You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
For centuries, physicians have been well aware that the oral cavity and oral tissues are a barometer of a person’s overall well being. More recently, numerous reports in the scientific literature have thrust further emphasis on the connection between oral disease and medical conditions. The US Surgeon General’s report in 2000 on oral health in America placed specific emphasis on the silent epidemic of oral disease, highlighting that oral infections can have a profound effect on total body health.1 Surgeon General Dr. David Satcher, author of that report, stressed that bacteria in the mouth can enter the systemic circulation and cause disease. In addition, he acknowledged that there are oral manifestations of systemic diseases, and that medications can have adverse oral effects. He noted that dental diseases can have a unique significance for immunocompromised patients and stressed the possible link between dental diseases and cardiovascular diseases and other medical illness.
There is, however, much confusion regarding the oral-systemic connection. Existing research has included mostly epidemiologic studies, some interventional studies, and fewer studies seeking to elucidate mechanisms of action. In many instances, study results have been contradictory. This is not surprising, however, given the variations in study design, the population studied, and the statistical analyses used. For practicing clinicians, it is important to be able to gather all of this emergent scientific evidence, synthesize it, and integrate it into their clinical practice. In essence, they must be able to authoritatively respond to their patients’ inquiries regarding relationships between oral and systemic disease.
Historical Background
Back in 1891, in a report by Dr. W.D. Miller titled, “The Human Mouth as a Focus of Infection,” the oral cavity became a focus of medicine as a potential source of all medical illnesses in what evolved as the focal infection theory.2 In 1931, Rosenow emphasized the importance of cooperation between dentists and physicians as well as the necessity of ensuring that the focus of oral infection was eliminated completely to improve medical health.3 However, by the mid-1930s, the validity of the focal infection theory began to be questioned when investigators found that when they considered the available real outcome data, there was no clear basis for ascribing the occurrence of much medical illness to the presence of oral foci of infection.4
It is clear that there is a large body of evidence suggesting association between oral and systemic disease. Yet, this consists mostly of epidemiologic and observational studies, which have inherent limitations that preclude establishing causal relationships. These epidemiologic studies include retrospective case control studies and prospective cohort studies, which cannot prove causality.5 They can, however, provide strong evidence for, and show the strength of, an association between a disease and putative causative factors. Randomized control trials, which can be used to test therapeutic and preventive measures, can provide presumptive evidence of disease causation in certain circumstances. However, these studies, too, have limitations in their design that can distort the results and need to be considered in evaluating the data. The principal question that remains is: “What is the association between oral disease and medical illness and causality?” Epidemiologic studies conducted to date suggest an association between periodontal disease and a number of medical illnesses. However, the strength and nature of these associations are not yet clear, because in some cases, the illness might result from confounding variables, including overlapping risk factors such as obesity, smoking, sedentary lifestyle, and lower socioeconomic status. Hill et al in a seminal paper in the Royal Society of Medicine in 1965 identified six areas that are necessary in evaluating the causal significance of observed associations.6 They include strength of association, consistency of findings among studies, temporality, biologic gradient, biologic plausibility, and experimental evidence. It is necessary to fulfill as many of these criteria as possible in the scientific data to establish causality. In order to effectively link oral disease as a cause of medical illness, these criteria need to be established.
Periodontal Disease and Pregnancy Complications
Emerging evidence is linking severe periodontal disease and pregnant women with an increased risk of delivering a preterm, low birth weight baby (PTLBWB).7-16 It is estimated that this risk is sevenfold; the National Institute of Dental and Craniofacial Research estimates that infectious oral disease may account for 18% of the 25 million PTLBWB born in the United States.17 According to the Institute of Medicine for Adverse Pregnancy Outcomes, current risk factors include a maternal age of less than 17 years old and greater than 34, African-American race, low socioeconomic status, inadequate prenatal care, drug use, alcohol and/or tobacco use, hypertension, genitourinary (GU) infections, diabetes, multiple pregnancies, and a history of PTLBW babies (Table 1).18
Currently, 12% of US pregnancies are complicated by preterm birth, meaning less than 37 weeks’ gestation. Low birth weight is categorized by less than 5 lbs. and 8 oz., and small gestational age refers to a baby in less than the 10% percentile. In addition, other complications such as miscarriage and preeclampsia may be related to periodontal disease.19 Generally, preterm birth results from three specific causes: 1) a preterm premature rupture of membranes (PPROM); 2) preterm labor from uterine contraction; or 3) a result of other complications, including induced labor due to preeclampsia.20 Preterm low birth weight babies in the United States have a staggering emotional, social, and economic cost. Hospital costs alone surpass $6 billion annually.21 In addition, there are significant risks to the mother, but primarily the risks are to the offspring, including neurodevelopmental problems, respiratory and behavioral problems, learning disabilities, and cardiovascular and metabolic problems. It is estimated that 25% of all complicated pregnancies occur without any known reason—in other words, they do not fulfill the currently known risk factors.19 This has prompted scientists and clinicians to research whether other causes might be at play.
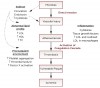
The potential relationship between pregnancy complications and periodontal disease is based on the assumption that the molecular pathogenesis would be similar to other maternal bacterial opportunistic infections such as GU infections. Currently, GU infections account for 30% to 50% of all cases of PTLBW babies.22 The mechanism proposed in these cases is that pathogens induce the release of inflammatory mediators, primarily Interleukin-1 (IL-1), Tumor Necrosis Factor-alpha (TNF-α), and Prostaglandin E2 (PGE2), which reach the placenta via the mother’s blood and trigger preterm labor. Periodontal bacteria is known to induce a local host immune response, including the release of inflammatory cytokines IL-1, TNF-α, and PGE2 and antibodies against the bacteria. Attempts by the maternal immune response and neutrophils to keep the infection localized, including a low maternal Immunoglobulin G (IgG) response to the bacteria, along with systemic access of the bacterial to the circulation represent the current hypothetical model (Figure 1). It is further postulated that the presence of bacteria in the blood trigger a secondary inflammatory response systemically by producing acute phase reactants such as C-reactive protein (CRP) from the liver. Bacteria, virulence factors, and cytokines all reach the placenta, creating a secondary site of bacterial challenge in a localized inflammatory response distant from the oral cavity. This fetal placental inflammation may cause placental tissue destruction, which can impair fetal growth and lead to low birth weight, and may affect maternal blood pressure, leading to preeclampsia. Also, cytokine production can lead to premature membrane eruption and uterine contraction, resulting in adverse pregnancy outcomes.23 Currently, cased controlled and cohort studies demonstrate an association in that both conditions exist in the same patients. In addition, cohort studies have shown temporality whereby periodontal disease precedes pregnancy complication and is not a consequence of it. However, many studies cannot fully exclude the possibility of other risk factors contributing to both conditions, thereby explaining this association.
Recent studies have tried to determine whether periodontal disease is a potentially reversible cause of adverse pregnancy outcomes. These studies divide women into two groups: those that receive periodontal therapy, and those that do not. One study by Mitchell-Lewis et al reported a 28% reduction in preterm low birth weights in the periodontal treatment group24; although, clinically significant, this was not a statistically significant difference from the nonperiodontal treatment group. Another study by Lopez et al indicated periodontal disease is an independent risk factor for preterm low birth weight,25 and a third study by Jeffcoat et al concluded that scaling and root planing may reduce preterm deliveries.26,27 In all three of these studies, periodontal treatment resulted in a reduction in the rate of preterm delivery and increased birth weight. However, the results were not statistically significant, and most women in the studies were African-American and/or of low socioeconomic status, which are characteristics associated with both diseases. Therefore, it may be possible that the data may not apply to the entire maternal population. Unfortunately, many questions remain, including: Can preventing or treating periodontal disease reduce the rate of pregnancy complications? Which pathogens induce adverse pregnancy outcomes or is a group of bacteria? After infection with periodontal pathogens, are the biologic events in animal studies true with humans? What is the best treatment for pregnant women with periodontal disease? When, in fact, should these women be treated?
Based on currently available scientific data, although many studies indicate a positive correlation, a causal relationship between periodontal disease and adverse pregnancy outcomes cannot be attributed yet. All studies suggest that periodontal treatment is safe for the mother and child; however, it is unclear if treatment improves pregnancy outcomes based on the available scientific evidence. Currently, multicenter randomized controlled trials are underway, which will provide the best evidence to support the idea that periodontal disease is a potential reversible cause of adverse pregnancy outcomes. It is currently recommended that oral healthcare providers inform patients of the biologic plausibility that untreated oral disease may affect pregnancy outcome and treat each patient according to the current standard of care.
Periodontal Infection and Cardiovascular Disease
Twenty years of research has firmly established that periodontal disease and cardiovascular disease are associated. However, two vital questions remain: What is the nature and relevance of this association? Does periodontal disease contribute causally to cardiovascular disease, or are they simply coincidentally associated? The answers to these questions have significant consequences, including whether or not dental health scores will ultimately be used to predict cardiovascular disease risks, and whether or not treatment of poor dental health reduces the risk of adverse cardiovascular outcomes. The central focus is the relationship of periodontal disease to atherosclerosis, a disease of the arteries in which fatty material and plaques are deposited in the wall of an artery, resulting in narrowing of the arterial lumen and eventual impairment of blood flow, which can lead to coronary artery disease, angina, heart attack, transient ischemic attacks and stroke, claudication, and organ damage.
Like low birth weight preterm deliveries, conventional risk factors for atherosclerosis account for only a portion of the atherosclerotic events in the general population. It is estimated that current known risk factors for atherosclerosis account for 50% to 70% of events.28 Other putative risk factors for atherosclerosis such as obesity, smoking, inflammation, and infection are shared with periodontal disease. Numerous traditional studies, including multiple cross-sectional studies, have demonstrated a higher incidence of atherosclerotic complications in patients with periodontal disease.29-31 These authors, however, drew careful conclusions because of overlapping risk factors for periodontal disease and cardiovascular disease, including older patients, male patients, cigarette smoking, diabetes, and low socioeconomic status. New research approaches have attempted to refine and test more specific hypotheses by directly measuring oral microbiology, by measuring systemic antibody titers, and by looking at the effect of intervention on subclinical markers. Desvarieux et al reported that carotid atherosclerosis, as measured by intima-media thickening increased with higher levels of periodontal bacterial after adjusting for traditional risk factors.32,33 Meyer et al noted that 95% of all examined coronary atheromas contained bacterial DNA.34 Of these, 49% were infected with at least one periodontal pathogen and 50% of those were associated with more than one periodontal pathogen. Pussinen and colleagues reported that antibodies to select periodontal pathogens, primarily Porphyromonas gingivalis (P. gingivalis), are associated with coronary heart disease.35-37 The implications of this study confirmed that edentulous status is, in fact, a marker for atherosclerotic coronary heart disease. In addition, the study confirmed that periodontal infection is associated with coronary disease and highlighted the specificity of antibodies to P. gingivalis for incident cardiovascular disease, suggesting that this bacterium is particularly deleterious for atherosclerotic complications. Intervention studies have attempted to assess the impact of periodontal treatment on subclinical markers of cardiovascular disease. Numerous studies have reported that periodontal treatment is associated with improved measures of systemic inflammation, including C-reactive protein.38-40 The studies, however, are not without flaw. In many instances, these interventions were not randomized and lacked untreated control groups with periodontal disease and the oral infection was not measured directly at baseline and follow-up to address the contributions of oral microbiology. In addition, many of these studies have small numbers of patients and failed to answer a vital question: Do subclinical findings translate into clinical events?
Mechanism by Which Periodontitis May Relate to Cardiovascular Disease
Currently, it is believed that the biological hypothesis for periodontal disease affecting or causing cardiovascular disease is via direct and indirect pathways (Figure 2). Oral microbes and their byproducts can gain systemic access to the circulatory system via the periodontium and oral cavity. These microbes have a potential to directly influence subclinical mediators of cardiovascular events, including atherosclerotic development and hypercoagulability. There is evidence that P. gingivalis can adhere to and infect cultures of coronary endothelial cells.41-43 Subsequent activation and expression of cell adhesion molecules may lead to endothelial dysfunction, which is an early event in the development of atherosclerosis. Atherosclerosis is mediated by an inflammatory process, and atheromas result from progressive accumulation of cholesterol and its esters in foam cells of the intima.44 Cytokines and atherosclerotic plaques regulate recruitment of other inflammatory cells, including vascular cell adhesion molecule (VCAM) and low-density lipoprotein (LDL). The release of cytokines from macrophages and foam cells is believed to be triggered by bacteria, leading to systemic activation of phagocytic cells. P. gingivalis is recognized as a key pathogen in this direct pathway process and a key player in periodontal disease. It has demonstrated a unique ability to invade epithelial cells, connective tissue cells, and endothelial cells. The invasion is mediated through up regulation of adhesion molecules, including intercellular adhesion molecules-1 (ICAM-1), VCAM, and E-selectins.44 Activation of these adhesion molecules is required to bind leukocytes to endothelium, which initiates the transmigration process and atherogenesis. Components of P. gingivalis increase uptake of LDL and foam cell macrophages. P. gingivalis has been shown to induce foam cell formation in human umbilical vein endothelial cells.45 Cholesterol accumulations and macrophages are, in fact, the hallmark of atherosclerotic lesions. In addition, oral bacterial play a role in platelet aggregation and thrombus-like formation in vitro. Matrix metalloproteinases (MMPs) identical to the ones involved in the periodontal attachment loss, derived from macrophages, promote atherosclerotic plaque rupture, and periodontal pathogens are known inducers of MMPs and could theoretically contribute to plaque rupture. In addition to these direct pathways, there is in an indirect effect. Atherosclerosis has a strong inflammatory component, and epidemiologic evidence suggests that increased levels of inflammation are, in fact, predictive of cardiovascular events. This includes C-reactive protein, which is found to be elevated in chronic periodontal disease.46,47 In addition, transient bacteremia and local release of bacterial byproducts, including lipopolysaccharides, trigger and enhance systemic inflammatory response. Combined with this, there exists molecular mimicry where antibodies towards bacteria cross-react with host cells (eg, heat shock protein 60).
Based on available scientific evidence, periodontal disease is clearly epidemiologically associated with cardiovascular disease. It is unclear if periodontal disease is a risk factor for, or contributes causally to, heart disease. Recent studies have enhanced specificity of infectious exposure definitions by measuring systemic antibodies to periodontal pathogens or quantifying oral microbota from dental plaque. However, several important risk factors are known to contribute to both conditions causally, making it extremely difficult to establish causality. Direct and indirect biological connections between periodontitis and atherosclerosis have been demonstrated at various stages of atherogenesis; however, based on available scientific evidence, currently recommending periodontal treatment solely for the prevention of atherosclerotic cardiovascular disease is not warranted. A recent systematic review of the level of evidence supporting periodontal disease as an independent risk factor for atherosclerotic vascular disease failed to demonstrate a causative relationship between the diseases. Furthermore, there is no evidence to support that periodontal interventions prevent atherosclerotic vascular disease or modify its outcomes.48 This review led to the American Heart Association’s scientific statement endorsed by the American Dental Association’s Council on Scientific Affairs and the World Heart Federation.48
Periodontal Disease and Diabetes
It is estimated that diabetes affects 20 million Americans, while 35% to 40% of Americans have not received a diagnosis.49 Currently, more than 9% of the US adult population has diabetes, and this number is expected to grow significantly in the future. The two main types of diabetes are classified primarily on the basis of their underlying pathophysiology. Type 1 diabetes, which constitutes about 5% to 10% of all cases in the United States, results from autoimmune destruction of insulin-producing beta cells in the pancreas, leading to total loss of insulin secretion. Insulin is used by the body to facilitate the transfer of glucose from the blood stream into target tissues such as muscle where glucose is used for energy. Since a person with Type 1 diabetes no longer produces endogenous insulin, glucose is unable to enter target cells and remains in the blood stream, resulting in sustained hyperglycemia. A patient with Type 1 diabetes, therefore, must take exogenous insulin to remain alive, hence, the former name “insulin-dependent diabetes.”
Type 2 diabetes constitutes about 85% to 90% of all diabetic cases and results from insulin resistance rather than from a total absence of insulin production. Autoimmune destruction of beta cells does not occur in these patients, and they do retain the capacity to secrete some insulin; although, production often diminishes over time. Type 2 diabetes patients remain undiagnosed for many years because hyperglycemia appears gradually and often without symptoms. Insulin resistance results in a decreased capacity to transfer glucose into target cells, resulting in hyperglycemia.
The relationship between diabetes and periodontal disease has been the subject of more than 200 articles published in English over the past 50 years.50-58 Interpretation of this research is made difficult by the numerous classifications for diabetes and periodontitis used over the years. Varying clinical and radiographic criteria used to assess periodontal disease and the extent and severity of the disease also make it challenging to establish relationships. In addition, evolving standards for the degree of glycemic control and changing methodology for assessing the complications associated with diabetes have made interpretation of the research difficult at best. Most of the research has focused on assorted populations and often has included relatively few subjects or has lacked controls. Evidence suggesting diabetes as a risk factor for gingivitis and periodontitis dates back to the 1950s. Increased gingival inflammation is seen in children with Type 1 diabetes, as is the greatest severity of gingivitis in patients with very poor glycemic control. Evidence suggests that diabetes mellitus increases the risk of developing periodontitis.59 Lalla has documented increased prevalence and severity of attachment loss in adults and children with diabetes, and there is increased loss with poor glycemic control.60 There appears, however, to be significant individual variability in the degree to which glycemic control influences periodontal status. How diabetes affects gum disease and how gum disease affects the metabolic state seem to be evolving narratives. Intervention trials have resulted in varied metabolic responses in patients with diabetes mellitus. A meta-analysis of 10 intervention trials (> 450 patients) found an average decrease in absolute hemoglobin HbA1c values of 0.4% after scaling and root planing.61 The addition of systemic antibiotic therapy resulted in mean absolute reduction of HbA1c of 0.7%; although, not statistically significant, clinical significance in the practice of medicine is defined by a change of 0.6%. The mechanism of interaction between diabetes and periodontal disease focuses on the function of cells, including altered neutrophil, monocyte, and macrophage function. In addition, there is up regulation of other immunoregulatory responses, including IL-1b and TNF-α.62 The altered wound healing that is seen in diabetes relates to poor fibroblast function in a high glucose environment. In addition, there is elevation of MMPs. In diabetes, changes in microvascular integrity lead to end-organ damage, which includes the presence of advanced glycosylated end-products in the periodontium.63,64 Inflammation is a common theme in both Type 1 and Type 2 diabetes, as is periodontal disease. Inflammation is a known cause of insulin resistance. Chronic periodontal disease can, therefore, increase insulin resistance and worsen glycemic control. This can happen secondarily as periodontal disease increases pro-inflammatory cytokines, which can lead to insulin resistance. Currently, commercially available salivary diagnostic tests attempt to identify the type and concentration of specific pathogenic bacteria known to cause periodontal disease. In addition, other tests claim to identify individual genetic susceptibility to periodontal disease to enable the clinician to establish which patients are at increased risk for more severe periodontal infection due to an exaggerated immune response. Those recommended to be tested include patients who have a family history of diabetes.
Conclusion
Although there is no conclusive evidence that treating oral disease will prevent heart attack, stroke, diabetes, or adverse pregnancy outcomes, dental professionals should promote a message that a strong biologic connection exists between oral and systemic health. Emergence of this topic and new data is leading to a convergence in oral and medical care that clearly benefits public health.
References
1. US Department of Health and Human Services. Oral Health in America: A Report of the Surgeon General. Rockville, MD: US Department of Health and Human Services, National Institute of Dental and Craniofacial Research, National Institutes of Health; 2000.
2. Miller WD. The human mouth as a focus of infection . Dental Cosmos. 1891;33(9):689-706.
3. Rosenow EC. Studies of elective localization: focal infection with special reference to oral sepsis. J Dent Res. 1919;1(3):205-267.
4. Pallasch TJ, Wahl MJ. The focal infection theory: appraisal and reappraisal. J Calif Dent Assoc. 2000;28(3):194-200.
5. Taubes G. Epidemiology faces its limits. Science. 1995;269(5221):164-169.
6. Hill AB. The environment and disease: association or causation? Proc R Soc Med. 1965;58:295-300.
7. Offenbacher S, Katz V, Fertik G, et al. Periodontal infections as a possible risk factor for preterm low birth weight. J Periodontol. 1996;67(10 suppl):1103-1113.
8. Dasanayake AP, Boyd D, Madianos PN, et al. The association between Porphyromonas gingivalis-specific maternal serum IgG and low birth weight. J Periodontol. 2001;72(11):1491-1497.
9. Dasanayake AP. Poor periodontal health of the pregnant woman as a risk factor for low birth weight. Ann Periodontol. 1998;3(1):206-212.
10. Moore S, Ide M, Coward PY, et al. A prospective study to investigate the relationship between periodontal disease and adverse pregnancy outcome. Br Dent J. 2004;197(5):251-258.
11. Lunardelli AN, Peres MA. Is there an association between periodontal disease, prematurity and low birth weight? A population-based study. J Clin Periodontol. 2005;32(9):938-946.
12. Moreu G, Tellez L, Gonzalez-Jaranay M. Relationship between maternal periodontal disease and low-birth-weight pre-term infants. J Clin Periodontol. 2005;32(6):622-627.
13. Moore S, Randhawa M, Ide M. A case-control study to investigate an association between adverse pregnancy outcome and periodontal disease. J Clin Periodontol. 2005;32(1):1-5.
14. Davenport ES, Williams CE, Sterne JA, et al. Maternal periodontal disease and preterm low birthweight: case-control study. J Dent Res. 2002;81(5):313-318.
15. Carta G, Persia G, Falciglia K, Iovenitti P. Periodontal disease and poor obstetrical outcome. Clin Exp Obstet Gynecol. 2004;31(1):47-49.
16. Jeffcoat MK, Hauth JC, Geurs NC, et al. Periodontal disease and preterm birth: results of a pilot intervention study. J Periodontol. 2003;74(8):1214-1218.
17. NIDCR: National Institute of Dental and Craniofacial Research web site. http://www.nidcr.nih.gov/. Accessed January 12, 2012.
18. Kramer MS. The epidemiology of adverse pregnancy outcomes: an overview. J Nutr. 2003;133(5 suppl 2):1592S-1596S.
19. Premature Birth. CDC Centers for Disease Control and Prevention web site. http://www.cdc.gov/features/prematurebirth/. Accessed April 11, 2012.
20. Goldenberg RL, Jennifer CF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371(9606):75-84.
21. Russell RB, Green NS, Steiner CA, et al. Cost of hospitalization for preterm and low birth weight infants in the United States. Pediatrics. 2007;120(1):e1-e9.
22. Bánhidy F, Acs N, Puhó EH, Czeizel AE. Pregnancy complications and birth outcomes of pregnant women with urinary tract infections and related drug treatments. Scand J Infect Dis. 2007;39(5):390-397.
23. Shub A, Swain JR, Newnham JP. Periodontal disease and adverse pregnancy outcomes. J Matern Fetal Neonatal Med. 2006;19(9):521-528.
24. Mitchell-Lewis D, Engebretson SP, Chen J, et al. Periodontal infections and pre-term birth: early findings from a cohort of young minority women in New York. Eur J Oral Sci. 2001;109(1):34-39.
25. Lopez NJ, Smith PC, Gutierrez J. Higher risk of preterm birth and low birth weight in women with periodontal disease. J Dent Res. 2002;81(1):58-63.
26. Jeffcoat MK, Geurs NC, Reddy MS, et al. Periodontal infection and preterm birth: results of a prospective study. J Am Dent Assoc. 2001;132(7):875-880.
27. Jeffcoat MK, Hauth JC, Geurs NC, et al. Periodontal disease and preterm birth: results of a pilot intervention study. J Periodontol. 2003;74(8):1214-1218.
28. Nieto FJ. Infections and atherosclerosis: new clues from an old hypothesis? Am J Epidemiol. 1998;148(10):937-948.
29. Grau AJ, Becher H, Ziegler CM, et al. Periodontal disease as a risk factor for ischemic stroke. Stroke. 2004;35(2):496-501.
30. Mattila KJ, Valtonen VV, Nieminen M, Huttunen JK. Dental infection and the risk of new coronary events: prospective study of patients with documented coronary artery disease. Clin Infect Dis. 1995;20(3):588-592.
31. Syrjanen J, Peltola J, Valtonen V, et al. Dental infections in association with cerebral infarction in young and middle-aged men. J Intern Med. 1989;225(3):179-184.
32. Desvarieux M, Demmer RT, Rundek T, et al. Relationship between periodontal disease, tooth loss, and carotid artery plaque: the Oral Infections and Vascular Disease Epidemiology Study (INVEST) . Stroke. 2003;34(9):2120-2125.
33. Desvarieux M, Schwahn C, Volzke H, et al. Gender differences in the relationship between periodontal disease, tooth loss, and atherosclerosis. Stroke. 2004;35(9):2029-2035.
34. Meyer MW, Gong K, Herzberg MC. Streptococcus sanguis-induced platelet clotting in rabbits and hemodynamic and cardiopulmonary consequences. Infect Immun. 1998;66(12):5906-5914.
35. Pussinen PJ, Alfthan G, Tuomilehto J, et al. High serum antibody levels to Porphyromonas gingivalis predict myocardial infarction. Eur J Cardiovasc Prev Rehabil. 2004;11(5):408-411.
36. Pussinen PJ, Nyyssonen K, Alfthan G, et al. Serum antibody levels to Actinobacillus actinomycetemcomitans predict the risk for coronary heart disease. Arterioscler Thromb Vasc Biol. 2005;25(4):833-838.
37. Pussinen PJ, Alfthan G, Rissanen H, et al. Antibodies to periodontal pathogens and stroke risk. Stroke. 2004;35(9):2020-2023.
38. Elter JR, Hinderliter AL, Offenbacher S, et al. The effects of periodontal therapy on vascular endothelial function: a pilot trial. Am Heart J. 2006;151(1):47.
39. Mercanoglu F, Oflaz H, Oz O, et al. Endothelial dysfunction in patients with chronic periodontitis and its improvement after initial periodontal therapy. J Periodontol. 2004;75(12):1694-1700.
40. Seinost G, Wimmer G, Skerget M, et al. Periodontal treatment improves endothelial dysfunction in patients with severe periodontitis. Am Heart J. 2005;149(6):1050-1054.
41. Desvarieux M, Demmer RT, Rundek T, et al. Periodontal microbiota and carotid intima-media thickness: the Oral Infections and Vascular Disease Epidemiology Study (INVEST) . Circulation. 2005;111(5):576-582.
42. Spahr A, Klein E, Khuseyinova N, et al. Periodontal infections and coronary heart disease: role of periodontal bacteria and importance of total pathogen burden in the Coronary Event and Periodontal Disease (CORODONT) study . Arch Intern Med. 2006;166(5):554-559.
43. Lalla E, Lamster IB, Hofmann MA, et al. Oral infection with a periodontal pathogen accelerates early atherosclerosis in apolipoprotein E-null mice. Arterioscler Thromb Vasc Biol. 2003;23(8):1405-1411.
44. Beck JD, Eke P, Lin D, et al. Associations between IgG antibody to oral organisms and carotid intima-medial thickness in community-dwelling adults. Atherosclerosis. 2005;183(2):342-348.
45. Kuramitsu HK, Kang IC, Qi M. Interactions of Porphyromonas gingivalis with host cells: implications for cardiovascular diseases. J Periodontol. 2003;74(1):85-89.
46. Loos BG, Craandijk J, Hoek FJ, et al. Elevation of systemic markers related to cardiovascular diseases in the peripheral blood of periodontitis patients. J Periodontol. 2000;71(10):1528-1534.
47. D’Aiuto F, Parkar M, Andreou G, et al. Periodontitis and systemic inflammation: control of the local infection is associated with a reduction in serum inflammatory markers. J Dent Res. 2004;83(2):156-160.
48. Lockhart PB, Bolger AF, Papapanou PN, et al; on behalf of the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, Council on Epidemiology and Prevention, Council on Peripheral Vascular Disease, and Council on Clinical Cardiology. Periodontal Disease and Atherosclerotic Vascular Disease: Does the Evidence Support an Independent Association? A Scientific Statement From the American Heart Association [published online ahead of print April 18, 2012]. Circulation. doi:10.1161/CIR.0b013e31825719f3.
49. National Center for Health Statistics. CDC Centers for Disease Control and Prevention web site. http://www.cdc.gov/nchs/. Accessed January 12, 2012.
50. Ervasti L, Knuuttila M, Pohjamo L, Haukipuro K. Relation between control of diabetes and gingival bleeding. J Periodontol. 1985;56(3):154-157.
51. Cutler CW, Machen RL, Jotwani R, Iacopino AM. Heightened gingival inflammation and attachment loss in type 2 diabetics with hyperlipidemia. J Periodontol. 1999;70(11):1313-1321.
52. Karjalainen K, Knuuttila M. The onset of diabetes and poor metabolic control increases gingival bleeding in children and adolescents with insulin-dependent diabetes mellitus. J Clin Periodontol. 1996;23(12):1060-1067.
53. Salvi GE, Kandylaki M, Troendle A, et al. Experimental gingivitis in type 1 diabetics: a controlled clinical and microbiological study. J Clin Periodontol. 2005;32(3):310-316.
54. Promsudthi A, Pimapansri S, Deerochanawong C, Kanchanavasita W. The effect of periodontal therapy on uncontrolled type 2 diabetes mellitus in older subjects . Oral Dis. 2005;11(5):293-298.
55. Stewart JE, Wager KA, Friedlander AH, Zadeh HH. The effect of periodontal treatment on glycemic control in patients with type 2 diabetes mellitus. J Clin Periodontol. 2001;28(4):306-310.
56. Janket SJ, Wightman A, Baird AE, et al. Does periodontal treatment improve glycemic control in diabetic patients? A meta-analysis of intervention studies. J Dent Res. 2005;84(12):1154-1159.
57. Engebretson SP, Hey-Hadavi J, Ehrhardt FJ, et al. Gingival crevicular fluid levels of interleukin-1β and glycemic control in patients with chronic periodontitis and type 2 diabetes. J Periodontol. 2004;75(9):1203-1208.
58. Kiran M, Arpak N, Unsal E, Erdogan MF. The effect of improved periodontal health on metabolic control in type 2 diabetes mellitus. J Clin Periodontol. 2005;32(3):266-272.
59. Cianciola LJ, Park BH, Bruck E, et al. Prevalence of periodontal disease in insulin-dependent diabetes mellitus (juvenile diabetes). J Am Dent Assoc. 1982;104(5):653-660.
60. Lalla E, Cheng B, Lal S, et al. Periodontal changes in children and adolescents with diabetes: a case-control study. Diabetes Care. 2006;29(2):295-299.
61. Mealey BL. Periodontal disease and diabetes: a two-way street. J Am Dent Assoc. 2006;137(suppl):26s-31s.
62. Mealey BL, Moritz AJ. Hormonal influences: effects of diabetes mellitus and endogenous female sex steroid hormones on the periodontium. Periodontol 2000. 2003;32:59-81.
63. Schmidt AM, Weidman E, Lalla E, et al. Advanced glycation end-products (AGEs) induce oxidant stress in the gingiva: a potential mechanism underlying accelerated periodontal disease associated with diabetes . J Periodontal Res. 1996;31(7):508-515.
64. Katz J, Bhattacharyya I, Farkhondeh-Kish F, et al. Expression of the receptor of advanced glycation end products in gingival tissues of type 2 diabetes patients with chronic periodontal disease: a study utilizing immunohistochemistry and RT-PCR. J Clin Periodontol. 2005;32(1):40-44.
Related content: For more information, read Education and Attitudes about the Association between Periodontal Disease and Diabetes at dentalaegis.com/go/cced153
About the Authors
Scott S. DeRossi, DMD
Chairman
Department of Oral Health and Diagnostic Sciences
Director
Clinical Center for Oral Medicine
Associate Professor of Oral Medicine
Associate Professor of Otolaryngology/Head Neck Surgery
Associate Professor of Dermatology
Georgia Health Sciences University
Augusta, Georgia
Thomas P. Sollecito, DMD, FDS, RCS(Ed)
Professor and Chairman of Oral Medicine
Department of Oral Medicine
School of Dental Medicine
University of Pennsylvania
Professor of Oral Medicine in Otolaryngology Head & Neck Surgery
Department of Otolaryngology
Perelman School of Medicine
University of Pennsylvania
Philadelphia, Pennsylvania