You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
A biopsy is the removal of tissue from a living organism for microscopic examination and diagnosis. The most consequential purpose of performing a biopsy is to enable clinicians to definitively diagnose dysplastic tissue changes that are potentially pre-cancerous or cancerous. The American Academy of Oral and Maxillofacial Pathology recommends that any abnormal tissue from the oral and maxillofacial region be removed and submitted to an oral pathologist. Exceptions to this recommendation include clinically normal-appearing extracted teeth, clinically normal extirpated dental pulp tissue, other clinically normal-appearing tissue, and excess donor tissue resulting from grafting procedures.1 With these guidelines in mind, even slightly abnormal tissue that is removed from the oral cavity should be analyzed, no matter how confident the clinician may be regarding its diagnosis.
Currently, several methods of tissue collection are available to clinicians. Biopsies can be performed via scalpel removal (ie, incision, excision, marsupialization), tissue punch, shave method, brush sampling, lasers, electrosurgery, radiosurgery, fine needle aspiration, core needle biopsy, vacuum-assisted biopsy, and other methods. The most common biopsies are incisional and excisional types using a scalpel. An incisional biopsy involves the removal of a representative portion of the lesion along with a portion of healthy tissue adjacent to it, while an excisional biopsy is the removal of the entire lesion. If a malignancy is strongly suspected, an initial incisional biopsy is the preferred approach to subsequently direct the definitive treatment. An excisional biopsy should be considered only if the lesion is thought to be almost certainly benign.2-4 Regarding small lesions (ie, <2 cm), as the medical community moves toward molecular-based and gene-targeted cancer therapies, an excisional biopsy of even small suspected malignancies would preclude the preservation of unfixed tissue samples for molecular pathological/genetic examinations. Thus, even with small lesions it is advisable to obtain the diagnosis first and then plan the definitive therapy.
Using a scalpel blade allows the clinician to obtain a tissue sample with well-defined margins and no structural alterations. However, this approach requires both local anesthesia and sutures. In contrast, with the use of a laser, because of its thermal effects, incisional margins may not be as well defined and could be inadvertently altered if the operator is not adequately skilled or is inexperienced. This problem may be exacerbated if there is no appropriately sized border of healthy tissue included in the sample. Also, because of the potential for specimen alteration or damage with a laser, its usage may not be appropriate in all cases, such as when interpretation of subtle cytomorphologic change is imperative to arriving at the definitive diagnosis (eg, suspected epithelial dysplasia).5-8
Before any biopsy is performed, the location, size, color, texture, and consistency of the lesion should be documented. Biopsy of epithelial lesions must include the full epithelial thickness and supporting connective tissue, as this allows for evaluation of invasive carcinomas and is also helpful for physical support. Even with a mucocele, for example, the lesion needs to include the supporting connective tissue, particularly the offending minor salivary gland, or the lesion may recur because the gland will continue to express mucus into the connective tissue where the body will attempt to wall it off and form another mucocele. Another example is with epithelial dysplasia and suspected superficial invasion; without the lamina propria there is no way to evaluate if the basement membrane is intact or has been violated. Thinner specimens may limit evaluation and proper diagnosis. If tissue is removed for a suspected vesiculobullous disease, the specimen must contain the ulcerative tissue along with adjacent epithelium to allow pathological analysis to detect the presence of intraepithelial or basement membrane separation. If direct immunofluorescence studies are necessary, tissue submission in a non-formalin-based transport medium will facilitate the ability to identify the culpable antigen. Clinicians should be wary of vascular lesions that may be prone to significant hemorrhage. These lesions are usually red, blue, or purple in color and often blanch with pressure. If a lesion of vascular origin is being harvested, use of a dental laser should be given serious consideration.9,10
As dental laser usage increases, many oral pathologists are expressing concern regarding poor biopsy technique, based on author conversations with multiple oral pathologists. Initially, dental lasers were intended solely for the purpose of hemostasis following scalpel or tissue-punch biopsies. However, lasers are increasingly being used to harvest specimens for histopathologic analysis.11,12 While many such samples are collected successfully, many are not diagnostic because of improper technique. Vescovi et al demonstrated that the photothermal effects of lasers can produce microscopic artifacts in the peri-incisional area of a lesion.13 Others have reported that epithelial and connective tissue artifacts, including nuclear changes, could lead to imprecise histological observations such as pseudodysplastic alterations that may interfere with diagnosis.14,15
Chen et al lists five main potential causes of an improper diagnosis: limitations in the biopsy technique, including sampling errors (eg, submission of tissue that was not representative of the lesion); insufficient tissue for diagnosis; presence of obscuring inflammation making diagnosis difficult; presence of other tissue artifacts; and pathologist discrepancy in interpretation between initial and subsequent specimens.16
The use of dental lasers avoids common sequelae seen with other novel methods of sample collection, like the edema, irritation, and delayed healing associated with cryosurgery.17,18 Various laser types may be used to harvest a soft-tissue biopsy. While each device presents advantages and disadvantages, generally lasers provide minimal trauma, uncomplicated healing, and faster recovery compared with conventional treatment modalities. Laser-treated wounds exhibit less scar formation due to fewer myofibroblasts in the healing tissue.19 The result is minimal wound contraction and scarring.
Any deficiencies associated with the use of a dental laser to collect a proper sample are related mostly to either insufficient tissue for diagnosis or other tissue artifacts, specifically thermal artifacts. This article, therefore, is intended to assist clinicians in enhancing biopsy technique when using a laser.
Diode Lasers
Because diode lasers are the most affordable of dental lasers, clinicians may be inclined to discount them as a viable instrument for biopsy. However, diode lasers provide a bloodless field and rapid healing, are easy to operate,18,20 and, moreover, the bactericidal effects they provide reduce the risk of bacteremia or infection following treatment.21 It has been suggested that diode lasers may stimulate clotting factor VII, which seems to enhance hemostasis, as a result of the "hot-tip" effect, causing thermocoagulation.22,23
In instances where thermal damage may be a concern, irrigating with saline while operating the laser can help to dissipate thermal energy. According to Fornaini et al, the effective penetration depth of a diode laser at 2 W is at least 2 mm, as measured with two thermocouples at different tissue depths in bovine tongue tissue.24 This may be adequate to seal small lymphatic vessels, minimizing postoperative edema.25 Research by Chawla et al showed that a biopsy may be collected using a diode laser with no alteration in the microanatomy of the specimen.26 Due to absorption characteristics of the various wavelengths of diode lasers, a 980 nm diode laser is able to cut slightly quicker than an 810 nm diode, as the former diode has greater absorption into water, which is the predominant component in most tissues in the oral cavity, than the 810 nm diode, which does not demonstrate much, if any, affinity to water.24 This difference is most noticeable when ablating tissues with minimal pigmentation.
Nd:YAG Lasers
Similar to diode lasers, neodymium-doped:yttrium aluminum garnet (Nd:YAG) lasers have a high affinity for pigmented tissues, which is why they are well suited for soft-tissue ablation. The difference between these two laser types is the high energy generated by the Nd:YAG lasers. The high peak power of the Nd:YAG laser makes it possible to deliver larger bursts of energy in shorter pulses, so the energy travels deeper into the tissue and generally causes a higher thermal tissue response in deep tissues with subsequent coagulation.27 In a study on histological specimens slightly less than 7 mm in size, serious thermal effects were noted with the use of an Nd:YAG. Vescovi et al reported that a comparison of Nd:YAG laser parameters indicated that the use of higher frequency with lower power produced a better incision with less tissue injury.13
CO2 Lasers
Carbon dioxide (CO2) lasers have been established for use in the treatment of benign and premalignant intraoral lesions.28,29 Among dental lasers, CO2 lasers are best suited to treating broad-based lesions because of their method of ablation, which is not delivered through a glass tip, but instead through a hollow light guide or waveguide, creating a collimated beam that can be focused. This wavelength of laser offers a reduction in pain, which may be due to creation of a thin layer of denatured collagen that acts as a biological wound dressing. This thin layer has the ability to seal sensory nerve endings and reduce pain.30 Yagüe-García et al noted that compared with a scalpel, the CO2 laser used in their clinical study resulted in more scarring, but this would be true with any laser device.31 Because of their high absorption into water, CO2 lasers cut much faster and cleaner than diode lasers. Devices that deliver their laser beam with an aerosolized water spray from the surgical handpiece offer increased safety.
Erbium Lasers
Due to their high absorption capabilities into water, which as stated earlier makes up most tissues in the oral cavity, erbium lasers (erbium-doped:yttrium aluminum garnet [Er:YAG] and erbium, chromium-doped:yttrium, scandium, gallium, and garnet [Er,Cr:YSGG]) have many applications in dentistry, including minimally invasive periodontal surgery, cavity preparation, and endodontics. Originally thought to be a hard-tissue device because of an inability to coagulate soft tissue (particularly the Er:YAG), erbium laser technology has developed to the point where predictable hemostasis is now possible.
Like CO2 lasers, erbium lasers have a high coefficient of absorption into water, which allows for a high-impact, superficial effect in soft tissue. Because the method of soft-tissue ablation with an erbium laser (including incorporation of an aerosolized water spray) is different than the more thermal effects seen in other devices, specimen integrity is able to be far better maintained,32 and the reduction of inflammation following tissue ablation produces less postoperative pain.33
While comparing thermal damage zones caused by CO2 and erbium lasers during histopathological evaluation, Suter and colleagues noted a thinner thermal zone in the erbium laser specimens. In that study, the zone of thermal damage for an excisional biopsy was 34 µm for the Er:YAG versus 74.9 µm created with the CO2 laser. While interesting, this data requires further research because of the small number of specimens in the study.34
KTP Lasers
Potassium titanyl phosphate (KTP) lasers have been used successfully for excisional biopsies; however, dental versions of this laser are not currently available in the United States. This is a particularly favorable wavelength because the green beam is completely absorbed into red tissues, allowing deep thermal injury to be safely avoided,35 whereas the Nd:YAG laser penetrates deeper into tissue and has the potential for deep thermal injury. This characteristic makes the KTP laser an excellent option for vascular lesions.
Factors to Consider Using a Laser
Romeo et al studied the effects of Er:YAG, Nd:YAG, and Er,Cr:YSGG lasers as well as 808 nm and 980 nm diode lasers. Tissue samples were harvested from a pig tongue. The authors determined that each of these laser types could be used to perform a soft-tissue biopsy. Even if slight alterations were present at the tissue margins, no sample was damaged to the point where histological examination was impossible. The Er,Cr:YSGG wavelength performed the best in these trials, with peripheral damage confined to less than 1 mm.36 It should be noted, however, that the cost of erbium lasers and some CO2 and Nd:YAG lasers make them an uneconomical choice if they are intended to be used only for the collection of tissue samples for biopsy.
Regardless of wavelength, laser-assisted treatment offers advantages such as photocoagulation with an erbium or CO2 laser or thermal coagulation when using a hot-tip diode device. As a result, sutures are rarely necessary following laser biopsy. To minimize thermal trauma and the generation of tissue artifacts, treatment time should be limited, with breaks incorporated, referred to as thermal relaxation time.37 Also, the addition of intraoperative water or saline spray provided by an assistant can help reduce thermal trauma and tissue artifacts. Lauritano et al noted that after surgical excision with a laser, no sutures were necessary, and after 7 days, complete epithelialization was observed in nearly all cases, and none of the cases resulted in bacterial infection of the wound.37 Laser coagulation often results in less postoperative pain and inflammation and improved wound healing by secondary intention,38 allowing healing by wound contraction with tissue movement from the margins toward the center of the wound.
As noted earlier, not every biopsy can or should be collected using a dental laser. The size and location of the tissue, as well as the potential for malignancy, should be thoroughly considered before opting for this modality. A sufficient sample size must be removed for the most accurate diagnosis. When a sample is placed into the fixative solution, it shrinks, hardens, and usually changes color. A larger sample size can compensate for the shrinkage and allow for better tissue manipulation.39 It is impossible to dictate specific laser parameters for every wavelength and device currently in use. A survey of several articles in the literature on this topic include eight different wavelengths.5,34,35,38,40-43 Average power ranged between 0.9 W and 7 W. Five different spot sizes were used (200 µm to 500 µm), all resulting in a wide range of laser fluences with no consistency.
Monteiro and colleagues measured the lateral thermal damage at tissue margins caused by four different wavelengths versus an electrosurgical device and scalpel. As might be expected, the most tissue damage was caused by the electrosurgical device, followed by the diode, Nd:YAG, CO2, and erbium lasers, respectively, and the steel blade. In addition, the most regular incision was created by the scalpel, followed by the CO2, erbium, and Nd:YAG lasers, respectively, and electrosurge, with the diode laser causing the most irregular incision.5 Despite the desire to minimize thermal artifacts, there may be benefit to using either a "hot" glass tip or more thermal laser, such as a diode or Nd:YAG. Employing a device that has the potential for increased thermal effects may allow clinicians to treat patients with clotting disorders.40,44,45
Enhancing Biopsy Collection
Despite the challenges mentioned above, adequate biopsy collection is achievable with dental lasers. The following suggestions by the authors may be implemented to enhance this procedure:
Local anesthetic should be injected at least 1 cm from the closest border of the lesion to minimize the potential for artifacts.
Since hemostasis is usually achieved and sutures are rarely necessary, laser incisions do not need to be elliptical.
Unless the lesion is quite large or appears malignant, laser biopsy should be excisional and include at least 1 mm of healthy tissue beyond the border. Because of the width of most laser fibers, the incision should start at least 1.5 mm to 2 mm from the border to achieve this goal.
The tissue to be removed should be secured with an orienting suture or tissue forceps, with care taken so as not to crush the sample.
If the laser being used does not feature an aerosolized water spray as part of its design, the surgical site should be irrigated with water or saline to minimize thermal artifacts.
Because most dental lasers are quite efficient, consideration should be given to the basic tenet of using the lowest power to achieve the desired results. Generally, 1 W to 2 W, preferably in an ultrashort pulsed mode, should suffice.
If charring becomes noticeable, steps should be taken immediately to minimize or eliminate the thermal effects. This can be accomplished by decreasing the average power, increasing the thermal relaxation time (the time the laser is not firing between pulses), lowering the pulses per second (Hz), and increasing the water spray or tissue irrigation.
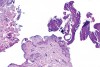
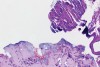
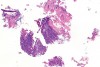
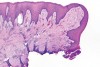
In Figure 1 through Figure 6, a variety of tissue biopsies collected via laser excision are presented. Figure 1 demonstrates the potential cellular distortion to the basement membrane region precluding detection of premalignant change. Figure 2 through Figure 4 highlight the cauterization and separation artifact caused in part by use of continuous wave versus pulsed mode. Figure 5 and Figure 6 demonstrate that by employing the advantages of laser surgical biopsy, it is possible to cleanly collect the specimen while preserving the marginal integrity of the area of suspected pathology without distortion.
Conclusion
Among the various methods of surgical biopsy collection, dental lasers present an option with multiple advantages. They enable the operator to achieve precise borders by sharply incising soft tissue. As a result, mechanical distortion to the tissue specimen can be minimized. Used by an experienced clinician with proper technique, a laser can abate obstacles such as mechanical distortion and imprecise borders that traditionally pose challenges to accurate diagnosis of the pathology. Laser biopsy allows efficient collection of soft-tissue specimens for pathological analysis and timely diagnosis.
About the Authors
Christopher J. Walinski, DDS
Associate Professor, Department of Dental Medicine, Touro College of Dental Medicine, Hawthorne, New York
Keng-Liang Ou, MS, PhD
Professor, Department of Dentistry, Taipei Medical University-Shuang Ho Hospital, New Taipei City, Taiwan; Professor, Department of Oral Hygiene Care, Ching Kuo Institute of Management and Health, Keelung, Taiwan; Chairman, 3D Global Biotech Inc., New Taipei City, Taiwan
Tracy M. Liang
DDS Candidate, Class of 2020, Touro College of Dental Medicine, Hawthorne, New York
Duane R. Schafer, DDS, MS
Professor, Department of Oral Diagnostic Services, Virginia Commonwealth University School of Dentistry, Richmond, Virginia
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. American Academy of Oral and Maxillofacial Pathology. Policy on Excised Tissue. AAOMP website. 2013. http://www.aaomp.org/wp-content/uploads/2016/12/Policy_on_Excised_Tissue-Final-11-9-2013.pdf. Accessed April 22, 2020.
2. Oliver RJ, Sloan P, Pemberton MN. Oral biopsies: methods and applications. Br Dent J. 2004;196(6):329-333.
3. Holzapfel BM, Lüdemann M, Holzapfel DE, et al. Open biopsy of bone and soft tissue tumors: guidelines for precise surgical procedures [article in German]. Oper Orthop Traumatol. 2012;24(4-5):403-415.
4. Lawrence W Jr. Operative management of soft tissue sarcomas: impact of anatomic site. Semin Surg Oncol. 1994;10(5):340-346.
5. Monteiro L, Delgado ML, Garcês F, et al. A histological evaluation of the surgical margins from human oral fibrous-epithelial lesions excised with CO2 laser, diode laser, Er:YAG laser, Nd:YAG laser, electrosurgical scalpel and cold scalpel. Med Oral Patol Oral Cir Bucal. 2019;24(2):e271-e280.
6. Liboon J, Funkhouser W, Terris D. A comparison of mucosal incisions made by scalpel, CO2 laser, electrocautery, and constant-voltage electrocautery. Otolaryngol Head Neck Surg. 1997;116(3):379-385.
7. Bornstein MM, Winzap-Kälin C, Cochran DL, Buser D. The CO2 laser for excisional biopsies of oral lesions: a case series study. Int J Periodontics Restorative Dent. 2005;25(3):221-229.
8. Awadallah M, Idle M, Patel K, Kademani D. Management update of potentially premalignant oral epithelial lesions. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125(6):628-636.
9. Costa Mendes L, Laurencin-Dalicieux S, Paul C, et al. Vascular Nd:YAG laser: a therapeutic alternative for large venous lakes of the oral mucosa. J Eur Acad Dermatol Venereol. 2020;34(4):e164-e165.
10. Ize-Iyamu IN, Saheeb BD, Edetanlen BE. Comparing the 810nm diode laser with conventional surgery in orthodontic soft tissue procedures. Ghana Med J. 2013;47(3):107-111.
11. Palaia G, Pergolini D, D'Alessandro L, et al. Histological effects of an innovative 445 nm blue laser during oral soft tissue biopsy. Int J Environ Res Public Health. 2020;17(8):E2651.
12. Suter VGA, Altermatt HJ, Bornstein MM. A randomized controlled trial comparing surgical excisional biopsies using CO2 laser, Er:YAG laser and scalpel. Int J Oral Maxillofac Surg. 2020;49(1):99-106.
13. Vescovi P, Corcione L, Meleti M, et al. Nd:YAG laser versus traditional scalpel. A preliminary histological analysis of specimens from the human oral mucosa. Lasers Med Sci. 2010;25(5):685-691.
14. Seoane J, Caballero TG, Urizar JM, et al. Pseudodysplastic epithelial artefacts associated with oral mucosa CO2 laser excision: an assessment of margin status. Int J Oral Maxillofac Surg. 2010;39(8):783-787.
15. González-Mosquera A, Seoane J, García-Caballero L, et al. Er,CR:YSGG lasers induce fewer dysplastic-like epithelial artefacts than CO2 lasers: an in vivo experimental study on oral mucosa. Br J Oral Maxillofac Surg. 2012;50(6):508-512.
16. Chen S, Forman M, Sadow PM, August M. The diagnostic accuracy of incisional biopsy in the oral cavity. J Oral Maxillofac Surg. 2016;74
(5):959-964.
17. Romanos GE, Pelekanos S, Strub JR. Effects of Nd:YAG laser on wound healing processes: clinical and immunohistochemical findings in rat skin. Lasers Surg Med. 1995;16(4):368-379.
18. Ahad A, Tandon S, Lamba AK, Faraz F. Minimally invasive management of pericoronal abscess using 810 nm GaAlAs diode laser. Int J Laser Dent. 2015;4(3):79-82.
19. Zeinoun T, Nammour S, Dourov N, et al. Myofibroblasts in healing laser excision wounds. Lasers Surg Med. 2001;28(1):74-79.
20. Walinski CJ. Irritation fibroma removal: a comparison of two laser wavelengths. Gen Dent. 2004;52(3):236-238.
21. Moritz A, Schoop U, Goharkhay K, et al. Treatment of periodontal pockets with a diode laser. Lasers Surg Med. 1998;22(5):302-311.
22. Coleton S. Lasers in surgical periodontics and oral medicine. Dent Clin North Am. 2004;48(4):937-962.
23. Goharkhay K, Moritz A, Wilder-Smith P, et al. Effects on oral soft tissue produced by a diode laser in vitro. Lasers Surg Med. 1999;25(5):401-406.
24. Fornaini C, Merigo E, Sozzi M, et al. Four different diode lasers comparison on soft tissues surgery: a preliminary ex vivo study. Laser Ther. 2016;25(2):105-114.
25. Pirnat S. Versatility of an 810 nm diode laser in dentistry: an overview. J Laser Health Acad. 2007;4:1-9.
26. Chawla K, Lamba AK, Faraz F, et al. Diode laser for excisional biopsy of peripheral ossifying fibroma. Dent Res J (Isfahan). 2014;11(4):525-530.
27. Merigo E, Clini F, Fornaini C, et al. Laser-assisted surgery with different wavelengths: a preliminary ex vivo study on thermal increase and histological evaluation. Lasers Med Sci. 2013;28(2):497-504.
28. Jerjes W, Upile T, Hamdoon Z, et al. CO2 laser of oral dysplasia: clinicopathological features of recurrence and malignant transformation. Lasers Med Sci. 2012;27(1):169-179.
29. Suter VG, Altermatt HJ, Dietrich T, et al. Pulsed versus continuous wave CO2 laser excisions of 100 oral fibrous hyperplasias: a randomized controlled clinical and histopathological study. Lasers Surg Med. 2014;46(5):396-404.
30. Wilder-Smith P, Arrastia AM, Liaw LH, Berns M. Incision properties and thermal effects of three CO2 lasers in soft tissue. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79(6):685-691.
31. Yagüe-García J, España-Tost AJ, Berini-Aytés L, Gay-Escoda C. Treatment of oral mucocele-scalpel versus CO2 laser. Med Oral Patol Oral Cir Bucal. 2009;14(9):e469-e474.
32. Romeo U, Libotte F, Palaia G, et al. Histological in vitro evaluation of the effects of Er:YAG laser on oral soft tissues. Lasers Med Sci. 2012;27(4):749-753.
33. Zaffe D, Vitale MC, Martignone A, et al. Morphological, histochemical, and immunocytochemical study of CO2 and Er:YAG laser effect on oral soft tissues. Photomed Laser Surg. 2004;22(3):185-189.
34. Suter VG, Altermatt HJ, Bornstein MM. A randomized controlled clinical and histopathological trial comparing excisional biopsies of oral fibrous hyperplasias using CO2 and Er:YAG laser. Lasers Med Sci. 2017;32(3):573-581.
35. Bargiela-Pérez P, González-Merchan J, Díaz-Sanchez R, et al. Prospective study of the 532 nm laser (KTP) versus diode laser 980 nm in the resection of hyperplastic lesions of the oral cavity. Med Oral Patol Oral Cir Bucal. 2018;23(1):e78-e85.
36. Romeo U, Del Vecchio A, Ripari F, et al. Effects of different laser devices on oral soft tissues: in vitro experience. J Oral Laser Appl. 2007;
7(3):155-159.
37. Lauritano D, Lucchese A, Gabrione F, et al. The effectiveness of laser-assisted surgical excision of leukoplakias and hyperkeratosis of oral mucosa: a case series in a group of patients. Int J Environ Res Public Health. 2019;16(2). doi: 10.3390/ijerph16020210.
38. Palaia G, Tenore G, Tribolati L, et al. Evaluation of wound healing and postoperative pain after oral mucosa laser biopsy with the aid of compound with chlorhexidine and sodium hyaluronate: a randomized double blind clinical trial. Clin Oral Investig. 2019;23(8):3141-3151.
39. Handlers JP. Diagnosis and management of oral soft-tissue lesions: the use of biopsy, toluidine blue staining, and brush biopsy. J Calif Dent Assoc. 2001;29(8):602-606.
40. Romeo U, Russo C, Palaia G, et al. Biopsy of different oral soft tissues lesions by KTP and diode laser: histological evaluation. ScientificWorldJournal. 2014:761704. doi: 10.1155/2014/761704.
41. Bombeccari GP, Garagiola U, Candotto V, et al. Diode laser surgery in the treatment of oral proliferative verrucous leukoplakia associated with HPV-16 infection. Maxillofac Plast Reconstr Surg. 2018;40(1):16.
42. Ahad A, Tandon S, Lamba AK, et al. Diode laser assisted excision and low level laser therapy in the management of mucus extravasation cysts: a case series. J Lasers Med Sci. 2017;8(3):155-159.
43. Calisir M, Ege B. Evaluation of patient perceptions after frenectomy operations: a comparison of neodymium-doped yttrium aluminum garnet laser and conventional techniques in the same patients. Niger J Clin Pract. 2018;21(8):1059-1064.
44. Palaia G, Del Vecchio A, Impellizzeri A, et al. Histological ex vivo evaluation of peri-incisional thermal effect created by a new-generation CO2 superpulsed laser. ScientificWorldJournal. 2014:345685. doi:10.1155/
2014/345685.
45. Romeo U, Libotte F, Palaia G, et al. Histological in vitro evaluation of the effects of Er:YAG laser on oral soft tissues. Lasers Med Sci. 2012;27(4):749-753.