You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Disclosure
Joseph Choukroun, MD, is a shareholder of Process for PRF in Nice, France.
Periodontal plastic surgery plays a more important role in periodontics today. In the United States, by age 60 years, 90% of the population will have at least 1 tooth with 1 mm of recession and 40% of the same group will have at least 1 site with > 3 mm of recession.1,2 Untreated progressive recession leads to a worsening periodontal status and may result in dental and pulpal complications. Periodontal plastic surgery procedures with the goal of root coverage are a series of techniques performed to correct soft-tissue deformities that are of traumatic or plaque-induced origin. The ultimate goals of root coverage are resolution of the defect by a thicker keratinized and attached tissue, restoration of esthetically seamless tissue, and reestablishment of a functional attached tissue.3
When gingival recession is present on multiple adjacent teeth, one surgical treatment is recommended to treat the entire area.4 A connective-tissue graft from the palatal area used in combination with a pedicle flap (coronal- or lateral-positioned flap) is a common alternative for the treatment of gingival recession. Limiting factors (eg, insufficient amount of connective tissue, patient’s refusal to have a second surgical site, morbidity) have led to the use of substitutes for autogenous connective tissue.3,4 Allograft and xenograft dermal-tissue substitutes are typically utilized in periodontal plastic surgery, with successful clinical outcomes. Acting as a scaffold and allowing fibroconduction, the nature of the obtained attached tissue and the amount of the keratinized tissue have been controversial.4
Enamel matrix derivative (EMD) products of xenographic origins used as a “boosting” agent have shown positive clinical and histologic results when combined with a coronally advanced flap to induce periodontal regeneration. Clinical results are significant only when compared to a coronally positioned flap used alone.4 There is no evidence supporting the use of EMD when connective or allograft tissue grafts are utilized as control sites.
In the last decade, blood-derived biological modifiers have been used to enhance hard- and soft-tissue healing. Platelet-rich plasma (PRP) is the first generation of such “boosters.’’5 Lack of hard-tissue growth factors and physical properties, a difficult protocol, and fast release have contributed to its unpredictable results in maxillofacial hard- and soft-tissue reconstruction.6 The results for PRP application for root-coverage and mucogingival procedures have been equivocal.
In 2013, Choukroun introduced blood-derived growth factors known as advanced platelet-rich fibrin (APRF). The ease of use (single spin without the employment of an anticoagulant agent), presence of bone morphogenetic proteins, and beneficial physical properties make APRF one of the predominant blood-concentrated derived growth factors employed in maxillofacial surgery today.7
The purpose of this paper is to discuss benefits and clinical applications of APRF in periodontal plastic surgery, especially in root-coverage procedures.
Fundamental Bases: PRF and APRF
Platelet-rich fibrin (PRF) is the second-generation platelet concentrate prepared from a single centrifugation without the use of an anticoagulant. The PRF clot obtained is a physiological tridimensional matrix saturated with platelets and leukocyte. The alpha granules secreted by the platelets trapped in the membranes will secrete their growth factors for a natural and slow resorbtion rate of the membranes (15 days).8,9 The growth factors released are: platelet-derived growth factor (PDGF), transforming growth factor-b (TGF-b), vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), and insulin-like growth factor-1 (IGF-1).8-11
APRF7 is based on the same protocol; however, the tubule composition is changed (from plastic to glass to increase the chelation effect) (Figure 1) and the centrifugation time and spin are reduced (from 2700 RPM/12 minutes to 1300 RPM/8 minutes). By changing both those parameters, Choukroun et al7 found the APRF membranes contain an increase of VEGF (responsible for fast neovascularization), monocytes (responsible for BMP’s release), and extracellular matrix (fibronectin).
Omar et al12 and Rocher et al13 determined monocytes are an important source of autogenous bone morphogenetic protein-2 (BMP-2) and bone morphogenetic protein-7 (BMP-7), thus making APRF an improved and more potent blood-concentrated derived growth factor compared with PRF.
Periodontal Application of PRF/APRF
In Vitro Studies
Tsai et al14 investigated the biologic effects of PRF on human gingival fibroblasts, periodontal ligament cells, oral epithelial cells, and osteoblasts. A culture revealed PRF-stimulated cell proliferation (20% increase) and the proliferation rates of epithelium (due to the use of TGF-b) were reduced. This suggested PRF membranes could be beneficial for periodontal regeneration. He et al15 conducted a study to determine the effects of PRF on cell attachment, proliferation, and phosphorylation on human osteoblasts. PRF could increase osteoblast attachment and proliferation while concurrently up-regulating collagen-related protein production, all of which would effectively promote bone regeneration.
The findings of Kang et al16 strongly support the distinctiveness of PRF as a bioscaffold and reservoir of growth factors for tissue regeneration. PRF extract (PRFe) increased proliferation and migration, and promoted differentiation of the human alveolar bone-marrow stem cells. In addition, transplantation of fresh PRF into mouse calvaria enhanced regeneration of the critical-sized defect. In another study by Chang and Zhao,17 PRF activation has shown to bring about the expression of phosphorylated extracellular signal-regulated protein kinase (p-ERK) and osteoprotegerin (OPG), signifying its benefits for bone regeneration. Furthermore, Huang et al18 demonstrated PRF stimulates proliferation and differentiation of dental pulp cells by upregulating OPG and ALP expression. A more recent study17 concluded enhancements of p-ERK, OPG, and ALP expression by PRF may provide benefits for periodontal regeneration.
Studies have also attempted to use PRF as a regenerative material in the treatment of periodontal defects. Chang and Zhao17 in their case report assessed the clinical and radiographic changes in periodontal intrabony defects treated with PRF. They concluded PRF was an effective treatment modality, as the results showed its application exhibited pocket reduction and gain in clinical attachment, along with increased postoperative radiographic density in the treated defects. Sharma and Pradeep19 found greater reduction in probing depth, greater gain in periodontal attachment level, and greater bone fill in 3-wall intrabony defects treated with PRF and open-flap debridement (OFD) when compared to therapy with OFD alone. In another study,20 regarding the treatment of mandibular Grade II furcation defects, the same authors showed statistically significant improvement at sites treated with PRF and OFD as compared to those with OFD alone. However, in a study by Pradeep et al,21 which explored the clinical and radiographic effectiveness of autologous PRF and PRP in the treatment of intrabony defects in chronic periodontitis subjects, no significant differences were found between the 2 platelet concentrates.
Periodontal Plastic Procedures
A paucity of data exists in the literature. Aroca et al22 compared 20 patients receiving treatment with a coronally positioned flap with without PRF membranes. At 6 months, the test group benefited from an increased zone of keratinized tissue but a lower percentage of complete root coverage. Aleksic et al23 and Jankovic et al24 compared the connective tissue grafts to the PRF as graft material for the treatment of Miller Class 1 and Class 2 in 19 patients. At 6 months, no statistical difference was found between the groups when comparing the amount of root coverage and increased zone of keratinized tissue. The comfort of the procedure and postoperatory complications were statistically less with the PRF group because no intraoral donor site was used. Tunali and Ozdemir25 conducted a prospective split-mouth design comparing PRF and connective tissue graft in 20 patients for 12 months follow-up. Both treatment methods reduced significantly the amount of recession (76% and 77%, respectively) and increased the clinical attachment levels (2.90 mm and 3.04 mm, respectively).
Mechanism of Action
The major issue we found in analyzing the literature is the comparison of PRF/APRF with a connective tissue graft. Even though these two materials have similar physical characteristics, the modes of operation and final products obtained are different.
The connective tissue graft harvested from the palate will transfer its genetic expression (keratinization) from the donor site to the recipient location. Karring et al26 have described this concept using an animal model. The receipted site has no input in the quality or the quantity of the tissue obtained. We termed this mechanism of action as fibrogenesis.
The APRF model works from a different pathway. The fibrin should be considered as a biological matrix; with an increased source of VEGF and growth factors, APRF will promote and induce the formation of a new tissue from the receipted site. Thus, the quality of the receipted-bed soft tissue is crucial to the success of the procedure. If we have an existing band of keratinized tissue, we will promote the fabrication of more keratinized tissue. If, on the contrary, we have only a loose mucosal unattached tissue, we will promote the formation of the same kind of poor-quality tissue. We described this mechanism of action as fibropromotion.
For fibropromotion to occur, 2 conditions are needed: biotensegrity and volume. Those factors are valid for augmentation procedures involving the use of any biological biomodifiers. The inability to satisfy those 2 parameters will translate into a failure of APRF and, thus, a clinical disappointment.
Biotensegrity
Ingber,27 a physician from Harvard school of medicine, demonstrated the positive or negative forces (tension and pressure) generated at the surface of a cell are transferred via the actomyosin filaments complex through the cytoskeleton and are finally transmitted to the nucleus. Therefore, a balance exists between the extracellular forces and the intracellular compartment if the forces applied are within the envelope of tolerance of each group of cells, thus organs. Biotensegrity helps to guide force transmission and orchestrate multimolecular response to stress at all size scales in all organ systems. When the mechanical external forces applied on the cell surface (cell-surface mechanoreceptors) are beyond the scope of intracellular tolerance, an alteration in the cytoskeletal structure occurring inside the cell leads into an intracellular biochemistry and gene expression changes from the original programmed expression. One of the clinical translational applications of this concept is the impact of flap tension and neoangiogenesis. Mammoto et al28 showed in animal models that stretching the mucosa of mice reduced the amount of VEGF produced, and, thus, a vascular reduction of the flap was observed. Pini Prato et al29 confirmed this hypothesis by conducting a randomized controlled clinical study to measure the tension of the coronally advanced flap before suturing and to compare the reduction in recession following coronally advanced flap therapy with or without tension. The statistical analysis showed minimal flap tension (0.0 g to 0.4 g) favored recession reduction, while a higher tension of the flap (4 g to 7 g) was associated with lower recession reduction. This negative impact of flap tension is observed in maxillofacial bone reconstruction. Marx et al30 attributed the success of their vertical bone augmentation to a passive, nontensile flap that maintained the cellular integrity of the bone graft.
Volume
To quantify any clinically relevant fibropromotion by the use of APRF, a certain quantity of platelets needs to be harvested so that a given number of growth factors is released. These growth factors (see above) embedded into the tight and well-organized fibrin mesh will be released “drop by drop” so to ensure a constant and stable flow for the next 12 to 15 days (resorption rate of the APRF membranes). For this reason, the quality and quantity of soft tissue obtained after the surgery is directly related to the amount of fibrin matrix grafted.
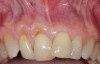
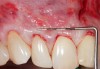
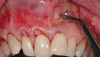
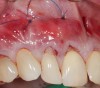
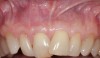
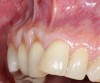
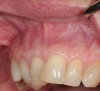
In a quantitative histomorphometric analysis of cell penetration, Gahnaati et al7 evaluated the composition of APRF membranes. Sixty-eight percent of the APRF membranes were colonized by the platelet-derived growth factors harvested by the samples. These findings, correlated with the authors’ clinical experience, led to a conclusion that 3 to 4 membranes are needed per pair of teeth to clinically observe a fibropromotion (Figure 2 through Figure 4).
Clinical Translation
The surgical technique proposed is a simplification of the vestibular incision subperiosteal technique access31 and an improvement of the tunnel technique.32
Incision
The vertical mucosal incision will allow a horizontal (mesiodistal) and apicocoronal full-thickness flap instrumentation, resulting in a total relaxation and passive coronal displacement of the mucogingival papillary complex (Figure 5 and Figure 6).
Root Preparation and Decontamination
The following are the same indications already proposed in periodontal muccogingival procedures. A thorough root preparation will create a flat or negative root surface, allowing for more volume of APRF placed and lesser flap tension, leading to a minimal APRF-membrane resorption (biotensegrity).
The root decontamination using ethylenediaminetetraacetic acid (EDTA) 17% (double application of 2.5 minutes) will remove the smear layer created by the root planing/preparation and will enable the collagen fibers of the tubule to be exposed and thus improve the quality of the type of attachment expected.
Volume Packing
For clinical significance, clinicians should use a minimum of 3 to 4 membranes per pair of teeth. A “backpacking concept” from the distal to mesial aspects will provide a homogenous density of APRF membrane volume (Figure 7).
As a result of the flap relaxation and dense backpacking, the flap will displace itself physiologically coronally without tension or pull. This tension-free concept is the core of the clinical application of biotensegrity.
Suturing
Apical periosteal mattress sutures are the core of the suturing technique and have several purposes. The goals are to avoid marginal suture tension on the APRF membranes, stabilize and maintain the membranes on the roots’ surface, and circumvent any traveling of the membranes into the mucosal area (Figure 8).
The clinical reproducibility of the root-coverage procedure associated with an increase of the zone of keratinized tissue depends on (Figure 9 through Figure 11) several factors.
One is the comprehensive understanding of the APRF mechanism of action and biological principle. The other is the precise execution of the surgical technique that relies on flap relaxation, easy access to membranes packing and positioning on roots (volume), physiologic displacement of the flap (biotensegrity), and passive closure without tension or sutures.
Conclusion
The fibrin-assisted soft-tissue promotion concept for the purpose of root coverage using APRF is a breakthrough in mucogingival procedures; the fundamentals of such a concept reside in the understanding of the established wound-healing principles in medicine and the clinical translation of those ideas. This innovative model will need more clinical and histologic studies to validate its long-term integration because only 2 years of data are available.
About the Authors
Alexandre-Amir Aalam, DDS
Clinical Assistant Professor
Herman Ostrow School of Dentistry
University of Southern California
Diplomate
American Board of Periodontology
Diplomate
American Board of Oral Implantology
Private Practice
Los Angeles California
Alina Krivitky Aalam, DDS
Clinical Assistant Professor
Herman Ostrow School of Dentistry
University of Southern California
Diplomate
American Board of Periodontology
Private Practice
Los Angeles, California
Joseph Choukroun, MD
Private Practice
Nice, France
References
1. Miller AJ, Brunelle JA, Carlos JP, et al. Oral Health of United States Adults. The National Survey of Oral Health in U.S. Employed Adults and Seniors: 1985-1986, National Findings. Bethesda, MD: US Department of Health and Human Services; 1987:3-11, 69-98. NIH publication 87-2868.
2. Albandar JM, Kingman A. Gingival recession, gingival bleeding, and dental calculus in adults 30 years of age and older in the United States, 1988-1994. J Periodontol. 1999;70(1):30-43.
3. Parameter on mucogingival conditions. American Academy of Periodontology. J Periodontol. 2000;71(5 suppl):861-862.
4. Tatakis DN, Chambrone L, Allen EP, et al. Periodontal soft tissue root coverage procedure: a consensus report from the AAP regeneration workshop. 2015;86(2 suppl):S52-S55.
5. Marx RE. Platelet-rich plasma: evidence to support its use. J Oral Maxillofac Surg. 2004;62(4):489-496.
6. Hamdan AA, Loty S, Isaac J, et al. Platelet-poor plasma stimulates proliferation but inhibits differentiation of rat osteoblastic cells in vitro. Clin Oral Implants Res. 2009;20(6):616-623.
7. Ghanaati S, Booms P, Orlowska A, et al. Advanced platelet-rich fibrin: a new concept for cell-based tissue engineering by means of inflammatory cells. J Oral Implantol. 2014;40(6):679-689.
8. Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e37-e44.
9. Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologic features. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e45-e50.
10. Dohan DM, Choukroun J, Diss A, et al. Platelet-rich fibrin (PRF): a second generation platelet concentrate. III. Leukocyte activation: a new feature for platelet concentrates? Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e51-e55.
11. Choukroun J, Diss A, Simonpieri A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: clinical effects on tissue healing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101(3):e56-e60.
12. Omar OM, Granéli C, Ekström K, et al. The stimulation of an osteogenic response by classical monocyte activation. Biomaterials. 2011;32(32):8190-8204.
13. Rocher C, Singla R, Singal PK, et al. Bone morphogenic protein 7 polarizes THP-1 cells into M2 macrophages. Can J Physiol Pharmacol. 2012;90(7):947-951.
14. Tsai CH, Shen SY, Zhao JH, Chang YC. Platelet-rich fibrin modulates cell proliferation of human periodontally related cells in vitro. J Dent Sci. 2009;4(3):130-135.
15. He L, Lin Y, Hu X, et al. A comparative study of platelet-rich fibrin (PRF) and platelet -rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(5):707-712.
16. Kang YH, Jeon SH, Park JY, et al. Platelet-rich fibrin is a bioscaffold and reservoir of growth factors for tissue regeneration. Tissue Eng Part A. 2011;17(3-4):349-359.
17. Chang YC, Zhao JH. Effects of platelet-rich fibrin on human periodontal ligament fibroblasts and application for the periodontal intrabony defects. Aust Dent J. 2011;56(4):365-371.
18. Huang FM, Yang SF, Zhao JH, Chang YC. Platelet-rich fibrin increases proliferation and differentiation of human dental pulp cells. J Endod. 2010;36(10):1628-1632.
19. Sharma A, Pradeep AR. Treatment of 3-wall intrabony defects in patients with chronic periodontitis with autologous platelet rich fibrin: a randomized controlled clinical trial. J Periodontol. 2011;82(12):1705-1712.
20. Sharma A, Pradeep AR. Autologous platelet rich-fibrin in the treatment of mandibular degree 2 furcation defects: a randomized clinical trial. J Periodontol. 2011;82(10):1396-1403.
21. Pradeep AR, Rao NS, Agarwal E, et al. Clinical evaluation of autologous platelet-rich fibrin and platelet-rich plasma in the treatment of intrabony defects in chronic periodontitis: a randomized controlled clinical trial. J Periodontol. 2012;83(12):1499-1507.
22. Aroca S, Keglevich T, Barbieri B, et al. Clinical evaluation of a modified coronally advanced flap alone or in combination with a platelet-rich fibrin membrane for the treatment of adjacent multiple gingival recessions: a 6-month study. J Periodontol. 2009;80(2):244-252.
23. Aleksić Z, Janković S, Dimitrijević B, et al. The use of platelet-rich fibrin membrane in gingival recession treatment. Srp Arth Celok Lek. 2010;138(1-2):11-18.
24. Jankovic S, Aleksic Z, Klokkevold P, et al. Use of platelet-rich fibrin membrane following treatment of gingival recession: a randomized clinical trial. Int J Periodontics Restorative Dent. 2012;32(2):41-50.
25. Tunali M, Ozdemir H. Evaluation of autologous platelet-rich fibrin in the treatment of multiple adjacent gingival recessions. A 12-month study. Int J Periodontics Restorative Dent. 2015;35(1):105-114.
26. Karring T, Lang NL, Löe H. The role of connective tissue in determining epithelial differentiation. J Periodontal Res.1975;10(1):1-11.
27. Ingber DE. Tensegrity-based mechanosensing from macro to micro. Prog in Biophys Mol Bio. 2008;97(2-3):163-179.
28. Mammoto T, Jiang A, Jiang E, Mammoto A. Platelet rich plasma extract promotes angiogenesis through the angiopoietin 1-Tie2 pathway. Microvasc Res. 2013;89:15-24.
29. Pini Prato G, Pagliaro U, Baldi C, et al. Coronally advanced flap procedure for root coverage. Flap with tension versus flap without tension: a randomized controlled clinical study. J Periodontol. 2000;71(2):188-201.
30. Marx RE, Schellenberger T, Wimsatt J, Correa P. Severely resorbed mandible: predictable reconstruction with soft tissue matrix expansion (tent pole) grafts. J Oral Maxillofac Surg. 2002;60(8):878-888.
31. Zadeh HH. Minimally invasive treatment of maxillary anterior gingival recession defects by vestibular incision subperiosteal tunnel access and platelet-derived growth factor BB. Int J Periodontics Restorative Dent. 2011;31(6):653-660.
32. Stimmelmayr M, Allen EP, Gernet W, et al. Treatment of gingival recession in the anterior mandible using the tunnel technique and a combination of epithelialized-subepithelial connective tissue graft—a case series. Int J Periodontics Restoratve Dent. 2011;31(2):165-173.