You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Osteocytes and bone lining cells (BLCs) play important roles in bone remodeling.Osteocytes are pivotal mechano-sensing cells that regulate bone mass via the coordination of bone forming osteoblasts and bone resorbing osteoclasts.1 While osteoblasts are derived from mesemchymal stem cells and synthesize new bone matrix, osteoclasts are terminally differentiated multinucleated cells from the monocyte-macrophage lineage.2 BLCs are the cells responsible for bone formation on new bone surfaces and are characterized by a flat-shaped architecture found primarily in cancellous bone.3,4 Various studies have shown that growth factors and cytokines are responsible for inducing the proliferation of BLCs and osteoblasts prior to bone formation under specific osteogenic signals.5,6 These external factors, including parathyroid hormone, bone morphogenetic proteins, fibroblast growth factor-2, as well as mechanichal loading, induce BLCs to exit the quiescent stage into an active functional phase.2,5,7 The presence of BLCs observed histologically on either a bone surface or bone biomaterial (bone graft or dental implant) indicates a strong osteogenic potential and often is regarded as a major signal of bone biomaterial integration.3
Loading and Bone Resorption
Osteocytes and BLCs form a working communication system that regulates their mechano-receptive function.1 Osteocytes sense a slight increase in mechano-signaling (which may be created by loading) and signal BLCs to increase bone formation and inhibit resorption.8,9 Conversely, bone disuse that is absent from mechanical loading is an example of low-strain-generated signaling with bone loss as a consequence. Moreover, if excessive loading or microdamage are present, higher osteocyte apoptosis may be observed.2-4,6 Osteocyte apoptosis is generally seen nearest to the cortical bone surface under high mechanical strain, because the bone surface mechanically is more sensitive when compared to the trabecular interior portion of bone.10,11
This theory is clinically highly relevant, and the osseodensification concept performed on dense cortical bone is more prone to mechanical sensivity/high strain/bone loss discussed later in this article. As a result, apoptosis of osteocytes and subsequent bone loss may follow.
Bone Remodeling Around Dental Implants
Shortly after dental implant placement, a sequence of immuno-inflammatory responses, followed by angiogenic and osteogenic events, takes place. This sequence is primarily influenced by the implant surface characteristics, including surface topography, chemistry, and material composition, which either facilitate or prevent the adsorption of proteins onto the implant surface. Accordingly, thrombin and fibrinogen adsorb to the implant surface and play a key role in the early homeostasis as the release of cytokines and growth factors stimulates future collagen matrix deposition around the titanium oxide layer of the implant, leading to newly formed woven bone (usually occurring by 5 days). In a matter of 8 to 12 weeks, lamellar bone initiates the biological stability, namely osseointegration.12
Afterwards, as with natural dentition, implants are subject to soft- and hard-tissue remodeling where the average biologic width around dental implants has been reported at approximately 3.5 mm.13 Much research has suggested that the microgap in a two-piece implant is associated with greater crestal bone loss.14 The connection between abutment and implant generally leaves a space of between 10 micrometers (µm) and 50 µm and, as a result, the pumping effect of a liquid contained in the implant cavity may facilitate bacterial colonization in the gap with gram-positive and gram-negative bacteria.14 This has led investigators to study the effects of internal implant connections versus external ones.15 Furthermore, study on the effects of abutment-to-implant connection (platform switching) on the impact of biologic widths around implants has been a topic of intense discussion in recent years.16
While continuous research has strived to optimize implant design either to improve implant stability and/or osseointegration or minimize marginal bone loss, much less attention has been placed on optimizing the implant site during osteotomy preparation. The following sections review the literature that investigates the effect of initial bone density on primary implant stability. Later, various surgical approaches are discussed, including the concept of osseodensification as a novel surgical method designed to improve initial primary implant stability by condensing/compacting available autogenous bone.
Effect of Initial Bone Quality on Osseointegration and Implant Torque Values
Implant insertion torque (IT) values in the range of 25 Ncm to 45 Ncm have been suggested as ideal during implant placement to prevent the micro-movement that could potentially lead to fibrous encapsulation.17 In contrast, high IT has been associated with a critical pressure that triggers microfractures and bone necrosis. Various animal models have shown that high IT can elicit complex microdamage that initiates bone remodeling. Moreover, cortical bone resorption occurs more rapidly compared to trabecular bone, which is due to its lower vascularity.18 These previous animal findings are in agreement with a radiographic and histomorphometrical human study that clearly revealed that implants placed with a high IT (>50 Ncm) may be subject to greater peri-implant bone loss in the early stages of healing compared to those placed more passively.19 A recent multiscale analysis also revealed that the effect of under-drilling an implant osteotomy to achieve high IT resulted in a double layer of apoptotic osteocytes when compared to low IT.20 Therefore, previous studies seem to indicate that a lack of primary stability may potentially jeopardize osseointegration, yet high IT might also compromise the long-term maintenance of peri-implant bone levels.17
An equally important factor affecting implant placement is the effect of initial alveolar bone density on primary stability. Early publications by Misch in the field of implantology classified alveolar bone ridges into four major types.21 Accordingly, denser bone is located in the anterior mandible, whereas more porous trabecular bone is found in the posterior maxilla. Though cortical bone provides greater compressive strength and higher initial implant stability, it is important to note that the lesser vascularity of compact bone impacts not only the angiogenic potential of the bone around the implant but also reduces the capacity for osteogenic cells to migrate to the site, which in turn may result in lower bone turnover and potential peri-implant bone loss.
Simons et al performed a study investigating the proportion of cancellous/cortical bone on marginal bone loss following implant placement.22 They noted that a higher proportion of cancellous bone, ie, >50% to 60%, significantly minimized early bone loss (~0.6 mm to 0.7 mm) when compared to implant recipient sites that had less cancellous bone than this and greater amounts of cortical content (~1.5 mm). It is, therefore, generally recommended that high IT should be minimized, particularly in the presence of a thick cortical layer. To avoid bone loss at the crestal region, Simons et al recommended tapping the osteotomy site to minimize high IT values at sites with thick cortical plates.22 (Editor's note: A second article on the osseodensification preparation technique highlights the clinical indications describing its use.)
In summary, cortical bone presents with several disadvantages compared to trabecular bone, including its limited blood supply and osteoprogenitor cell presence.23 Furthermore, the crestal bone has the highest strain levels yet is most mechanically sensitive.10,11 As a result, osteocyte apoptosis (caused by microdamage and high strain) is more common in cortical bone and promotes a more potent resorptive signal (greater and faster bone loss).10,11 For these combined reasons, cortical crestal bone is a risk factor during implant placement, and the initial bone quality and characterization must be considered.
Outcomes in Immediate Implants Under High and Low Torque
Because high primary stability is believed to lead to better osseointegration, a number of clinical studies have investigated this hypothesis. For immediate implant placement with or without immediate loading, primary stability certainly is needed (>32 Ncm).24,25 Nevertheless, various research groups have shown that when implants are placed under high IT (≥50 Ncm), an increase in marginal bone loss and recession, notably in the presence of a thin buccal bone, may be observed.26 When extremely high IT values (>70 Ncm) are used during implant placement, it's been found that marginal bone loss was substantially higher.27 Similarly, failure rates are also increased when implant stability is too low at the time of implant placement.
These findings further demonstrate that initial torque values play a pivotal role in the fate of the peri-implant bone level. Interestingly, more than 20 years have passed since Summers demonstrated that bone compaction utilizing the osteotome technique to increase the primary stability of dental implants without removing bone tissue could potentially improve final bone healing.28-30 Nevertheless, caution is warranted with this technique because others, including Büchter et al and Stavropoulos et al, have demonstrated that it may also potentially lead to a compromise in bone healing due to the related microfractures within the peri-implant bone that are correlated with high insertion implant torque.31,32
For these reasons, various approaches with specific guidelines for implant placement have been highly debated over the years. Examples include the use of simplified drilling methods, such as placement of wider implants under higher IT values.26,27 When installed under higher IT, adequate secondary stability and high bone-to-implant contact (BIC) have been reported, though it was also reported that the healing process was slightly delayed due to initial necrosis of the existing bone.33 Similarly, findings from another research team also showed favorable results even when implants submerged at 0 Ncm torque demonstrated similar final outcomes to implants placed at 30 Ncm or 70 Ncm by 4 months.34 Hence, the impact of high IT values on peri-implant bone loss as compared to low IT remains yet uncertain.
Osseodensification
The biomechanical stability of implants typically has been dependent on several factors. At time of placement, implant macro and micro design, as well as the quality and quantity of surrounding bone, have been important parameters regarding initial implant stability (Figure 1).35 Over the years, several protocols to increase implant primary stability have been identified, including: (1) drilling protocol that involves under-preparation of the osteotomy; (2) implant type with regard to macrotexture and microtexture; (3) use of longer implants that provide greater BIC; and (4) techniques involving osseo-condensation of bone. Bone has long been considered an ideal tissue in the body because it possesses the ability to be flexible (absorbs energy); changes shape via deformation (without necessarily breaking or cracking); withstands compression, widening when this occurs; and is able to lengthen during tension.36
Typically, bone is cut prior to implant placement with the use of standard drill burs. While drill bits rotate, the cutting resistance vector has been described as "constantly changing," and this can cause the drill bit to shave along the bone surface away from the initial osteotomy site, inducing less-than-optimal site preparations (Figure 2 through Figure 4).37 Since fresh, hydrated trabecular bone is a ductile material, it has a good capacity for plastic deformation. Osseodensification is essentially a burnishing process that redistributes bone material on the bony surface through plastic deformation.37 The counterclockwise rotation of osseodensification burs causes the lands of the bur to slide across the surface of the bone by means of low plastic deformation that is purposefully designed to have a compressive force less than the ultimate strength of the bone.
For these reasons, osseodensification burs were created. As opposed to removing bone from the implant osteotomy site, the osseodensification concept utilizes compact autografting/condensation, which aims to maintain bone volume, resulting in higher initial bone density and better BIC following implant placement and accelerating bone healing. While, initially, higher bone stability can be achieved, an increase in residual strain that enhances osteogenic activity through mechanobiology may be observed. Increased implant stability leads to higher IT values and implant stability quotient (ISQ) values, thereby reducing micromotion. Caution must be taken to place implants within the ideal range.
Osseodensification burs create real-time haptic feedback, which informs the surgeon if more or less force is needed and allows immediate adjustments to be made to the advancing force, depending on the given bone density. These burs rotate in a counterclockwise direction and do not "cut" as expected with standard burs (Figure 5). They, therefore, densify bone by rotating in the noncutting direction (counterclockwise at 800 to 1,200 rotations per minute). Copious amounts of irrigation fluid should be used during the procedure to provide adequate lubrication between the bur and bone surfaces to minimize overheating. When used correctly, at the proper high speed and in a reversed, noncutting direction with steady external irrigation, a strong, dense layer of bone tissue can be formed along the walls and base of the osteotomy.
Since the osseodensification burs produce compression waves in which pressure is applied outwardly, bone laterally compresses during the continuous rotation and concurrent advancement of the bur. This facilitates "compaction autograft" or "osseodensification." During this process, bone debris is redistributed up the flutes of the bur and is pressed into the trabecular walls of the osteotomy. The autografting supplements the basic bone compression, and the condensation effect acts to further densify the inner walls of the osteotomy.38 Trisi et al studied the osseodensification technique in an animal model and found that osseodensification increased the percentage of bone density/BIC values around dental implants inserted in low-density bone with respect to conventional implant drilling techniques.38 They reported that osseodensification may play a role in enhancing implant stability by reducing micromotion.
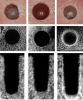
Huwais and Meyer also reported on the osseodensification technique and stated that the accumulation of autogenous bone along the internal component of the implant osteotomy sites promoted primary stability of the placed implant (Figure 6).39 The authors confirmed the essentiality for sufficient bone volume and density at the implant site (especially in low-density bone) in order to achieve good BIC and primary stability, both of which are crucial factors for osseointegration. The results from their study showed clearly that osseodensification significantly increased insertion and removal torques compared to standard drilling and extraction drilling. No significant differences in ISQ readings or temperature increases were demonstrated among the three groups, confirming the safety of osseodensification. It was concluded that by preserving bone using osseodensification, the healing process was accelerated and primary implant stability was increased while in situ autograft bone was maintained along the surface of the osteotomy site.
In summary, preliminary animal models have revealed a number of key findings. Osseodensification increases the bone mineral density around the periphery of the osteotomy and enhances primary stability by facilitating higher insertion and removal torques on the implant. Clinicians should, however, be cautioned not to over-compress bone, especially in the cortical region of the implant.40 (This concept is further addressed in detail in the aforementioned second article on this topic, which focuses on clinical use of osseodensification.) From a clinical safety perspective, osseodensification is similar to standard drilling when proper rotary speed, penetration speed, and irrigation are used. With osseodensification, bone is autografted through the reapplication of ground particles in a compacted manner along the entire depth of the osteotomy, particularly at its base. The process creates a smaller osteotomy than can be achieved with conventional drilling techniques due to recovery elastic strain when the osteotome is removed from the hole. The result of these characteristics is BIC values that have been reported to be three times higher for osseodensification when compared to standard drilling.38,39
Conclusions
The osseodensification preparation technique has been shown to preserve bone volume in two ways: via the compression of cancellous bone due to viscoelastic and plastic deformation, and compaction autografting of bone particles along the length and at the apex of the osteotomy. These strategies come after an array of previously discussed osteotomy techniques that compact bone through deformation. Also, impaction autografting has been used for more than two decades in the field of orthopedic surgery to improve stability of total hip replacements.41 The philosophy of these techniques differs from that of bone drilling, in which bone is removed. Instead, healthy bone is maintained within the osteotomy, especially in regions where bone density is already compromised.
About the Authors
Michael A. Pikos, DDS
Oral Maxillofacial Surgeon, CEO, Pikos Institute, Tampa, Florida
Richard J. Miron, Dr. med. dent., DDS, PhD
Adjunct Professor, Periodontology, Nova Southeastern University,
Fort Lauderdale, Florida
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Klein-Nulend J, Nijweide PJ, Burger EH. Osteocyte and bone structure. Curr Osteoporos Rep. 2003;1(1):5-10.
2. Florencio-Silva R, Sasso GR, Sasso-Cerri E, et al. Biology of bone tissue: structure, function, and factors that influence bone cells. Biomed Res Int. 2015;2015:421746. doi: 10.1155/2015/421746.
3. Miller SC, de Saint-Georges L, Bowman BM, Jee WS. Bone lining cells: structure and function. Scanning Microsc. 1989;3(3):953-960.
4. Bowman BM, Miller SC. The proliferation and differentiation of the bone-lining cell in estrogen-induced osteogenesis. Bone. 1986;7(5):351-357.
5. Matic I, Matthews BG, Wang X, et al. Quiescent bone lining cells are a major source of osteoblasts during adulthood. Stem Cells. 2016;34(12):2930-2942.
6. Chow JW, Wilson AJ, Chambers TJ, Fox SW. Mechanical loading stimulates bone formation by reactivation of bone lining cells in 13-week-old rats. J Bone Miner Res. 1998;13(11):1760-1767.
7. Donahue HJ, McLeod KJ, Rubin CT, et al. Cell-to-cell communication in osteoblastic networks: cell line-dependent hormonal regulation of gap junction function. J Bone Miner Res. 1995;10(6):881-889.
8. Huiskes R, Ruimerman R, van Lenthe GH, Janssen JD. Effects of mechanical forces on maintenance and adaptation of form in trabecular bone. Nature. 2000;405(6787):704-706.
9. van Oers RF, Ruimerman R, Tanck E, et al. A unified theory for osteonal and hemi-osteonal remodeling. Bone. 2008;42(2):250-259.
10. Jahani M, Genever PG, Patton RJ, et al. The effect of osteocyte apoptosis on signalling in the osteocyte and bone lining cell network: a computer simulation. J Biomech. 2012;45(16):2876-2883.
11. Adachi T, Aonuma Y, Taira K, et al. Asymmetric intercellular communication between bone cells: propagation of the calcium signaling. Biochem Biophys Res Commun. 2009;389(3):495-500.
12. Davies JE. Understanding peri-implant endosseous healing. J Dent Educ. 2003;67(8):932-949.
13. Tomasi C, Tessarolo F, Caola I, et al. Morphogenesis of peri-implant mucosa revisited: an experimental study in humans. Clin Oral Implants Res. 2014;25(9):997-1003.
14. Ujiie Y, Todescan R, Davies JE. Peri-implant crestal bone loss: a putative mechanism. Int J Dent. 2012;2012:742439. doi: 10.1155/2012/742439.
15. Berberi A, Maroun D, Kanj W, et al. Micromovement evaluation of original and compatible abutments at the implant-abutment interface. J Contemp Dent Pract. 2016;17(11):907-913.
16. Atieh MA, Ibrahim HM, Atieh AH. Platform switching for marginal bone preservation around dental implants: a systematic review and meta-analysis. J Periodontol. 2010;81(10):1350-1366.
17. Insua A, Monje A, Wang HL, Miron RJ. Basis of bone metabolism around dental implants during osseointegration and peri-implant bone loss. J Biomed Mater Res A. 2017;105(7):2075-2089.
18. Wang L, Ye T, Deng L, et al. Repair of microdamage in osteonal cortical bone adjacent to bone screw. PLoS One. 2014;9(2):e89343.
19. Duyck J, Corpas L, Vermeiren S, et al. Histological, histomorphometrical, and radiological evaluation of an experimental implant design with a high insertion torque. Clin Oral Implants Res. 2010;21(8):
877-884.
20. Cha JY, Pereira MD, Smith AA, et al. Multiscale analyses of the bone-implant interface. J Dent Res. 2015;94(3):482-490.
21. Misch CE. Bone classification, training keys to implant success. Dent Today. 1989;8(4):39-44.
22. Simons WF, De Smit M, Duyck J, et al. The proportion of cancellous bone as predictive factor for early marginal bone loss around implants in the posterior part of the mandible. Clin Oral Implants Res. 2015;26(9):1051-1059.
23. Kristensen HB, Andersen TL, Marcussen N, et al. Osteoblast recruitment routes in human cancellous bone remodeling. Am J Pathol. 2014;
184(3):778-789.
24. Trisi P, Perfetti G, Baldoni E, et al. Implant micromotion is related to peak insertion torque and bone density. Clin Oral Implants Res. 2009;20
(5):467-471.
25. Ottoni JM, Oliveira ZF, Mansini R, Cabral AM. Correlation between placement torque and survival of single-tooth implants. Int J Oral Maxillofac Implants. 2005;20(5):769-776.
26. Barone A, Alfonsi F, Derchi G, et al. The effect of insertion torque on the clinical outcome of single implants: a randomized clinical trial. Clin Implant Dent Relat Res. 2016;18(3):588-600.
27. Khayat PG, Arnal HM, Tourbah BI, Sennerby L. Clinical outcome of dental implants placed with high insertion torques (up to 176 Ncm). Clin Implant Dent Relat Res. 2013;15(2):227-233.
28. Summers RB. A new concept in maxillary implant surgery: the osteotome technique. Compendium. 1994;15(2):152-158.
29. Nkenke E, Kloss F, Wiltfang J, et al. Histomorphometric and fluorescence microscopic analysis of bone remodelling after installation of implants using an osteotome technique. Clin Oral Implants Res. 2002;13(6):595-602.
30. Nóbrega AR, Norton A, Silva JA, et al. Osteotome versus conventional drilling technique for implant site preparation: a comparative study in the rabbit. Int J Periodontics Restorative Dent. 2012;32(3):e109-e115.
31. Büchter A, Kleinheinz J, Wiesmann HP, et al. Biological and biomechanical evaluation of bone remodelling and implant stability after using an osteotome technique. Clin Oral Implants Res. 2005;
16(1):1-8.
32. Stavropoulos A, Nyengaard JR, Lang NP, Karring T. Immediate loading of single SLA implants: drilling vs. osteotomes for the preparation of the implant site. Clin Oral Implants Res. 2008;19(1):55-65.
33. Jimbo R, Janal MN, Marin C, et al. The effect of implant diameter on osseointegration utilizing simplified drilling protocols. Clin Oral Implants Res. 2014;25(11):1295-1300.
34. Rea M, Lang NP, Ricci S, et al. Healing of implants installed in over- or under-prepared sites-an experimental study in dogs. Clin Oral Implants Res. 2015;26(4):442-446.
35. Meyer U, Vollmer D, Runte C, et al. Bone loading pattern around implants in average and atrophic edentulous maxillae: a finite-element analysis. J Craniomaxillofac Surg. 2001;29(2):100-105.
36. Seeman E. Bone quality: the material and structural basis of bone strength. J Bone Miner Metab. 2008;26(1):1-8.
37. Bertollo N, Walsh WR. Drilling of bone: practicality, limitations and complications associated with surgical drill-bits. In: Klika V, ed. Biomechanics in Applications. London, UK: InTech; 2011.
38. Trisi P, Berardini M, Falco A, Podaliri Vulpiani M. New osseodensification implant site preparation method to increase bone density in low-density bone. Implant Dent. 2016;25(1):24-31.
39. Huwais S, Meyer EG. A novel osseous densification approach in implant osteotomy preparation to increase biomechanical primary stability, bone mineral density, and bone-to-implant contact. Int J Oral Maxillofac Implants. 2017;32(1):27-36.
40. Wang L, Wu Y, Perez KC, et al. Effects of condensation on peri-implant bone density and remodeling. J Dent Res. 2017;96(4):413-420.
41. Keblish PA, Neufeld SK, Varma C. Impaction autograft enhancement of femoral stem fixation in primary cementless THA. Orthopedics. 1996;19(9):759-762.