You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Understanding the etiology, prevention, and management of oral infections helps facilitate oral health maintenance. When an acute infection occurs, it is prudent to resolve it promptly, because an infection can lead to troubling morbidity.
The word infection is used to describe a variety of conditions; therefore, several terms are defined to clarify intraoral situations. Basically, when there is an immune response to a bacterial challenge, it is considered an infection.1 Subclinical infections commonly occur in pockets around teeth and dental implants. The term subclinical connotes that the immune response has controlled the bacterial challenge and there are no clinical signs of an infection (eg, redness, swelling).1 If the inflammatory process is not thwarted, tissue destruction occurs and the situation is regarded as a disease.1 For example, gingivitis, which histologically demonstrates an increased number of inflammatory cells and collagen-poor areas, is considered a disease.2 Similarly, periodontitis, peri-implant mucositis, and peri-implantitis are all diseases associated with chronic infections of the periodontium or peri-implant tissues. These chronic conditions, however, are not associated with general signs or symptoms of infection or bacteremia (eg, fever, malaise).3,4 In contrast, acute infections—eg, periodontal or peri-implant abscess, cellulitis—that can occur in the oral cavity may be characterized as dynamic, polymicrobial (mixed aerobic and anaerobic bacteria), opportunistic, usually painful, and may result in dysfunction.5,6 This article covers diagnosis and management of these latter conditions and reviews relevant information.
Manifestations of Acute Infections and Associated Bacteria
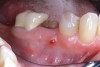
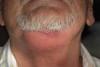
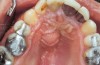
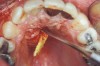
Manifestations of acute infection include abscess and cellulitis. Abscess is an accumulation of pus in an enclosed space7 (Figure 1), while cellulitis is a diffuse inflammatory process within tissues, which may manifest edema, redness, pain, and interference with function (Figure 2). With cellulitis, the infection may be caused by various organisms, and it typically occurs in loose tissues beneath the skin or mucous membranes or around muscle bundles or surrounding organs.7 Also with cellulitis, the severity of the infection is initially determined by virulence of invading bacteria and the host’s response.
Oral infections are usually polybacterial. Approximately 5% are due to aerobic bacteria, 35% are caused by anaerobic bacteria, and 60% are triggered jointly by aerobes and anaerobes.8 When aerobic bacteria are involved, the most common offending agents are gram-positive aerobic cocci (eg, streptococci spp, staphylococci spp). If there is an anaerobic or mixed infection, bacteria usually found are anaerobic cocci (eg, streptococci spp, peptostreptococci spp) and gram-negative anaerobic bacilli (Prevotella spp, Porphyromonas spp, Bacteroides spp, and Fusobacterium spp).8 In general, anaerobic bacteria are the main etiological agents causing orofacial infections. The following is a common scenario with respect to initiating an infection: After inoculation of tissue with microbes, virulent and invasive aerobic bacteria proliferate and decrease the tissue’s oxidation-reduction potential. This creates an environment conducive for multiplication of anaerobic bacteria, which will predominate during the suppurative phases of an infection.8
Incidences of Infections Post-Surgically: Correlation to Antibiotic Use
Infections may involve soft tissue and bone, and their occurrence varies after different surgical procedures. Following periodontal procedures, their incidence ranges from 1% to 5.4%, according to studies by Pack and Haber9 and Checchi et al.10 In the same studies, individuals administered antibiotics before, during, or after surgery had an infection rate that varied from 2.33% to 5.4 %.9,10 These data indicate that among patients undergoing routine periodontal surgical procedures, there is no difference in the incidence of infections among patients who receive or do not receive antibiotics. However, it should be noted that these patients were randomly selected and do not necessarily reflect specific types of situations. According to Powell et al,11 subsequent to implant therapy there was a 1.14% (2/175 procedures) chance of patients experiencing an infection after stage 1 and stage 2 therapies. The complication rate was so low after implant surgery, these authors questioned whether clinicians actually need to prophylactically provide antibiotics for 1 week after a “routine” surgical implant procedure.
The aforementioned studies question the need to routinely prescribe antibiotics prophylactically. Furthermore, there is debate in the literature concerning whether antibiotics should be used before or after routine dental implant placement. A systematic review (six studies) by Ahmed and Sadd12 addressed implant success rates (11,406 implants, n = >2600) with respect to antibiotic usage. The following success rates were reported 3 months after implant insertions: no antibiotic therapy, 92%; preoperative antibiotics, 96%; postoperative antibiotics, 97%; both pre- and postoperative antibiotics, 96.5%. They concluded that all therapies achieved at least a 90% success rate; therefore, there was no benefit associated with the prophylactic use of antibiotics in low- and moderate-risk cases. However, it was noted that 50% of the studies indicated there was a benefit to using antibiotics pre- or postoperatively as an adjunct to implant therapy. Furthermore, in the authors’ opinion, an 8% failure rate after 3 months is too high.
Contrastingly, Esposito et al13 recently published a systematic review addressing antibiotic usage with respect to dental implant placement. Based on this review, which included six randomized clinical trials with a follow-up of at least 3 months (N = 1162), the investigators reported that antibiotics are beneficial in reducing implant failure rates. Their data assessment indicated that antibiotic utilization would prevent one patient from early implant loss for every 25 individuals taking antibiotics. Specifically, 2 g amoxicillin given 1 hour before procedures increased implant success rates. It was also stated that the benefit of postoperative antibiotics is unknown.
Since there are contradicting conclusions from the two cited systematic reviews (ie, Ahmed and Sadd12 and Esposito et al13), clinicians must interpret the data for themselves. However, it is the authors’ opinion that if 50% of the studies in the first systematic review found there was a benefit with respect to using antibiotics, and the second review also demonstrated an advantage, it can be surmised that for routine dental implant placement, a preoperative dose of 2g of amoxicillin (or 600 mg clindamycin for penicillin-allergic patients) is a prudent protocol. Furthermore, there are many situations—eg, a complicated surgical procedure, bone graft placement, multiple dental implant insertion, a lengthy surgery, a medically compromised patient—when it is a judgment call by the clinician as to the necessity of prescribing antibiotics postoperatively for a prolonged period of time (eg, 1 week).
Concerning the incidence of infections after third-molar extractions among healthy individuals, Oomens and Forouzanfar14 assessed 24 studies to determine if there was a benefit with respect to prophylactically prescribing antibiotics. They concluded there was limited data to support this practice, and there is no consensus as to whether an adjunctive antibiotic reduces the occurrence of infections.14
Ultimately, the decision of whether to prescribe an antibiotic before or after various surgical procedures discussed above needs to be made with recognition that drug overuse has led to the development of resistant bacterial strains and unwanted side effects. Therefore, in each situation the clinician must assess the risk/benefit ratio of prescribing an antibiotic.
Inflammatory Response: After Surgery, Pathogens, or Irritants
Inflammation is a biological response by vascular tissues to detrimental stimuli (eg, after a surgical procedure, or irritants or pathogens).15 The presence of inflammation does not necessarily signify that there is a frank infection. Classically, signs of an inflammation include redness, heat, pain, swelling, and loss of function.15 However, these manifestations of inflammation are not the inflammatory response; the inflammatory reaction consists of sequential changes that can alter tissue function or structure.16 After cellular injury, injured cells leak their contents into extracellular spaces, and chemical mediators that regulate the inflammatory reaction, such as histamine and bradykinin, are released. These mediators are associated with amplified leukocyte migration, phagocytosis, and increased permeability of blood vessels. Hemodynamic alterations, such as vasodilatation, result in additional blood circulating to injured areas. Furthermore, normal blood supply between capillaries and the tissue is disturbed, and more fluid flows into the harmed tissue than out, causing edema. As a result of two processes—hemorrhage and edema—initial swelling can commence minutes to hours after injury.
Alterations listed above arise early after impairment and, depending upon wound severity, can persist for several days. Edema does not transpire equally in all directions from the site of injury. It is guided by muscle attachments, fascia, and structures, such as bone and teeth.17 The above tissue modifications may be associated with discomfort and reduced function.
Understanding Clinical Signs of an Infection
Sometimes, it is difficult to clinically differentiate between post-surgical edema and swelling due to an infection. In this regard, certain characteristics can be used to distinguish between them.
Edema is a common initial finding after a surgical procedure, and an infection does not usually manifest itself for at least 48 hours. Upon palpation, edema is usually soft and not painful and decreases after the initial swelling period (24 to 72 hours). In contrast, swelling associated with an infection can continue to get worse with time. An infected area also is often painful when palpated and may be accompanied by pus, fever, malaise, tender lymph nodes, and trismus; in addition, the site may be hot and begin to get harder (indurated).
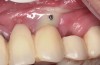
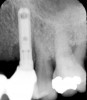
Suppuration is a cardinal sign of an infection, yet it may not be present. Pus is usually a thick viscous fluid that is often created in response to an infection. As the neutrophils die, they are destroyed by macrophages, thereby forming pus. Pus is usually white or yellow, but it can be red, green, or blue (Figure 3 and Figure 4).18 White and yellow pus typically reflect an infection with Streptococcus pyogenes or Staphylococous aureus.19 A red discharge reflects blood mixing with the pus. If the pus is green, it is due to incorporation of myleperoxidase, which is produced by neutrophils.19 A clear fluid (serum) reflects healing and is not considered a sign of an infection.19
Exudate is the medical term for drainage and can appear in a variety of forms. Its appearance is an indication of whether the wound is healing normally. The following types of discharges may be seen after surgery20:
Serous drainage—Typically, in the first 48 to 72 hours after an incision is made, a serous exudate is normal from a wound. It is clear and may be slightly yellow in appearance.
Serosanguinous drainage—This is a thin, watery drainage that is composed of both blood and serum. It may appear slightly pink from the red blood cells that are present, and it is normal in the early stages of healing.
Sanguinous drainage—The exudate has a larger amount of blood present than in serosanguinous drainage. This discharge is not typical of a healing wound.
Purulent discharge—Pus is not a normal finding after a surgical procedure.
Diagnosing an Infection
Clinical and radiographic evaluations are useful in diagnosing infection.
Visual inspection—During visual inspection, the face and neck should be examined for redness and asymmetry due to swelling, and the patient should be assessed intraorally for localized or diffuse swelling and tissue color alterations.
Palpate—The infected area can be gently palpated for fluctuance. If an infection is present, palpation usually elicits a painful response, and the submandibular and sublingual lymph nodes may be tender.
Radiograph evaluation—Radiographs can help determine if a tooth, implant, or sequestering bone are contributing to an infection. In specific situations—for example, various forms of osteomyelitis—infections may affect the appearance of the bone. A non-vital tooth can manifest a radiolucent apical area; sinus infections usually demonstrate a cloudy sinus; and other alterations of bone density or trabecular pattern may be associated with an infection.
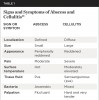
Distinguishing Between an Abscess and Cellulitis
Criteria to discern between an abscess and cellulitis are listed in Table 1.21
Differentiating Between Various Abscesses
A periodontal abscess usually manifests itself as red, painful swelling adjacent to a tooth, where it is typically associated with deep probing depths; a draining fistula is rarely present. In contrast, an endodontic abscess often demonstrates an inflamed fistulous tract, which is typically apical to the mucogingival junction. In a radiograph, there often is radiolucency at the apex of the tooth.
Peri-implant infections may be chronic or acute. An acute infection is similar to a periodontal abscess, but is usually circumferential around an implant.
Testing for Infection
Inspect incision line for pus—Subsequent to surgery, if the patient reports discomfort, it is prudent to assess for purulence. This can be done by pressing the tissue adjacent to an incision in a manner that would express suppuration through the incision line.
Culturing an infection—At the time of incision and drainage, it is sometimes recommended that a sample of pus or body fluid be obtained for culture and sensitivity testing. A culture can be performed to find out what type of microorganism(s) is causing an infection. A sensitivity test determines what kind of antibiotics will be effective. For localized dental abscesses, a culture is usually not taken. However, if cellulitis is present, it is recommended that a culture be obtained to have an exact diagnosis with respect to bacterial sensitivity to antibiotics.
Complete blood count or blood panel—Laboratory assessments can be ordered when dealing with infections that are recurring or atypical or may reflect an underlying disease. One of the components of a complete blood count (CBC) is an assessment of the number of white blood cells (WBC count). A decreased WBC count may be due to various conditions, such as bone marrow failure due to infection, radiation, or disease of the liver or spleen, etc.22 High numbers of WBCs—ie, leukocytosis—can result from infectious diseases, inflammatory disease (such as rheumatoid arthritis or allergy), leukemia, severe emotional or physical stress, and tissue damage (such as burns).22 The chief types of white blood cells are neutrophils, lymphocytes, monocytes, eosinophils, and basophiles. An increased number of neutrophils may indicate bacterial infection or an acute viral infection.
Treatment of Infections
The objective of antibiotic therapy is to decrease the bacterial challenge and to prevent additional complications due to the spread of an infection. Resolution of an infection may require systemic antimicrobials, surgery, or a combination of both modalities.
Non-Surgical Therapy
The pathogenicity of bacteria is enhanced due to synergism between two or more microorganisms that dominate the infectious process.23 Therefore, an antibiotic to control an infection must interfere with the synergism between key offending organisms, and it is not necessary to have a drug that is specific for every bacteria involved in the infection.23 An antibiotic is usually administered when a patient manifests signs of an infection. If there is no fluctuance, most antibiotics are prescribed for 7 to 10 days (Table 2). Typically, the patient’s physical response will reflect the efficacy of drug therapy. Also, compliance with respect to taking the drug is required.
Commonly Used Antibiotics for Treating Acute Oral Infections
Betalactams—eg, penicillin, ampicillin, amoxicillin—are broad-spectrum, bactericidal drugs and they are effective for most oral infections.24 However, betalactamase-generating bacterial strains have resulted in resistance to betalactam antibiotics, causing treatment failures.25-27 The combination of a betalactam antibiotic with a betalactamase inhibitor, such as amoxicillin plus clavulanic acid (Augmentin), furnishes activity against resistant bacteria.8,23 Penicillin VK and amoxicillin are often prescribed for odontogenic infections; however, some authors consider Augmentin to be the best antibiotic to control oral bacterial infections8,23 for individuals not allergic to penicillins. Cephalosporins are not very effective against gram-negative anaerobes and do not provide any benefit compared to penicillins.23
Macrolides—eg, erythromycin, azithromycin—are bacteriostatic antibiotics and they are not very effective against anaerobes belonging to Bacteroides, Fusobacterium, Porphyromonas, and Prevotella genera. Therefore, they are not considered efficacious for oral infections.23
Tetracyclines are bacteriostatic and broad spectrum; however, they are not the drugs of choice for oral infections,23 because about 12% of oral bacteria are resistant to tetracyclines.28
Metronidazole is specific for anaerobic gram-negative bacteria. It is often prescribed in combination with other antibiotics—eg, amoxicillin with and without clavulanic acid.23
Clindamycin is effective against anaerobic, aerobic gram-positive, and betalactamase-producing organisms. In patients who are allergic to betalactams, clindamycin is the drug of choice for oral infections.23
Other Aspects of Drug Administration
Other issues surrounding antibiotics commonly used to treat infection include treatment duration, bacterial resistance, and combination therapy. (Dosages of antibiotics commonly used to treat acute infections are found in Table 2.)5
Duration—Drug treatment duration has not been established for each type of infection, but drugs are generally administered for 7 to 10 days.5 With respect to azithromycin, it is only prescribed for 3 to 5 days. Bascone et al5 specified 500 mg/day for 3 days (Table 2); however, it is commonly prescribed as follows: 500 mg on day 1, followed by 250 mg daily for 4 days.
Bacterial resistance—The number of resistant microbes to antibiotics has doubled in the last 10 to 15 years.29 Accordingly, antibiotics should not be prescribed casually. It should be noted that higher doses of betalactams can provide better results than low doses; therefore, the maximum dose should be prescribed.30 In contrast, increased doses of macrolides (eg, erythromycin) do not provide better results against resistant bacteria.30
Combinations of antibiotics—Since there is an increased number of bacteria-resistant strains, combinations of antibiotics have been used that exert a synergistic effect against diverse pathogenic flora—eg, amoxicillin plus clavulanic acid, amoxicillin plus metronidazole, or in penicillin-allergic individuals, a combination of ciprofloxacin plus metronidazole.31 However, combinations of drugs should be reserved for severe or refractory infections.
Impact of Infected Tissue pH on Anesthetizing Technique
The pH-infected tissue affects the anesthetizing technique. A normal blood pH is about 7.4; a lower pH is considered acidic and a higher pH as alkaline. When an abscess is present, the pH of the tissues is reduced to pH 5.532 or lower. Therefore, when an anesthetic, which is a weak acid-base salt, is applied, it does not dissociate well, and there is less free base available for diffusing into the lipid-rich nerves. To compensate for this, the clinician should anesthetize around the infected site and consider giving regional blocks instead of infiltration injections.
Incision and Flap Design when Managing Oral infections
Two different approaches to incision and flap design when managing oral infections are addressed below: 1) simple incision and drainage to allow pus to escape; and 2) a flap approach to accomplish the same thing, but providing access for removal of an offending agent (eg, infected implant).
Incision and Drainage
An infected area that is is fluctuant should be lanced, with anatomic factors dictating the incision line. The incision can be made with a surgical blade and must be extended to the periosteum, because there may be pus accumulated under it. A periosteal elevator is placed into the incision line and under the periosteum to seek out pus deposits. A hemostat can also be inserted into the incision and opened around 5 mm to allow pus to escape. After the incision is made, the adjacent tissue is manipulated to drive out residual pus. An antibiotic is usually prescribed for 1 week.
If pus was evacuated, the surgeon places a drain (¼-inch-thick Iodoform gauze or a Penrose drain for patients not allergic to iodine) (Figure 5 and Figure 6). An inch is cut off and ¾ of an inch is placed under the tissue. To ensure the drain does not become misplaced, a suture can be positioned through the tissue and the drain. After 3 days, if there is no pus, the drain is removed. If the site is still purulent, the drain should be removed, the wound irrigated, and a new drain placed to maintain drainage. At this juncture, it may be desirable to switch antibiotics and perform more aggressive debridement. In addition, if the patient is not responding to therapy, it would be prudent to obtain a sample for culturing and antibiotic sensitivity testing, especially if this was not done initially when the site was drained.
If the preliminary problem was a periodontal or peri-implant abscess associated with a deep probing depth, it can be quickly managed. After local anesthesia is administered, an ultrasonic tip is inserted into the pocket to drain the abscess. This technique works well as a temporary measure if the clinician does not have time to surgically debride an infected site. Infections of endodontic origin require a pulpectomy and canal enlargement for sufficient drainage. When dealing with a tooth, the occlusion needs to be checked to determine if it needs to be relieved, because an infection often induces hyper-eruption of the tooth, leading to hypermobility and discomfort upon mastication. The patient is placed on antibiotics for 1 week.
Flap Approach
If a flap needs to be elevated to remove a tooth, implant, or granulomatous tissue, incision placement must take the following points into account: anatomical considerations; adequate size; broad base for the flap; and margins should be made on sound bone.33 In addition, to allow for sufficient drainage, it is best to avoid primary closure. As tissues that are infected become friable due to the increased number of inflammatory cells and decreased collagen, suturing infected tissue requires taking a large bite of tissue so the suture does not pull though the wound’s edge. Furthermore, when treating an infection in conjunction with a therapeutic intervention, especially when general symptoms of infection are manifested, recommendation should be made with respect to analgesics, hydration, nutritional support, and anti-inflammatory drugs.
Spread of Dental Infections: Deep Space Infections
Localized infections have the potential to spread via the blood system, lymphatic vessels, or tissue planes.23,34 They can spread through the blood system due to a bacteremia or an infected thrombus. Pathogens can travel in the lymph through the lymphatic vessels that connect a series of nodes. An involved lymph node will enlarge and be palpable. An inflammatory exudate can proceed along facial planes involving adjacent and distant sites (Table 3).7,34 The spread of the infection is facilitated by bacteria such as streptococci, which produce streptokinase and hyaluronidase. These enzymes destroy fascia around muscles and organs and turn potential spaces into real tissue compartments. Many of the deep spaces are adjacent to each other, and an infection can spread from one space to another.
In the maxilla, an infection can extend into the vestibular, buccal, canine, and infratemporal spaces and palatal areas (Table 3). In the mandible, an infection can also spread into the vestibular, submental, sublingual, submandibular, pterygomandibular, and parapharyngeal spaces (Table 3). Failure to promptly manage cellulitis can result in life-threatening sequelae: cavernous sinus thrombosis, septicemia, airway obstruction, and mediastinitis.7 Osteomyelitis and bone sequestration are also possible complications of untreated infections. Patients who are swollen and experience trismus and pain upon swallowing need aggressive therapy. Specific warning signs of an unresponding cellulitis include dysphagia, dyspnea, trismus, and progressive swelling.7 If there is an inadequate response to initial therapy, or the signs and symptoms of the infection are advanced, these patients should be referred to an oral surgeon and possibly be hospitalized.
Conclusion and Summary
A complete medical history facilitates safe treatment of medically compromised patients, and adherence to a strict aseptic protocol limits postsurgical orofacial infections. It has been suggested that a main reason for early dental implant loss is bacterial contamination at implant placement.35 Therefore, despite a lack of consensus on the use of prophylactic antibiotics prior to dental implant placement, it is the authors’ opinion that preoperative administration of antibiotics is recommended to ensure there is an effective blood level during surgical procedures. In this regard, data were cited to support the contention that this protocol (2 g, 1 hour before surgery) reduces the rate of implant failure.
With respect to complicated surgeries—eg, medically compromised patients, surgery performed in infected sites, extensive bone augmentation procedures, prolonged surgical interventions—it is up to the clinician’s judgment as to whether antibiotics should be prescribed pre- and postoperatively as a prophylactic measure.
If a postoperative infection occurs, early diagnosis and management usually preclude it from spreading. When there is fluctuance, incision and drainage typically provides immediate relief, and removal of an offending agent is critical to resolving an infection. This latter statement is critically important and needs to be underscored. Antibiotics useful for managing oral infections are amoxicillin/clavulanic acid, clindamycin, penicillin VK, amoxicillin, and metronidazole. Failure to achieve a good clinical result dictates frequent follow-up visits and altering the prescribed antibiotic, and surgical debridement may also be required. Improper antimicrobial dosage and lack of sufficient patient compliance should also be considered as possible reasons for lack of response by the patient. If the infection continues to worsen, referral to an oral maxillofacial surgeon or a hospital is necessary.
Disclosure
The authors had no disclosures to report.
About the Authors
Gary Greenstein, DDS, MS
Clinical Professor, College of Dental Medicine, Columbia University, New York, New York; Private Practice, Surgical Implantology and Periodontics, Freehold, New Jersey
Ben Greenstein, DMD
Clinical Assistant Professor, College of Dental Medicine, Rutgers University, Newark, New Jersey; Private Practice, Surgical Implantology and Periodontics, Freehold, New Jersey
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Evans AS. Epidemiological concepts. In: Evans AS, Brachmen PS, eds. Bacterial Infections of Humans: Epidemiology and Control. New York, NY: Plenum Press; 1991:3-58.
2. Greenstein G, Caton J, Polson AM. Histologic characteristics associated with bleeding after probing and visual signs of inflammation. J Periodontol. 1981;52(8):420-425.
3. Sans M, van Winkelhoff AJ. Periodontal infections: understanding the complexity—consensus of the Seventh European Workshop on Periodontology. J Clin Periodontol. 2011;38(suppl 11):3-6.
4. Armas J, Culshaw S, Savarrio L. Treatment of peri-implant diseases: a review of the literature and protocol proposal. Dent Update. 2013;40(6):472-474.
5. Bascones Martínez A, Aguirre Urízar JM, Bermejo Fenoll A, et al. Consensus statement on antimicrobial treatment of odontogenic bacterial infections [in Spanish]. Med Oral Patol Oral Cir Bucal. 2004;9:369-376.
6. Gutiérrez-Pérez JL, Perea-Pérez EJ, Romero-Ruiz MM, Girón-González JA. Orofacial infections of odontogenic origin. Med Oral. 2004;9(4):280-287.
7. Osborn TM, Assael LA, Bell RB. Deep space neck infection: principles of surgical management. Oral Maxillofac Surg Clin North Am. 2008;20(3):353-365.
8. de-Vicente-Rodríguez JC. Maxillofacial cellulitis [in Spanish]. Med Oral Patol Oral Cir Bucal. 2004;9 suppl:133-138.
9. Pack PD, Haber J. The incidence of clinical infection after periodontal surgery. A retrospective study. J Periodontol. 1983;54(7):441-443.
10. Checchi L, Trombelli L, Nonato M. Postoperative infections and tetracycline prophylaxis in periodontal surgery: a retrospective study. Quintessence Int. 1992;23(3):191-195.
11. Powell CA, Mealey BL, Deas DE, et al. Post-surgical infections: prevalence associated with various periodontal surgical procedures. J Periodontol. 2005;76(3):329-333.
12. Ahmad N, Saad N. Effects of antibiotics on dental implants: a review. J Clin Med Res. 2012;4(1):1-6.
13. Esposito M, Grusovin MG, Worthington HV. Interventions for replacing missing teeth: antibiotics at dental implant placement to prevent complications. Cochrane Database Syst Rev. 2013 Jul 31;7:CD004152.
14. Oomens MA, Forouzanfar T. Antibiotic prophylaxis in third molar surgery: a review. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114(6):e5-e12.
15. Gurenlian JR. Inflammation: the relationship between oral health and systemic disease. Dent Assist. 2009;78(2):8-10,12-14,38-40.
16. Kohl BA, Deutschman CS. The inflammatory response to surgery and trauma. Curr Opin Crit Care. 2006;12(4):325-332.
17. Deal DN, Tipton J, Rosencrance E, et al. Ice reduces edema. A study of microvascular permeability in rats. J Bone Joint Surg Am. 2002;84-A(9):1573-1578.
18. Nordqvist C. What Is Pus? Medical News Today Web site. Updated September 5, 2014. http://www.medicalnewstoday.com/articles/249182.php. Accessed December 8, 2014.
19. Howard K. What factors affect the color of pus? wiseGEEK Web site. http://www.wisegeek.com/what-factors-affect-the-color-of-pus.htm. Accessed December 8, 2014.
20. Heisler J. Types of drainage from a surgical wound. About.com Web site. Updated May 30, 2014. http://surgery.about.com/od/aftersurgery/a/Types-Of-Drainage-Exudate-From-A-Surgical-Wound.htm. Accessed December 8, 2014.
21. Balaji SM. General principles of oral surgery. In: Balaji SM, ed. Textbook of Oral and Maxillofacial Surgery. New Delhi, India: Elsevier; 2007:9-48.
22. George-Gay B, Parker K. Understanding the complete blood count with differential. J Perianesth Nurs. 2003;18(2):96-114.
23. Maestre-Vera JR. Treatment options in odontogenic infection [in Spanish]. Med Oral Patol Oral Cir Bucal. 2004;9(suppl):25-31.
24. Kuriyama T, Karasawa T, Nakagawa K, et al. Bacteriologic features and antimicrobial susceptibility in isolates from orofacial odontogenic infections. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90(5):600-608.
25. Legg JA, Wilson M. Prevalence of beta-lactamase producing bacteria in subgingival plaque and their sensitivity to Augmentin. Br J Oral Maxillofac Surg. 1990;28(3):180-184.
26. Kinder SA, Holt SC, Korman KS. Penicillin resistance in the subgingival microbiota associated with adult periodontitis. J Clin Microbiol. 1986;23(6):1127-1133.
27. Heimdahl A, Von Konow L, Nord CE. Betalactamase producing Bacteroides species in the oral cavity in relation to penicillin therapy. J Antimicrob Chemother. 1981;8(3):225-229.
28. Lacroix JM, Walker CB. Detection and incidence of the tetracycline resistance determinant tet(M) in the microflora associated with adult periodontitis. J Periodontol. 1995;66(2):102-108.
29. Herrera D, Van Winkelhoff AJ, Dellemijn-Kippuw N, et al. Beta-lactamase producing bacteria in the subgingival microflora of adult patients with periodontitis. A comparison between Spain and the Netherlands. J Clin Periodontol. 2000;27(7):520-525.
30. García Rey C, Martín Herrero JE, Prieto J. Relación entre consumo de antibióticos y selección de resistencias en el género Streptococcus. Medicina Preventiva. 2002;8(3):23-30.
31. Slots J, Jorgensen MG. Efficient antimicrobial treatment in periodontal maintenance care. J Am Dent Assoc. 2000;131(9):1293-1304.
32. Lardner A. The effects of extracellular pH on immune function. J Leukoc Biol. 2001;69(4):522-530.
33 Urolagin SB, Kale TP, Patil S. Intraoral incisions, design of flaps and management of soft tissue. Guident Web site. http://www.guident.net/oral-surgery/intraoral-incisions-design-of-flaps-and-management-of-soft-tissue.html. Accessed September 4, 2014.
34. Fehrenbach MJ, Herring SW. Spread of dental infections. Practical Hygiene. 1997;September-Ocotber:13-18.
35. Esposito M, Hirsch JM, Lekholm U, Thomsen P. Biological factors contributing to failures of osseointegrated oral implants. (II). Etiopathogenesis. Eur J Oral Sci. 1998;106(3):721-764.