. (To view Table 1, "Studies Analyzing Effect of Cannabis or Electronic Cigarettes on Bone Graft/Metabolism or Association With Peri-implant Disease," visit compendiumce.com/go/2106.)
Effects of Tobacco Smoking
The effects of tobacco smoking are well documented in the literature. Detrimental effects of tobacco in the oral cavity include gingivitis, periodontitis, and malignant neoplasms as well as having a negative impact on the success rate of bone grafts and sinus augmentation procedures.20,21 Adrenergic effects of tobacco by-products such as nicotine and carbon monoxide, which cause vasoconstriction and reduced peripheral microvascularization and subsequently decrease the tissue oxygen, are well known.22,23 In MSA procedures, these effects would compromise the neoangiogenesis and future amalgamation of the bone graft materials with the sinus floor and increase the risk of impaired wound healing and bone graft failure.
The previous statement is consistent with existing literature. Lindfors et al evidenced the effects of tobacco on ridge augmentation, observing inflammation and decreased success rates-a success rate of only 62.5% was observed in cigarette smokers compared to 94.7% in nonsmokers.24 Also, Schwarz et al analyzed 407 sinus lift procedures and found a higher rate of sinus membrane perforations, wound dehiscence, and sinusitis in smokers.25 This finding is supported by Levin et al and Bain et al, who observed that the maxillary bone structure is more susceptible to the harmful effects of tobacco smoking.4,26 Additionally, Testori et al observed a reduced implant survival rate in tobacco smokers who smoke heavily (more than 15 cigarettes per day) and deficient alveolar bone height (less than 4 mm) after MSA.11
Effects of Cannabis Use
Regarding cannabinoids and tetrahydrocannabinol (THC), the main psychoactive component of cannabis, the literature is unclear and yet contradictory. Napimoga et al asserted that THC and cannabidiol, another major component found in the cannabis plant or sativa, may possess anti-inflammatory and antiresorptive properties by decreasing the expression of the receptor activator of nuclear factor-Kß ligand (RANKL/RANK) and by lowering cytokine production, thus inhibiting neutrophil migration.27 On the other hand, the literature lacks reports about bone grafting failure due to marijuana abuse. Nevertheless, Versteeg et al reported that the use of cannabis could increase the incidence of plaque accumulation and gingivitis as well as produce such side effects as xerostomia, leukoedema, and increased prevalence and density of Candida albicans.28 Moreover, it is established that increased use of cannabis sativa may possibly alter the microcirculation by causing carboxyhemoglobinemia from combustion of cannabis; as a result, these effects decrease the oxygen-carrying capacity of the blood and compromise the wound vascularization of grafted bone sites and, therefore, negatively impact the wound healing process in surgical sites.7
In addition, Idris et al stated that cannabinoid receptors type 1 and type 2 regulate bone remodeling; consequently, the use of recreational cannabis may act as an agonist at the cannabinoid receptors, enhancing osteoclast activity and bone loss.29 This supposition is supported by Pacher et al who posited that marijuana could alter bone healing by increasing osteoclastic activity, thereby inducing bone resorption of the peri-implant bone and compromising implant success.6
Effects of Vaping
Electronic cigarettes are used worldwide as an alternative to tobacco cigarettes and have become particularly popular among young individuals. In the oral cavity, propylene glycol, a chemical contained in e-cigs, breaks down into acetic acid, lactic acid, and propionaldehyde, which in turn, become toxic elements for enamel and soft tissues. Due to its hygroscopic properties, propylene glycol can also lead to tissue desiccation, causing xerostomia; hence, it may increase the risk of caries and periodontal problems.30 Among other potential harmful by-products in e-cig liquid vaporization are acrolein, propanal, styrene, and the carcinogens 2,3-benzofuran and allylthiourea. Acrolein is a glycerol breakdown product that is poisonous, irritant, and flammable. This harmful chemical significantly induces the release of matrix metallopeptidase-9 (MMP-9) from neutrophils, triggering inflammatory responses. For instance, a strong association has been shown between MMP-9 and degree of inflammation and disease severity in patients with chronic obstructive pulmonary disease.31
As stated earlier, e-cigs may be used with or without nicotine. Even if an e-cig does not contain nicotine, it may induce vasoconstriction and, therefore, reduce microvascularization.32 Javed et al noted that vaping may be an etiologic factor of periodontitis. Like tobacco cigarettes, e-cigs could induce the release of destructive inflammatory cytokines in the periodontium contributing to bone loss acceleration.16 A recent study by Otero et al, showed that e-cig use could alter bone metabolism by altering the mRNA and osteoblast genes.17 Furthermore, Rouabhia et al analyzed the effects of e-cigs on dental implants and demonstrated that their use, with or without nicotine, could exponentially reduce the osteoblastic attachment on implant surfaces (P < .001). For this reason, the authors concluded that e-cig vapors increase the risk for implant failure.18 This deduction was confirmed by other recent studies that reported similar results concerning vaping13-15,33; these studies all suggested that use of e-cigs increases the risk for peri-implant inflammation by producing cytokines such as tumor necrosis-α (TNF-α) and interleukin-1β (IL-1β), which may contribute to peri-implant bone loss.
Higham et al studied the effects of e-cigs on the neutrophil inflammatory response. They observed a significant increased secretion of MMP-9 and CXCL8 and an increased activity of neutrophil elastase and MMP-9 when neutrophils were exposed to e-cig vapors, causing a proinflammatory response from neutrophils.31 Abduljabbar et al assessed the levels of proinflammatory cytokines in water pipe smokers compared to nonsmoking patients. Their results were in accordance with the aforementioned cited studies, showing statistically significant results in the water pipe group (P < .01) compared to the nonsmoker group.12
In gingival tissues, e-cigs can alter fibroblast morphology, growth, and density. Several studies have demonstrated that e-cigarette vapors promote fibroblast apoptosis, whether the e-cigs contain nicotine or not.34-36 Fibroblast apoptosis may be induced by an increased expression of the Bcl-2 associated X (BAX) protein, a pro-apoptotic protein that regulates apoptotic cell death and causes an increase in reactive oxygen species production.35 Furthermore, Rouabhia et al observed an increased activity of the caspase-3 pathway, a catalytic enzyme involved in apoptosis, in gingival epithelial cells due to e-cig vapor. This increased caspase-3 activity may stimulate fibroblast apoptosis and, thus, dysregulation of gingival epithelial function.36 Impairment of fibroblast function, migration, and viability may inhibit the role of fibroblasts in wound closure and wound healing.34,37
E-Cigarettes' Effects on Genetic Polymorphism in Progression of Implant Failure
Recent studies reported a possible correlation between genetic polymorphism and implant failure in e-cig users. Different reports that analyzed the level of IL-1β and TNF-α yielded relevant information regarding oral inflammatory diseases such as periodontitis and peri-implantitis.38,39 Furthermore, other studies have demonstrated that levels of IL-1β and TNF-α are significantly increased in patients with peri-implantitis and this is characteristic in peri-implant diseases.13,15,30-32
Another proteolytic enzyme present in peri-implant disease is MMP-8. The effect of MMP-8 is the reduction of angiogenesis, which could be a critical factor for osseointegration. Costa-Junior et al proposed that MMP-8 is associated with early implant failure,40 and this was confirmed by de Araujo Munhoz et al, who verified that this enzyme is a possible risk factor for implant loss.41
The present review has attempted to summarize results published in the scientific literature about a possible correlation among gene polymorphisms, peri-implantitis occurrence, and e-cig use. There are some polymorphisms for which different studies state consistent results. However, the etiology and pathogenesis of peri-implantitis and the immune/inflammatory cytokine response to e-cigs and vaping are not currently well understood, and many studies report conflicting results for specific polymorphisms. Therefore, no robust conclusions can be drawn from the current literature regarding the association of gene polymorphisms and peri-implantitis risk or e-cig use (despite the large number of polymorphisms that have been studied). The inequality of the designs of these studies (mainly concerning population samples) necessitates the conduction of further studies using larger population samples.
Can Vaping Be Considered a Contraindication for Sinus Lift?
MSA has been a highly successful surgical procedure and is considered one of the most predictable bone augmentation methods.42 A complete healing of the flap and incision line along with initial mineralization of the bone graft material can be expected 4 weeks after a sinus graft procedure.43 In the clinical case presented, however, the consistency of the scaffolding material at 4 weeks was fairly similar to the day of surgery. The results of this clinical case suggest a possible correlation between cannabis use, e-cig use, and early bone graft failure. In a young, otherwise healthy patient who was not medically compromised and had no history of cigarette smoking, a possible synergic effect between cannabis sativa and e-cigs may have contributed to the MSA failure. The authors suspect that marijuana smoking and vaping habits may have led to a reduced vascularization of the area and that the increased release of collagenase and gelatinase, MMP-8 and MMP-9, by leukocytes may have impaired neoangiogenesis and bone formation in the grafted sinus site. In this case, the histopathological examination confirmed by the intraoperative findings revealed avascular bone grafting material that caused acute and chronic inflammation.
Clinical Recommendations
According to evidence-based literature as cited herein, the event described in the present case report cannot be explained by a simple and premature graft failure associated with a postoperative sinus augmentation infection. The patient had proper antibiotic coverage and did not display any postoperative signs of infection such as swelling, purulence, or pain around the surgical site. Even the histopathological evaluation confirmed the total absence of any infectious process, displaying only a regular inflammatory process consistent with necrosis of the material.
With incoming generations, a trend in the increased use of electronic cigarettes and cannabis can be expected, and this might lead to further complications. Therefore, clinicians must carefully evaluate a patient's social history, including the use of recreational drugs like cannabis and e-cigarettes. Marijuana and e-cig smoking should be considered risk factors for bone augmentation techniques. Because e-cigs seem to have effects similar to tobacco smoking, it is recommended that patients stop smoking e-cigs 10 days after surgery for single-unit implant and minor surgeries. Furthermore, quitting smoking for 2 weeks before and after elective surgical procedures such as MSA or multiple implant placement might be suggested to prevent postoperative complications, achieve proper wound healing, and maximize the chances of a successful surgical outcome. Finally, clinicians should encourage patients to quit cannabis use prior to surgical procedures, especially in the presence of a coexisting chronic disease and other risk factors such as vaping, because it may exponentially increase wound healing impairment. Nevertheless, more research, including randomized controlled trials, is needed to validate the result of the current study.
Conclusion
Smoking is a common risk factor for MSA failure because it compromises wound healing and the integration, vascularization, and mineralization of bone grafts. Therefore, patients should be carefully evaluated preoperatively. Clinicians must thoroughly assess the patient's social history, including the use of recreational drugs like cannabis and e-cigarettes. Some studies have suggested that genetic polymorphisms of MMP-8, IL-1β, and TNF-α may predispose patients for early implant loss; however, more research is necessary to evaluate this correlation regarding e-cig use and vaping.
The clinical case presented can only suggest that the use of cannabis in synergic association with e-cigs is a possible risk factor for even a highly predictable procedure such as MSA in healthy patients. Because the literature revealed the unicity of the case, more research is needed to better understand the pathophysiology of the use of cannabis and e-cigs on bone grafting.
Acknowledgment
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from the individual participant included in the study.
About the Authors
Marco Bergamini, DDS
Alumnus of Advanced Program for International Dentists in Oral Surgery, Department of Oral and Maxillofacial Surgery, and International Implant Resident at Department of Periodontology and Implant Dentistry, New York University College of Dentistry, New York, New York
Thamar D. Silvestre, DDS
Alumnus of Advanced Program for International Dentists in Oral Surgery, Department of Oral and Maxillofacial Surgery, New York University College of Dentistry, New York, New York
Huzefa S. Talib, BDS
Clinical Associate Professor, Department of Oral and Maxillofacial Surgery, New York University College of Dentistry, New York, New York
Thomas G. Wiedemann, MD, DDS, PhD
Clinical Assistant Professor, Department of Oral and Maxillofacial Surgery,
New York University College of Dentistry, New York, New York
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Jensen OT, Shulman LB, Block MS, Iacono VJ. Report of the Sinus Consensus Conference of 1996. Int J Oral Maxillofac Implants.1998;13 suppl:11-45.
2. Ragucci GM, Elnayef B, Suarez-Lopez Del Amo F, et al. Influence of exposing dental implants into the sinus cavity on survival and complications rate: a systematic review. Int J Implant Dent. 2019;5(1):6.
3. Lindhe J, Karring T, Lang NP, eds. Clinical Periodontology and Implant Dentistry. 4th ed. Blackwell Munskgaard: 2003:3-48.
4. Levin L, Schwartz-Arad D. The effect of cigarette smoking on dental implants and related surgery. Implant Dent. 2005;14(4):357-361.
5. Baggio S, Deline S, Studer J, et al. Routes of administration of cannabis used for nonmedical purposes and associations with patterns of drug use. J Adolesc Health. 2014;54(2):235-240.
6. Pacher P, Batkai S, Kunos G. Cardiovascular pharmacology of cannabinoids. Handb Exp Pharmacol. 2005;(168):599-625.
7. National Academies of Sciences, Engineering, and Medicine. The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. Washington, DC: The National Academies Press; 2017.
8. Nogueira-Filho G. Cannabis and oral health: deleterious effects on periodontitis and dental implants. In: Preedy VR, ed. Handbook of Cannabis and Related Pathologies. Academic Press, Elsevier: 2017:chap e8:e72-e79.
9. Polosa R, Benowitz NL. Treatment of nicotine addiction: present therapeutic options and pipeline developments. Trends Pharmacol Sci. 2011;32(5):281-289.
10. ASA Physical Status Classification System. American Society of Anesthesiologists. https://www.asahq.org/standards-and-guidelines/asa-physical-status-classification-system. Accessed January 15, 2021.
11. Testori T, Weinstein RL, Taschieri S, Del Fabbro M. Risk factor analysis following maxillary sinus augmentation: a retrospective multicenter study. Int J Oral Maxillofac Implants. 2012;27(5):1170-1176.
12. Abduljabbar T, Akram Z, Vohra F, et al. Assessment of interleukin-1β, interleukin-6, and tumor necrosis factor-Α levels in the peri-implant sulcular fluid among waterpipe (narghile) smokers and never-smokers with peri-implantitis. Clin Implant Dent Relat Res. 2018;20(2):144-150.
13. Al-Aali KA, Alrabiah M, ArRejaie AS, et al. Peri-implant parameters, tumor necrosis factor-alpha, and interleukin-1 beta levels in vaping individuals. Clin Implant Dent Relat Res. 2018;20(3):410-415.
14. Akram Z, Vohra F, Bukhari IA, et al. Clinical and radiographic peri-implant parameters and proinflammatory cytokine levels among cigarette smokers, smokeless tobacco users, and nontobacco users. Clin Implant Dent Relat Res. 2018;20(1):76-81.
15. ArRejaie AS, Al-Aali KA, Alrabiah M, et al. Proinflammatory cytokine levels and peri-implant parameters among cigarette smokers, individuals vaping electronic cigarettes, and non-smokers. J Periodontol. 2019;90(4):367-374.
16. Javed F, Abduljabbar T, Vohra F, et al. Comparison of periodontal parameters and self-perceived oral symptoms among cigarette smokers, individuals vaping electronic cigarettes, and never-smokers. J Periodontol. 2017;88(10):1059-1065.
17. Otero CE, Noeker JA, Brown MM, et al. Electronic cigarette liquid exposure induces flavor-dependent osteotoxicity and increases expression of a key bone marker, collagen type I. J Appl Toxicol. 2019;39(6):888-898.
18. Rouabhia M, Alanazi H, Park HJ, Gonçalves RB. Cigarette smoke and e-cigarette vapor dysregulate osteoblast interaction with titanium dental implant surface. J Oral Implantol. 2019;45(1):2-11.
19. Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097.
20. Akinkugbe AA, Slade GD, Divaris K, Poole C. Systematic review and meta-analysis of the association between exposure to environmental tobacco smoke and periodontitis endpoints among nonsmokers. Nicotine Tob Res. 2016;18(11):2047-2056.
21. Winn DM. Tobacco use and oral disease. J Dent Educ. 2001;65(4):306-312.
22. Galindo-Moreno P, Fauri M, Avila-Ortiz G, et al. Influence of alcohol and tobacco habits on peri-implant marginal bone loss: a prospective study. Clin Oral Implants Res. 2005;16(5):579-586.
23. Jensen JA, Goodson WH, Hopf HW, Hunt TK. Cigarette smoking decreases tissue oxygen. Arch Surg. 1991;126(9):1131-1134.
24. Lindfors LT, Tervonen E, Sándor G, Ylikontiola LP. Guided bone regeneration using a titanium-reinforced ePTFE membrane and particulate autogenous bone: the effect of smoking and membrane exposure. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(6):825-830.
25. Schwarz L, Schiebel V, Hof M, et al. Risk factors of membrane perforation and postoperative complications in sinus floor elevation surgery: review of 407 augmentation procedures. J Oral Maxillofac Surg. 2015;73(7):1275-1282.
26. Bain CA, Moy PK. The association between the failure of dental implants and cigarette smoking. Int J Oral Maxillofac Implants. 1993;8(6):609-615.
27. Napimoga MH, Benatti BB, Lima FO, et al. Cannabidiol decreases bone resorption by inhibiting RANK/RANKL expression and pro-inflammatory cytokines during experimental periodontitis in rats. Int Immunopharmacol. 2009;9(2):216-222.
28. Versteeg PA, Slot DE, van der Velden U, van der Weijden GA. Effect of cannabis usage on the oral environment: a review. Int J Dent Hyg. 2008;6(4):315-320.
29. Idris AI, Van't Hof RJ, Greig IR, et al. Regulation of bone mass, bone loss and osteoclast activity by cannabinoids receptors. Nat Med. 2005;11(7):774-779.
30. Froum S, Neymark A. Vaping and oral health: it's worse than you think. Perio-Implant Advisory website. January 10, 2019. https://www.perioimplantadvisory.com/clinical-tips/article/16412201/vaping-and-oral-health-its-worse-than-you-think. Accessed January 15, 2021.
31. Higham A, Rattray NJW, Dewhurst JA, et al. Electronic cigarette exposure triggers neutrophil inflammatory responses. Respir Res. 2016;17(1):56.
32. Pywell MJ, Wordsworth M, Kwasnicki RM, et al. The effect of electronic cigarettes on hand microcirculation. J Hand Surg Am. 2018;43(5):432-438.
33. AlQahtani MA, Alayad AS, Alshihri A, et al. Clinical peri-implant parameters and inflammatory cytokine profile among smokers of cigarette, e-cigarette, and waterpipe. Clin Implant Dent Relat Res. 2018;20(6):1016-1021.
34. Alanazi H, Park HJ, Chakir J, et al. Comparative study of the effects of cigarette smoke and electronic cigarettes on human gingival fibroblast proliferation, migration and apoptosis. Food Chem Toxicol. 2018;118:390-398.
35. Rouabhia M, Park HJ, Semlali A, et al. E-cigarette vapor induces an apoptotic response in human gingival epithelial cells through the caspase-3 pathway. J Cell Physiol. 2017;232(6):1539-1547.
36. Sancilio S, Gallorini M, Cataldi A, di Giacomo V. Cytotoxicity and apoptosis induction by e-cigarette fluids in human gingival fibroblasts. Clin Oral Investig. 2016;20(3):477-483.
37. Chen IL, Todd I, Fairclough LC. Immunological and pathological effects of electronic cigarettes. Basic Clin Pharmacol Toxicol. 2019;125(3):237-252.
38. Akram Z, Vohra F, Bukhari IA, et al. Clinical and radiographic peri-implant parameters and proinflammatory cytokine levels among cigarette smokers, smokeless tobacco users, and nontobacco users. Clin Implant Dent Relat Res. 2018;20(1):76-81.
39. Petkovic-Curcin A, Zeljic K, Cikota-Aleksic B, et al. Association of cytokine gene
polymorphism with peri-implantitis risk. Int J Oral Maxillofac Implants. 2017;32(5):e241-e248.
40. Costa-Junior FR, Alvim-Pereira CC, Alvim-Pereira F, et al. Influence of MMP-8 promoter polymorphism in early osseointegrated implant failure. Clin Oral Investig. 2013;17(1):311-316.
41. de Araujo Munhoz F, Branco F, Souza R, dos Santos M. Matrix metalloproteinases gene polymorphism haplotype is a risk factor to implant loss: a case-control study. Clin Implant Dent Relat Res. 2018;20(6):1003-1008.
42. Starch-Jensen T, Aludden H, Hallman M, et al. A systematic review and meta-analysis of long-term studies (five or more years) assessing maxillary sinus floor augmentation. Int J Oral Maxillofac Surg. 2018;47(1):103-116.
43. Moy PK, Lundgren S, Holmes RE. Maxillary sinus augmentation: histomorphometric analysis of graft materials for maxillary sinus floor augmentation. J Oral Maxillofac Surg. 1993;51(8):857-862.