You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
A 72-year-old edentulous Caucasian woman who presented for a denture adjustment complained of painful skin and intraoral mucosal blisters, which were causing a crippling deterioration in daily functioning. Both her maxillary and mandibular dentures had been fabricated at a local dental center about a year earlier. She just wanted her dentures adjusted and an opinion as to whether they were causing her skin blisters.
The patient was a married retired mother of one. Though well-educated and intelligent, she expressed an intense aversion to going to doctors and had not been seen by a physician for several years, despite borderline hypertension. She was overweight at 175 pounds and 5’3” and had been a one-pack-a-day smoker for more than 40 years. Her past medical history indicated three unsuccessful surgeries to treat pelvic organ prolapse with a polypropylene surgical mesh. Her vital signs were normal (BP 137/88) and she had no known allergies. She reported regular use of multivitamin supplements and occasionally naproxen for headaches and arthritic joint pain. A history of general anxiety and insomnia was noteworthy.
Dental Exam
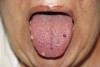
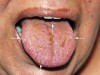
Intraoral examination revealed mucosal peeling, scars, and several eroding ulcers on the tongue, lip, palate, and bilaterally along both load-bearing and nonload-bearing surfaces of the alveolar ridges. Hemorrhagic blisters and petechiae were also present on the dorsum of the tongue and posterior pharyngeal wall, compounded with the additional complication of candidiasis (Figure 1 and Figure 2). Burning, itching, and redness, as well as spontaneous peeling and bleeding of the buccal mucosa made eating uncomfortable.
Mucosal fragility required gentle manipulation of the dentures since any abrupt movement could potentially cause additional blistering. Both dentures, however, appeared to be clinically acceptable and noncontributory to intraoral pathology. Denture retention was adequate. Flange extensions and vertical dimension were within normal limits. Occlusion was lingualized and balanced. Close examination of the intaglio surface of the dentures likewise presented no conspicuous burs or rough spots that would account for intraoral blisters. There was no indication that the dentures were ill-fitting or in need of relining due to bone resorption. Without evidence of trauma, allergy to acrylic, or other dental etiology, the chief concern was systemic pathology.
History of Present Illness
Inflamed blisters, scars, and milia covered much of the skin of her hands, fingers, elbows, knees, and ankles (Figure 3). She said these areas were especially trauma-prone. If she bumped them, the skin could peel away and bleed. She experienced periodic remissions and exacerbations, and said that some lesions would heal, while others left scars. She also suffered from nail dystrophy, hair loss, a loss of appetite, and said that eating could cause esophageal rupturing and bowel irritation. Lesions could break out in the ocular mucosal membranes as well by simply rubbing her eyes.
One blister on the extensor surfaces of her digits was hemorrhagic and purulent. The others were fluid-filled. All these changes, she stated, had a rapid onset and presented within a period of about 2 months during the summer and progressively got worse over time. She said that the summer heat and heat in general seemed to make it worse. She avoided showers since the force of the water could unpredictably open up wounds on her torso and buttocks. Pointing to the tiny white bumps or pimples or milia dispersed along the extensor surfaces of her finger joints, she started crying and stated that she looked like “a leper.” When asked why she hadn’t been examined by a doctor, she said she was afraid that it was cancer and if it was, she didn’t want to know. The patient was mollified with a few reassuring words and persuaded to seek immediate and appropriate medical attention.
Differential Diagnosis
Differential diagnosis included bullous systemic lupus erythematosus, mucous-membrane pemphigoid, linear IgA disease, paraneoplastic pemphigus, and other related skin disorders. The patient was referred to a dermatologist who, subsequently, ordered comprehensive testing.
Dental Treatment
Due to the seriousness and complexity of this case, referral to an ear, nose, and throat (ENT); ophthalmology; gastrointestinal (GI); or dermatology specialist was not optional. It was imperative to refer the patient to an appropriate specialist to obtain an accurate diagnosis and formulate an effective treatment plan. Initial palliative treatment, however, included the following: The patient was instructed to eat just soft foods and avoid hard or brittle foods and foods with high acid content such as tomatoes and orange juice. She was advised to stop smoking, avoid trauma, and cover the extensor skin surfaces with protective pads, at least until she could receive appropriate medical attention. Gentle but thorough daily oral hygiene was also encouraged until an appropriate treatment plan was determined. The primary focus was diagnosis, prevention of new lesions, and to guard against infection. Mycelex troches were prescribed for candidiasis.
A temporary soft denture reline was applied to cushion compressive forces. She was instructed, however, to keep the dentures out of her mouth whenever possible. The dentures were relined with an ethyl alcohol-free soft reline tissue conditioner based on an n-butyl methacrylate/i-butyl methacrylate copolymer. Conventional soft-tissue liners containing ethyl alcohol that could potentially leach out and cause further mucosal irritation were avoided. Triamcinolone acetonide (0.1%) dental paste was prescribed to treat mucosal erosions, as well as to evaluate the effectiveness of an intermediate potency corticosteroid to decrease the severity of symptoms.
The patient returned for follow-up 1 week later. Candidiasis had resolved. There was nominal improvement of mucosal redness, itching, and pain, but blisters were still present and cause for concern. After consulting the specialist, the patient was prescribed dexamethasone elixir 0.5 mg/ml and told to rinse with 1 tsp. for 2 to 3 minutes and spit. Concurrent maintenance of oral hygiene was also important to reduce the severity of symptoms, and the patient was instructed to avoid abrasive dentifrices and alcohol-containing mouthwashes that could potentially induce exacerbation. The rationale for treatment with dexamethasone rinse was to decrease oral discomfort due to symptomatic lesions, to improve function (eating, speaking, sleeping, wearing dental prostheses, etc), as well as to prevent the frequency and severity of exacerbations. Dexamethasone is a more potent anti-inflammatory agent than triamcinolone acetonide and indicated for local relief of pain in multiple ulcerated areas. Each teaspoonful (5 ml) of the rinse contains 0.5 mg of dexamethasone, 0.1% benzoic acid, and 5.1% alcohol as a preservative. The inactive ingredients consist primarily of purified water, propylene glycol, and liquid sugar. Thus, the alcohol content is unlikely to induce exacerbations. It should be noted, however, that dexamethasone is contraindicated whenever there is candidiasis anywhere in the body. Since candidiasis in this case had resolved before prescribing the rinse, there was no contraindication to dexamethasone. Nevertheless, the patient needed to be closely monitored for recurrence of candidiasis.
The patient was asked to check in at 10-day intervals, and a compound intraoral lidocaine rinse (a three-drug mouthwash: lidocaine, diphenhydramine, and sodium bicarbonate in normal saline) was also prescribed to palliate oral burning. At each visit, the patient was reminded of the gravity of her situation and the importance of compliance with the diagnostic appointments and subsequent recommendations made by the dermatologist. In order to achieve a positive outcome, the complexity of this case required collaboration with an experienced immunomodulating prescribing clinician, which was outside the normal scope of the dentist.
It is important to note, however, that patient compliance in following through with the referral process should not be assumed. In this case, compassionate bedside manner proved to be as important as palliation in convincing the elderly woman to seek appropriate medical evaluation. This included making several phone calls to simply ask the patient how she was feeling, how the evaluation process was going, and if there was anything the clinician could do to encourage the patient to remain in compliance. It is important that the patient understand and trust that the clinician and specialist are there to help her through the problem.
Medical Assessment
The patient was evaluated at a university hospital several days later, and the following tests were performed: histopathology from the edge of selected blisters; direct immunofluorescence on normal-appearing perilesional skin; and indirect immunofluorescence with the patient’s serum on salt-split normal human skin substrate. Histopathology documented the separation between the epidermis and the dermis, confirming the presence of subepidermal blisters, and revealing a mixed inflammatory cell dermal infiltrate. Direct immunofluorescence of a skin biopsy detected a thick band of IgG and C3 deposited at the epidermal basement membrane zone. Indirect immunofluorescence detected the presence of IgG circulating autoantibodies in the patient’s serum that targeted the skin basement membrane component, Type VII collagen, and was bound to the dermal floor on 1 mol/L salt-split normal human skin substrate. Immunoblot analysis of dermal extracts demonstrated IgG antibodies against Type VII collagen, whereas no reaction was seen with epidermal extracts or by enzyme-linked immunosorbent assay using a recombinant form of bullous pemphigoid 180. About 6 weeks later, a diagnosis of the generalized inflammatory variant of the rare autoimmune skin disease epidermolysis bullosa acquisita (EBA) was confirmed.
Medical Treatment
EBA is difficult to treat. Systemic corticosteroids are the gold standard in treatment, but there is no proof that they are the best treatment.1 The primary objective is to protect the skin and stop blister formation, promote healing, and prevent complications. Immunosuppressive agents can be used to modify or reduce autoimmune responses and decrease the production of autoantibodies. They may, however, increase the risk of secondary infection. These drugs include: azathioprine, dapsone, colchicine, cyclophosphamide, corticosteroids, mycophenolate mofetil, gold, and intravenous immunoglobulin. None of these immunomodulatory agents, however, are target-specific in blocking the autoimmune response.2-6
Various adjuvants and therapeutic modalities include cyclosporine, colchicine, plasmapheresis, extracorporeal photochemotherapy, and intravenous gammaglobulins.4,5,7-9 For patients on long-term systemic corticosteroid treatment, daily calcium and vitamin D supplements are essential to reduce steroid-induced osteoporosis. They should also take bisphosphonate to inhibit osteoclast-mediated bone resorption.4,6,8,9
Patients with EBA—especially the elderly—need to be monitored regularly during treatment. Since treatment requires long-term medications, potentially severe adverse effects—including internal malignancies—are a concern.2-6 Coordination of the patient’s care and good communication between the patient’s primary physician and all other healthcare professionals involved in treatment are important for the overall management of the patient.10
Patient education is also important. Patients should not delay reporting any change to their health status and must avoid direct physical trauma to their skin surfaces. They can wear protective pads to cover their extensor skin surfaces, and excellent skin care will help prevent infection.4,5,8,9 Skin grafting for denuded or ulcerated areas of the skin may be necessary. Other surgical procedures include dilation of the esophagus if there is a stricture, repair of hand deformities, and removal of any malignant lesions that may develop. Working with a physical therapist can help keep the full range of motion in the joints and minimize contractures.4,5
Dental Management
EBA often results in significant oral scarring and is a major complication that directly impacts dentists from the standpoints of being able to access the oral cavity for care and having a role in preventing additional scarring. Manipulation of structures in the oral cavity must, therefore, be conducted cautiously in order to minimize trauma. Good dental hygiene is very important, including regular dental visits, since patients are highly likely to develop dental caries11 and are susceptible to infection. Proper nutrition is also important to help recovery. Patients should avoid eating hard or brittle foods, as well as foods with high acid content. If swallowing difficulties are present, oral steroids for short periods of time may be prescribed. It is suggested, however, that the dentist refer the patient to an ENT or GI specialist to evaluate dysphagia rather than empirically prescribe steroids without consulting a specialist. If steroids are used for longer than 1 month, calcium and vitamin D supplements may be necessary to prevent osteoporosis. If there is candida in the mouth or esophagus, medication would be prescribed as well.4,5 Periodontal surgery, including gingivectomy, has been performed in patients with EBA,11 but its general usefulness in maintaining gingival health in patients with EBA has not been confirmed.
There is no standard regimen to treat EBA and no single recommended therapy. Treatment by the dentist should be individually tailored and aimed at reducing inflammation. Close collaboration with the patient’s physician is imperative, since the combination of steroids prescribed by the dentist together with immunomodulating drugs prescribed by the physician can cause side effects. Referral to a specialist in oral medicine may be necessary.
Prolonged use (longer than 2 weeks) of topical steroids may result in secondary candidiasis and mucosal atrophy (though rarely),12 and may increase the potential of systemic absorption. Prescribing antifungal therapy together with the topical steroids may be necessary. Due to alternating symptomatic exacerbations and remissions, a phased sequence of therapy with a more potent regimen of topical corticosteroid for 1 to 2 weeks followed by a second phase regimen of less potent topical corticosteroids will often provide effective therapy. During periods of acute exacerbation, combinations of more than one topical corticosteroid may also be effective—for example, first a dexamethasone oral rinse followed by application of fluocinonide or triamcinolone ointment. There are many combinations that may be utilized, depending upon the individual clinical scenario. Topical steroids of various potencies that may be helpful in treating the patient and as an adjunct to systemic therapy are presented in Table 1. Several prescription options based on case severity that the clinician may find helpful are presented in Table 2.
A “magic mouthwash” containing benadryl, kaopectate (or carafate), and milk of magnesia as a base to which nystatin and/or lidocaine may be added (depending upon the clinical indications) can be very effective as a therapy for mildly symptomatic cases or as maintenance therapy. Pharmacies can customize each prescription formulation specifically to the patient’s individual requirements. The previously mentioned three-drug mouthwash (lidocaine, diphenhydramine, and sodium bicarbonate in normal saline) can provide effective symptomatic relief in patients with mucositis. Compound dental washes or rinses offer numerous advantages over commercially available dosage forms. Elixirs, syrups, and suspensions often contain preservatives such as alcohol, which can cause mucosal reactions or gastrointestinal irritation, or sugar, which makes the preparation undesirable for prolonged use in the mouth or for diabetic patients. A customized preparation without unnecessary excipients can eliminate these concerns. Various anesthetic/analgesic and antibiotic/anti-infective mouthwashes are also available.
Prognosis
There is no cure for EBA. The prognosis is generally good but depends on disease severity. Mild forms improve with time. If treated properly, patients can expect to live a normal life span.4,5
Long-term remission in EBA patients, however, is difficult to achieve. In patients who are resistant or develop side effects to conventional immunosuppressive therapy, a prolonged clinical remission is highly unlikely. Prolonged remissions have successfully been induced in some patients treated with regimens that include dapsone, colchicine, mesalazine, cyclosporine, mycophenolic acid, intravenous immunoglobulin, rituximab, daclizumab, extracorporeal photochemotherapy, and plasmapheresis; but the long-term benefits are inconclusive and require additional study.13
Discussion
The patient returned 6 weeks after the initial consult to coordinate future dental treatment. Medical treatment had already started and the patient was put on cyclosporine (6 mg/kg per day) and prednisone (5mg/day). At 3-months follow-up, no side-effects were observed. There was some improvement in her condition, but the results were transient and not lasting. The patient still complained of mucosal “itching” and redness, but blisters on her hands, elbows, and ankles were reduced.
Both dentures were relined with a permanent resilient reliner made of a rubber compound, polyphosphazene, which does not harden like plasticized acrylics or require surface coatings to restrict the leach out of toxic plasticizers. It resists fungus overgrowth and obviates the need for periodic antimicrobial treatments, in comparison to silicone and urethane materials.14,15 The patient was examined at 3 monthly follow-ups and did not present with new intraoral blisters at those visits.
At 1-year follow-up, the patient was free of candida and, except for residual pretreatment scarring, there was no evidence of new lesions on the load-bearing surfaces of the alveolar ridges. There were two small clusters of petechiae on the anterior dorsum of the tongue and posterior pharyngeal wall, but no dysphagia, eroding ulcers, or blisters. Mucosal itching and pain were diminished, but redness of the buccal mucosa persisted. Continued smoking was a contributing factor. Despite this, her attitude towards treatment had greatly improved and was now positive. The patient was attempting to cut back on smoking and was receptive to making behavioral and nutritional life changes necessary for a successful outcome.
Though it cannot be stated that the patient was cured or the disease was in complete remission, the patient was doing comparatively well. The overall combination of these life changes, together with protective pads to shield external areas from trauma, as well as the various treatments resulting from the multidisciplinary approach, had brought the disease under control. The patient was eating better with less discomfort and pain, and functioning better with an improved quality of life. With an increased willingness to comply, the patient’s cooperation was undoubtedly vital to the success of the case, enabling her to cope with the vulnerabilities imposed by the disease and eventually overcoming them.
Conclusion
Good communication between all health professionals involved in the multi-disciplinary management of patients with EBA is imperative. More important, a good dentist-patient relationship can motivate the most anxious patient to stop the imprudent delay of proper diagnosis and treatment. The progression of EBA can be rapid, and improper diagnosis will inevitably lead to high morbidity and potential mortality. The clinician’s patience and clearly delivered reasonable advice can make a crucial difference in the efforts of patients—especially the susceptible elderly—to seek effective treatment.
References
1. Kirtschig G, Murrell D, Wojnarowska F, Khumalo N. Interventions for mucous membrane pemphigoid/cicatricial pemphigoid and epidermolysis bullosa acquisita: a systematic literature review. Arch Dermatol. 2002;138(3):380-384.
2. Fine JD. Management of acquired bullous skin diseases. N Engl J Med. 1995;333(22):1475-1484.
3. Chan LS, Chen M, Woodley DT. Epidermolysis bullosa acquisita in the elderly: Clinical manifestations, diagnosis and therapy. J Geriat Dermatol. 1996;4(2):47-52.
4. Chan LS, Woodley D. Epidermolysis Bullosa Acquisita. Available at: http://emedicine.medscape.com/article/1063083-overview. Accessed January 16, 2012.
5. Chan LS. Epidermolysis bullosa acquisita. In: Lebwohl MG, Heymann WR, Berth-Jones J, Coulson I, eds. Treatment of Skin Disease: Comprehensive Therapeutic Strategies. 2nd ed. St. Louis, MO: Mosby; 2005:191-193.
6. Mutasim DF. Autoimmune bullous dermotoses in the elderly: an update on pathophysiology, diagnosis and management. Drugs Aging. 2010;27(1):1-19.
7. Weinberg MA, Insler MS, Campen RB. Mucocutaneous features of autoimmune blistering diseases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1997;84(5):517-534.
8. Yeh SW, Ahmed B, Sami N, Razzaque Ahmed A. Blistering disorders: diagnosis and treatment. Dermatol Ther. 2003;16(3):214-223.
9. McCuin JB, Hanlon T, Mutasim DF. Autoimmune bullous diseases: diagnosis and management. Dermatol Nurs. 2006;18(1):20-25.
10. Luke MC, Darling TN, Hsu R, et al. Mucosal morbidity in patients with epidermolysis bullosa acquisita. Arch Dermatol. 1999;135(8):954-959.
11. Hakki SS, Celenligil-Naliel H, Karaduman A, et al. Epidermolysis bullosa acquisita: clinical manifestations, microscopic findings, and surgical periodontal therapy. A case report. J Periodontol. 2001;72(4):550-558.
12. Ship JA, Brennan MT, Greenberg M, et al. Pharmacology. In: Burket LW, Greenberg MS, Glick M, Ship JA. Burket’s Oral Medicine. 11th ed. Hamilton, Ontario: BC Decker Inc.; 2008:17-40.
13. Gürcan HM, Ahmed AR. Current concepts in the treatment of epidermolysis bullosa acquisita. Expert Opin Pharmacother. 2011;12(8):1259-1268.
14. Klein, IE, Lennon CA. A comprehensive approach to tissue conditioning for complete dentures. J Prosthet Dent. 1984;51(2):147-151.
15. Dootz ER, Koran A, Craig RG. Physical property comparison of 11 soft denture lining materials as a function of accelerated aging. J Prosthet Dent. 1993;69(1):114-119.
About the Authors
Chris S. Ivanoff, DDS
Assistant Professor
Department of Prosthodontics
Assistant Director Quality Assurance
University of Tennessee
College of Dentistry
Memphis, Tennessee
Timothy L. Hottel, DDS, MS, MBA
Professor
Department of Prosthodontic
Dean
University of Tennesse
College of Dentistry
Memphis, Tennessee
Related content:
For more information, read Surgical Considerations and Management of Patients with Mucocutaneous Disorders at dentalaegis.com/go/cced94