You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
At the dawn of the 21st century the emergence of cone-beam computed tomography (CBCT) imaging technology was, without much debate, the most significant advancement in extraoral dental imaging since the introduction of panoramic radiography.1 CBCT enabled dentistry to realize the third dimension of imaging with its diagnostic advantages being widely reported.2,3
Additive manufacturing (AM) was pioneered in 1984 by Charles Hull, when he presented and later patented his technique of forming physical objects from a series of 3-dimensional (3D) computer-assisted design data.4 In 1989, a separate firm developed and later patented the filament deposition modeling technique.5 These technologies maintained proprietary rights and, as a consequence, were expensive and affordable for use only by large companies and laboratories. Expiration of the patents and creation of the RepRap project in 2004 enabled the use of AM technology by a wider audience.6 In a dental clinic context, the synergy of CBCT and AM has applications in guided implant surgery,7 design and fabrication of splints for orthognathic surgery,8 maxillofacial prosthetics,9 endodontics,10 and fabrication of anatomical models for diagnostic and educational purposes.11
The aim of this article is to describe the applications of CBCT hardware for AM purposes given the cost limitations of typical private dental practices. Its focus of interest will be static surgical guides, and it will outline the workflow, techniques, and potential pitfalls dentists may encounter.
General Workflow
Any AM process includes three consecutive stages, which in the scope of guided implant surgery consist of: (1) data acquisition, (2) design of the 3D guide (computer-assisted design [CAD]), and (3) fabrication and post-processing of the physical guide (computer-assisted manufacturing [CAM]).
Data Acquisition
CBCT Scanning of the Patient
To begin the process, the patient should be positioned with the teeth separated so as to leave 3 mm to 7 mm of interocclusal space at the point of the incisors. The occlusal level of the teeth should be maintained horizontal to the floor, and the patient should be instructed to keep completely still for the duration of the scan.
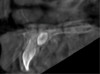
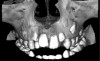
Patient movement is a major concern in CBCT imaging. Movement may present as double contours and/or an overall lack of sharpness of the image. In computer-guided implant surgery, a sharp image is necessary for a successful registration of the surface model scan to the CBCT anatomy. Motion correction hardware and software algorithms have been proposed and commercially applied in some CBCT units and appear to enhance image quality somewhat (Figure 1 through Figure 6).12
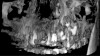
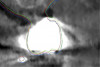
A similar concern relates to artifacts resulting from metal objects. These artifacts present as shadows and streaks around and between objects of high density, effectively deteriorating the overall shape of the crowns of the teeth in the scan. Any kind of metal prosthesis can deteriorate the scan and render the eventual registration unsuccessful. Although many CBCT manufacturers incorporate metal artifact reduction algorithms, their usefulness remains untested in the realm of implant dentistry, and the improvement they offer in the diagnostic effectiveness of the scan is questionable (Figure 7 through Figure 11).13 Positioning the patient with teeth separated and on a horizontal occlusal plane may reduce the number of artifacts originating from the opposite jaw.14
CBCT Operational Parameters
Implant dentistry is a low-demand imaging task and presents an excellent opportunity for the clinical implementation of the renowned principle of ALARA (as low as reasonably achievable). As a general rule, low-dose protocols with the shortest scan time must be employed. A voxel resolution of 200 μm is adequate for the depiction of bone and teeth, the partial rotation angle (ie, 180 degrees) must be preferred where available, and mA should be kept to the minimum. The field of view (FOV) must cover the whole jaw and part of the opposite teeth, thus FOV of 8 cm (d) x 5 cm (h) is the smallest recommended. The kV must be the highest available in the unit because fewer metal artifacts are anticipated with higher kV.15
Virtual Model of the Dentition
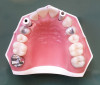
A digital model of the dentition is required for the fabrication of the surgical guide. The digital model can be acquired indirectly through the digitization of the stone model or impression material using either a desktop scanner or CBCT device. Direct digitization of the dentition is accomplished with the use of an intraoral scanner. Studies have shown that CBCT devices can be used to create accurate virtual models with x-ray scanning of not only stone models but also impressions.16,17 Intraoral scanners have been shown to demonstrate relative inaccuracy when scanning involves the complete arch,18 with scanning being somewhat challenging in edentulous cases.
Design of the 3D Guide (CAD)
Using the proprietary software, data acquired from the CBCT device are exported in DICOM (digital imaging and communication in medicine) format, while data from intraoral and desktop scanners are exported as stereolithography (STL) files. These data files must be imported to a dedicated implant planning and guide design software. It is this very software that will be used for: the virtual waxing on the surface model of the dentition; the virtual placement of implants, taking into account any surgical and prosthetic considerations; the design of the guide based on the particular implant system each clinic uses; and, finally, export of the designed guide as a printable STL file.
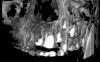
Whatever the software solution may be, the first steps after the importing of the DICOM and STL files are the segmentation of the CBCT anatomy and the registration of the 3D surface model of the teeth to the anatomy. In the present context, segmentation refers to the process where the needed voxels of the CBCT crowns of the teeth are selected to be represented in the 3D model of the anatomy. Registration refers to the process of the alignment of the two data sets (CBCT and surface model of the teeth). This is a necessary process taking into account that the implants are virtually placed using information from the CBCT anatomy, but the surgical guide is fabricated on the 3D surface model of the dentition. This registration procedure is achieved when a number of distinct points on the anatomy of the teeth, both on the CBCT image and on respective places on the 3D surface model, can be discerned and aligned. However, in edentulous cases where few teeth are remaining and/or placed in unfavorable positions and where excessive metal artifacts are present, this may be difficult to accomplish (Figure 12 through Figure 15).
The Need for a Radiographic Template
The need for radiographic templates in computer-aided implant dentistry seems to be limited. Current editions of various implant planning software offer users the ability to virtually place the implant in not only the correct anatomical position but also the optimal prosthetic location with the help of virtual waxing.
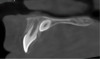
In the case of a fully edentulous patient, CBCT scanning cannot capture the soft tissues with the accuracy needed given the presence of the tongue and the movable tissues of the lips and cheeks collapsing to the edentulous arch. Retraction of the soft tissues with the use of cotton wools19 or lip retractors has been proposed, however the effectiveness of these techniques has not been thoroughly evaluated in the literature. If available, the patient's relined prosthesis remains the preferred solution if there is no metal in the denture to distort the x-ray scan and the prosthetic alignment of the teeth is adequate. If these conditions cannot be met, a set-up is performed and a scanning appliance is fabricated. This radiographic template is scanned while it is seated in the patient's mouth and also separately. The use of special radiographic markers placed on the denture shortly before scanning is advocated. Such markers consist of spherical glass beads that, unlike asymmetric gutta-perchas, make subsequent registration easier (Figure 16 through Figure 18).
A radiographic template is also necessary when a limited number of teeth are in the arch or if multiple metal scatter artifacts are anticipated. As a general rule, the authors advise such a template be used either with five teeth or fewer when there are no restorations, or with seven teeth or fewer when some of them have metal restorations. Exposure parameters have to be individualized for each CBCT scanner model, but use of the maximum mAs and resolution setting available is advisable for the extraoral scanning of the radiographic templates.
Fabrication of the Physical Guide (CAM)
After the virtual planning of the implant surgery and as soon as the guide design is accomplished in the software, a file of the guide must be exported and ultimately imported to a different software used for the actual printing. The de facto file format used for export from the surgical planning and guide design software is STL. This file is imported into the proprietary software that comes with every AM device for manipulation and slicing.
In the manipulation stage the virtual guide is oriented and positioned, the printing parameters are selected based on the material and the required resolution, and the virtual guide is sliced. Slicing refers to the procedure where the STL file of the surgical guide is transformed into a series of thin individual axial slices parallel to the horizontal plane of the printer's table and converted into another file form that contains instructions for the 3D printer.
Various terms have been used to describe the processes and techniques for fabricating physical models from CAD data. AM classifies the processes used to fabricate objects by controlled material addition, ie, via the extrusion of material through a fine nozzle, or use of an energy source applied to powders or polymers. In the context of the present article, with the focus on low-cost, affordable manufacturing available in an implant clinic, two groups of AM will be discussed: vat polymerization and material extrusion.
Vat Polymerization
According to the ISO/ASTM 52900:2015 international standard, "vat polymerization" is the collective term used for all technologies that use a vat to store resin.20 In short, vat polymerization technology utilizes a vat of liquid photopolymer resin, from which a model is constructed layer by layer. An ultraviolet (UV) light is used to cure the resin while a platform conveys the object being made upward after each new layer is cured. Subcategories of vat polymerization include stereolithography (SLA), digital light processing (DLP), and liquid crystal display (LCD) devices.
SLA devices use a laser beam as the energy source. The laser beam is directed by mirrors and guided to outline dot by dot each layer of the object to be printed. In DLP, a digital projector screen is used to flash a single image of each layer across the entire platform at once. In LCD, a liquid crystal display screen is used to mask off a UV light source, revealing only the pixels necessary for the current layer in the printing process. Although LCD is the most cost-effective technique, its main disadvantage is the relatively short life-cycle of the screen.
Material Extrusion
Printers that use melted material to add layers and form the object are in the AM category called fused deposition modeling (FDM), a term trademarked in the United States by Stratasys Ltd. An alternative term used for the same process but without infringing any trademarks is fused filament fabrication (FFF). Unlike the vat polymerization devices, which use a liquid resin to construct the object, FFF devices use a variety of materials, including acrylonitrile butadiene styrene (ABS), polycarbonate (PC), polylactic acid (PLA), high-density polyethylene, PC/ABS, polyethylene terephthalate (PETG), polyphenylsulfone, and high-impact polystyrene. In the context of implant dentistry PLA, ABS, and PETG are mainly used.
AM Considerations: The 3D Printer
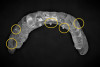
Both the vat polymerization and FFF AM technologies can be used in a dental environment, with the cost of 3D printers ranging for both technologies from as low as US$200 to several thousand dollars. Both technologies are capable of producing 3D surgical guides with the necessary accuracy for the task (Figure 19 and Figure 20).21, 22 However, there are distinct differences.
Vat polymerization, which is capable of printing with resolution down to 50 μm, produces guides that are visually smoother and presumably greater in detail, while FFF printers, with an operational limit of resolution of around 100 μm, produce more "coarse" guides. In the realm of implant dentistry this is relatively unimportant. The voxel size of the CBCT is around 200 μm and this defines the upper limit of accuracy achievable by computer-aided implant dentistry. If the material properties used for the printed guide are adequate, the resolution of the printing will not affect the end accuracy of the procedure. Vat polymerization, however, can be a messy procedure, as the guide demands post processing with cleaning in an isopropyl alcohol bath and additional polymerization. FFF guides are almost ready for use after printing with fewer post-processing steps.
Nevertheless, vat printers offer an advantage in that the development of biocompatible resins for guide fabrication that are suitable for steam sterilization render this technology ideal for the task. For FFF printers, biocompatibility is more of an issue, although several materials have been introduced to address it. Such materials available for use with low-cost FFF printers include: a PLA registered with the US Food and Drug Administration (FDA) that has antimicrobial capabilities and an ISO 10993-10:2010 certification23 for biocompatibility with skin/mucosa contact; a high-strength PETG-based material that fulfills 11607-1:2019,24 ISO 10993, and USP Class VI25 and is listed with the FDA; and an ABS with an ISO 10993-1:2018 certification.26 However, steam sterilization cannot be used with these filaments and only chemical disinfection is possible.27
Finally, several critical elements that may affect quality control in AM must be considered. These include the experience of the clinician with the 3D printing procedure, the build location and orientation (affecting the structural properties of the guide), and the need for an absence of any residual unset material especially in vat-based systems.
Conclusion
The synergy of CBCT units with low-cost AM devices presents unique opportunities for clinicians in the dental clinic, promising the realization of guided implant surgery with the accuracy and quality expected from professionally made guides but with limited fiscal and temporal cost. Considerations related to CBCT imaging and the AM process have been presented. Competent clinicians are exhorted to explore and exploit current technology to offer improved care for their patients.
About the Authors
Dimitrios Apostolakis, DDS, MSc, MSc
Private Practice in Dental Radiology, Chania, Crete, Greece
George Michelinakis, DDS, MSc, MPhil
Private Practice in Prosthodontics and Esthetic and Implant Dentistry, Heraklion, Crete, Greece
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Farman AG, Scarfe WC. Historical perspectives on CBCT. In: Scarfe WC, Angelopoulos C, eds. Maxillofacial Cone Beam Computed Tomography.Cham, Switzerland: Springer; 2018:3-11.
2. American Dental Association Council on Scientific Affairs. The use of cone-beam computed tomography in dentistry: an advisory statement from the American Dental Association Council on Scientific Affairs. J Am Dent Assoc. 2012;143(8):899-902.
3. Radiation Protection No. 172. Cone Beam CT for Dental and Maxillofacial Radiology. Evidence-Based Guidelines. European Commission. 2012.
4. Hull CW, inventor; UVP Inc, assignee. Apparatus for production of three-dimensional objects by stereolithography. US patent 4,575,330. March 11, 1986.
5. Crump SS, inventor; Stratasys Inc, assignee. Apparatus and method for creating three-dimensional objects. US patent 5,121,329. June 9, 1992.
6. Bowyer A. 3D printing and humanity's first imperfect replicator. 3D Printing and Additive Manufacturing. 2014:1(1):4-5.
7. Abduo J, Lau D. Accuracy of static computer-assisted implant placement in anterior and posterior sites by clinicians new to implant dentistry: in vitro comparison of fully guided, pilot-guided, and freehand protocols. Int J Implant Dent. 2020;6(1):10.
8. Park JH, Lee YB, Kim SY, et al. Accuracy of modified CAD/CAM generated wafer for orthognathic surgery. PLoS One. 2019;14(5):e0216945.
9. Michelinakis G. The use of cone beam computed tomography and three dimensional printing technology in the restoration of a maxillectomy patient using a dental implant retained obturator. J Indian Prosthodont Soc. 2017;17(4):406-411.
10. Connert T, Zehnder MS, Amato M, et al. Microguided endodontics: a method to achieve minimally invasive access cavity preparation and root canal location in mandibular incisors using a novel computer-guided technique. Int Endod J. 2018;51(2):247-255.
11. Msallem B, Sharma N, Cao S, et al. Evaluation of the dimensional accuracy of 3D-printed anatomical mandibular models using FFF, SLA, SLS, MJ, and BJ printing technology. J Clin Med. 2020;9(3):817.
12. Santaella GM, Wenzel A, Haiter-Neto F, et al. Impact of movement and motion-artefact correction on image quality and interpretability in CBCT units with aligned and lateral-offset detectors. Dentomaxillofac Radiol. 2020;49(1):20190240.
13. Vasconcelos KF, Codari M, Queiroz PM, et al. The performance of metal artifact reduction algorithms in cone beam computed tomography images considering the effects of materials, metal positions, and fields of view. Oral Surg Oral Med Oral Pathol Oral Radiol. 2019;127(1):71-76.
14. Scarfe W. Cone beam computed tomography. In: Mallya S, Lam E, eds. White and Pharoah's Oral Radiology. Principles and Interpretation.8th ed. St. Louis, MO: Elsevier; 2019.
15. Freitas DQ, Fontenele RC, Nascimento EHL, et al. Influence of acquisition parameters on the magnitude of cone beam computed tomography artifacts. Dentomaxillofac Radiol. 2018;47(8):20180151.
16. Apostolakis D, Michelinakis G, Kourakis G, Pavlakis E. Accuracy of triangular meshes of stone models created from DICOM cone beam CT data. Int J Implant Dent. 2019;5(1):20.
17. Peng L, Chen L, Harris BT, et al. Accuracy and reproducibility of virtual edentulous casts created by laboratory impression scan protocols. J Prosthet Dent. 2018;120(3):389-395.
18. Michelinakis G, Apostolakis D, Pavlakis E, et al. Accuracy of IOS in full-arch dentate patients compared to CBCT cast-scanning. An in-vivo study. Eur J Prosthodont Restor Dent. 2019;27(3):122-130.
19. Scherer MD, Roh HK. Radiopaque dental impression method for radiographic interpretation, digital alignment, and surgical guide fabrication for dental implant placement. J Prosthet Dent. 2015;113(4):343-346.
20. ISO/ASTM 52900:2015. Additive manufacturing - General principles - Terminology. December 2015. https://www.iso.org/standard/69669.html. Accessed September 25, 2020.
21. Hatz CR, Msallem B, Aghlmandi S, et al. Can an entry-level 3D printer create high-quality anatomical models? Accuracy assessment of mandibular models printed by a desktop 3D printer and a professional device. Int J Oral Maxillofac Surg. 2020;49(1):143-148.
22. Nestler N, Wesemann C, Spies BC, et al. Dimensional accuracy of extrusion- and photopolymerization-based 3D printers: in vitro study comparing printed casts. J Prosthet Dent. 2020;S0022-3913(19)30748-6.
23. ISO 10993-10:2010. Biological evaluation of medical devices - Part 10: Tests for irritation and skin sensitization. August 2010. https://www.iso.org/standard/40884.html. Accessed September 25, 2020.
24. ISO 11607-1:2019. Packaging for terminally sterilized medical devices - Part 1: Requirements for materials, sterile barrier systems and packaging systems. February 2019. https://www.iso.org/standard/70799.html. Accessed September 25, 2020.
25. USP Class VI. Material Biocompatibility and Standard for Plastic Manufacturing Systems/Components. US Pharmacopeia. usp.org.
26. ISO 10993-1:2018. Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process. August 2018.
https://www.iso.org/standard/68936.html. Accessed September 25, 2020.
27. Rankin TM, Giovinco NA, Cucher DJ, et al. Three-dimensional printing surgical instruments: are we there yet? J Surg Res. 2014;189(2):193-197.