You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Periodontal disease is a prevalent condition affecting millions of individuals worldwide and remains a leading cause of tooth loss in adults.1 Its management requires both active treatment and long-term supportive periodontal therapy.2,3 Routine supportive periodontal therapy often involves ultrasonic instrumentation and air polishing to remove soft and hard deposits—including biofilm, calculus, and extrinsic stains—from teeth and dental implants.4
While these common debridement procedures are essential for establishing and sustaining periodontal health, they carry the risk of inadvertent damage to adjacent dental restorations.5,6 This risk represents a significant clinical concern, particularly given the high prevalence of direct restorations and fixed dental prostheses, such as crowns and bridges, among patients receiving periodontal care.5,7
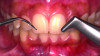
In addition to patients with periodontal disease, routine dental care for the general population often involves scale-and-polish procedures aimed at biofilm control and esthetic improvement.8 These procedures are typically performed at regular intervals to maintain oral hygiene and facilitate visual caries assessment. However, laboratory studies indicate that repeated instrumentation can compromise the integrity of dental restorations, leading to increased surface roughness and marginal deterioration over time.9 This underscores the need for debridement methods that effectively remove deposits while minimizing the risk of restoration damage—both in routine dental maintenance and during supportive periodontal therapy (Figure 1).
The repeated use of ultrasonic and air polishing devices in long-term maintenance care raises important questions regarding their effects on the surface quality and marginal integrity of various restorative biomaterials.5,6 This article provides a concise overview of laboratory evidence on these interactions and offers evidence-based recommendations to aid clinicians in reducing iatrogenic damage to restorations during periodontal care (Table 1).
Surface Roughness and Its Clinical Relevance
A smooth restoration surface is important for maintaining oral hygiene and minimizing biofilm accumulation.10 Rough surfaces enhance microbial adhesion, foster biofilm formation, and predispose the restoration to staining.10 Increased biofilm accumulation, in turn, entails risks of promoting secondary caries and periodontal disease.4 Many studies have demonstrated that the use of ultrasonic instrumentation can increase surface roughness, particularly in materials with lower mechanical resilience such as resin-modified glass-ionomer cements and resin-based composites.5 These materials are susceptible to abrasion owing to their microstructural properties and relatively low hardness.5
Ra values, which quantify average surface roughness, serve as a standard metric for evaluating material surface integrity.5 In the studies included in a recent systematic review, resin-modified glass-ionomer cements exposed to ultrasonic instrumentation exhibited Ra increases as high as 2.52 mm, indicative of substantial surface degradation.5 Similarly, resin-based composites demonstrated surface alteration, although nanohybrid composites fared slightly better due to improved filler technology.5
On the other hand, high-strength ceramics like zirconia and lithium disilicate displayed minimal surface roughness changes even after repeated debridement.5 These biomaterials have superior mechanical properties, including high hardness and low porosity, making them more resistant to mechanical insult.11 Zirconia specimens maintained Ra increases below 0.064 mm, suggesting that zirconia is well suited as a restorative biomaterial for patients requiring frequent periodontal maintenance care.5
Surface roughness not only affects biofilm accumulation but also plays a critical role in staining susceptibility and patient satisfaction.12 Rougher surfaces can lead to discoloration, necessitating frequent polishing or even replacement of restorations.12 This is particularly significant in the esthetic zone, where even minor surface imperfections can be visually noticeable.
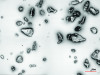
Air polishing techniques also impact surface roughness. Powders such as sodium bicarbonate and calcium carbonate are very effective in stain removal.5 However, these powders are significantly more abrasive than glycine and erythritol.5,6 Sodium bicarbonate–based air polishing has been reported to result in surface roughness increases that are comparable to or greater than those induced by ultrasonic instrumentation.5 Erythritol and glycine powders, because of their smaller particle size and lower Mohs hardness, caused significantly less damage and are now advocated for both supragingival and subgingival applications (Figure 2).5
Marginal Integrity and Restoration Longevity
Beyond surface alterations, marginal integrity is another key factor in the long-term success of dental restorations.9 A compromised margin may lead to microleakage, discoloration, material fractures, and ultimately restoration failure. Laboratory studies using microscopy and dye penetration techniques have shown that both ultrasonic instrumentation and air polishing can negatively affect the marginal seal of restorations.5
A systematic review indicated that restorations made from resin-modified glass-ionomer cements and amalgam show high rates of marginal deterioration as a result of repeated ultrasonic instrumentation.5 Additionally, data from a comprehensive laboratory investigation suggest that indirect restorations with narrow margins, particularly porcelain-fused-to-metal (PFM) crowns with all-ceramic shoulders of 0.7 mm, are especially susceptible to marginal chipping and cracking.9 The study simulated 10 years of supportive periodontal therapy, with sessions involving ultrasonic instrumentation conducted every 3 months (ie, four times a year).9 Repeated ultrasonic instrumentation resulted in a significant increase in marginal defects over time.9 Interestingly, the study revealed that wide shoulder preparations (1.5 mm) exhibited significantly fewer defects, suggesting that preparation design plays a role in resisting debridement damage.9
In addition to preparation design, the choice of luting agent significantly influences the susceptibility of restorations to damage from ultrasonic instrumentation. Marginal chipping occurs more frequently and earlier in restorations luted with glass-ionomer cement compared with those using resin-based luting materials.9 These findings suggest that both the restoration material and the luting agent affect restoration resilience to mechanical debridement. Resin-based luting materials, in particular, enhance the mechanical properties of glass-ceramic restorations, thereby preventing or delaying the onset of marginal defects.9
Comparative Effects of Debridement Methods
When comparing ultrasonic instrumentation and air polishing, both methods have distinct mechanical interactions with dental biomaterials. Ultrasonic scalers use high-frequency vibrations to break apart calculus, which can lead to pitting and microcracks in soft or brittle restorative surfaces (Figure 3).5 The concentrated and repeated application of force in a narrow area increases the likelihood of material breakdown.5
Air polishing, by contrast, uses a pressurized stream of abrasive powder and water.5 The abrasiveness of this method largely depends on the type of powder used. Studies comparing powders have consistently shown that sodium bicarbonate and calcium carbonate cause substantial surface alterations in resin-modified glass-ionomer cements, resin-based composites, amalgams, and veneering ceramics.5 On the other hand, low-abrasive powders, such as glycine and erythritol, offer a gentler cleaning process, preserving the structural and esthetic integrity of restorations.5
Evidence from multiple studies suggests that erythritol and glycine are the powders of choice for air polishing when restorative biomaterials are present.5,6 These powders are particularly suitable for maintenance around esthetic restorations, implants, and exposed root surfaces.5,6 Outcomes associated with these low-abrasive powders include improved patient comfort, reduced gingival trauma, and lower frequency of restorative damage.5,6
Ultrasonic scalers are all but indispensable for removing calculus deposits, especially in deep periodontal pockets or furcation areas. However, their use should be limited around delicate restorations to minimize potential damage.5,6 When ultrasonic instrumentation is necessary, a targeted approach—focusing on calculus and biofilm removal while avoiding prolonged contact with restoration margins—can help preserve restorative integrity. Training and calibration of the clinical operator further ensure the judicious application of force and precise instrument handling.
Table 2 provides a summary and comparison of debridement methods.
Mitigating Iatrogenic Damage
To mitigate the risk of iatrogenic damage during debridement, dental practitioners and hygienists should employ several precautionary strategies. First, the choice of debridement technique should be guided by the nature of the deposits that require removal and the type and location of any restorative material. For instance, ultrasonic instrumentation should be avoided on resin-modified glass-ionomer cements and amalgam restorations unless strictly necessary.5 When ultrasonic instruments are used, light contact with the calculus alone is often sufficient, while avoiding adjacent and subjacent tooth or restorative surfaces. The use of magnification devices can facilitate targeted calculus removal.
Second, the use of rubber cup and paste polishing following debridement has been shown to reduce surface roughness.5 This technique is particularly beneficial for resin-based composite and glass-ionomer restorations, as it can help restore a smoother surface and reduce subsequent biofilm adherence. However, its effectiveness is limited; in cases of significant surface roughening, polishing alone is insufficient to fully reverse the damage.5
Third, when delivering fixed dental prostheses, dental practitioners should consider using resin-based luting materials, especially in patients who need supportive periodontal therapy.9 Resin-based luting materials demonstrate higher flexural strength and improved marginal sealing compared with traditional cements. By enhancing marginal integrity, resin-based luting materials reduce the risk of microleakage and marginal breakdown over time.9
Another crucial strategy involves patient education and communication.13 Patients should be informed about the potential risks of restoration damage during maintenance procedures and involved in decisions regarding the choice of biomaterials and debridement techniques.13 Informed consent processes should include a discussion about the long-term implications of restorative care in the context of periodontal health.
Dental teams should also implement standardized protocols that prioritize both periodontal health and the preservation of restorations during debridement. Such protocols may include regular review of the patient’s restorative status, thorough documentation of vulnerable areas—from both periodontal and restorative perspectives—and use of magnification devices to enhance precision during procedures.
Material-Specific Recommendations
The following recommendations are suggested when performing periodontal maintenance around different restorative materials:
Resin-modified glass-ionomer cements: These materials are prone to surface roughening and marginal breakdown. For removal of soft deposits on or near these restorations, air polishing with erythritol or glycine powders is recommended. Ultrasonic instrumentation should be avoided where feasible.5
Resin-based composites: Ultrasonic instrumentation near the gingival margins of resin-based composite restorations should be avoided whenever possible, as these areas are susceptible to damage.5
Amalgams and gold alloys: Caution should be exercised when using ultrasonic instruments around amalgam and gold alloy restorations to prevent marginal deterioration.5
Zirconia and lithium disilicate: These materials demonstrate high resistance to debridement-induced damage and are preferred in patients undergoing supportive periodontal therapy.5
Porcelain-fused-to-metal restorations: PFM restorations are susceptible to marginal chipping, especially at narrow shoulder preparations and at sites with all-ceramic margins.9 Ultrasonic instrumentation should be avoided at these sites unless strictly necessary for calculus removal.5
Clinical Implications
Understanding the interactions between debridement tools and restorative materials is critical to providing high-quality periodontal maintenance. Damage to restorations not only reduces their functional longevity but also increases the need for costly replacements and can lead to adverse patient outcomes such as hypersensitivity and esthetic compromise.13
A site-specific, tailored approach is essential for effective removal of deposits—including biofilm, calculus, and extrinsic stains—and preservation of restoration integrity.14,15 For example, air polishing with erythritol or glycine powders can be employed safely and effectively for biofilm management around restored teeth, reserving ultrasonic instrumentation primarily for calculus and biofilm removal in nonrestored areas.5
Moreover, comprehensive treatment planning should account for not only immediate clinical outcomes but also the long-term maintenance needs of the patient.13 This includes selecting durable restorative biomaterials, providing patient education on the care of restorations, and implementing regular follow-up. Equally important is interdisciplinary collaboration among periodontists, general dentists, and hygienists to ensure that periodontal and restorative care plans are well integrated. Such coordinated care enhances clinical outcomes, mitigates complications, and supports a preventive model focused on long-term oral health, restoration longevity, and patient satisfaction.13
Summary
Ultrasonic and air polishing techniques are important tools in periodontal care but must be used judiciously when teeth with restorations are involved.5,6 Laboratory evidence suggests that high-strength ceramics such as zirconia and lithium disilicate tolerate debridement procedures better than resin-modified glass-ionomer cements and resin-based composites.5,6 Air polishing with low-abrasive powders, such as glycine and erythritol, is recommended to minimize surface alterations and marginal damage to dental restorations.5,6 It is important to recognize that laboratory findings, while informative, do not fully replicate clinical conditions. Thus, conclusions for patient care should be drawn with caution. Nevertheless, it appears prudent that clinical decisions consider biomaterial type, margin design, and debridement method to optimize outcomes and preserve restoration integrity over time.
ACKNOWLEDGMENT
The authors thank Mirka Solomon and Jeanine Béatrice Wyss, B Dent Med, for their assistance in the preparation of the images shown in Figure 1, and Figures 2 and 3, respectively.
ABOUT THE AUTHORS
Florin Eggmann, PD Dr. med. dent.
Lecturer, Department of Periodontology, Endodontology, and Cariology, University Center for Dental Medicine Basel (UZB), University of Basel, Basel, Switzerland
Jeronim Esati, Dr. med. dent.
Resident, Department of Periodontology, Endodontology, and Cariology, University Center for Dental Medicine Basel (UZB), University of Basel, Basel, Switzerland
Markus B. Blatz, DMD, PhD
Professor of Restorative Dentistry, Chair, Department of Preventive and Restorative Sciences, and Assistant Dean, Digital Innovation and Professional Development, University of Pennsylvania, School of Dental Medicine, Philadelphia, Pennsylvania
Queries to the author regarding this course may be submitted to
authorqueries@conexiant.com.
REFERENCES
1. GBD 2021 Oral Disorders Collaborators. Trends in the global, regional, and national burden of oral conditions from 1990 to 2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet. 2025;405(10482):897-910.
2. Sanz M, Herrera D, Kebschull M, et al. Treatment of stage I-III periodontitis – the EFP S3 level clinical practice guideline. J Clin Periodontol. 2020;47(suppl 22):4-60.
3. Afrashtehfar KI, Assery NM, Alblooshi KAK, Schmidlin PR. Maintaining periodontally compromised teeth seems more cost-effective than replacing them with dental implants. Evid Based Dent. 2024;25(3):129-130.
4. Mombelli A. Maintenance therapy for teeth and implants. Periodontol 2000. 2019;79(1):190-199.
5. Esati J, Amran T, Weiger R, et al. Adverse effects of ultrasonic instrumentation and air polishing on dental restorations: a systematic review of laboratory studies. J Esthet Restor Dent. 2025;37(6):1372-1383.
6. Harman A, Murchie B. Do ultrasonic instrumentation and air polishing procedures adversely affect dental restorations? Evid Based Dent. 2025;26(3):125-127.
7. Schmidt JC, Vogt S, Imboden M, et al. Dental and periodontal health in a Swiss population–based sample of older adults: a cross-sectional study. Eur J Oral Sci. 2020;128(6):508-517.
8. Lamont T, Worthington HV, Clarkson JE, Beirne PV. Routine scale and polish for periodontal health in adults. Cochrane Database Syst Rev. 2018;12(12):CD004625.
9. Eggmann F, Schiavone F, Amato J, et al. Effect of repeated ultrasonic instrumentation on single-unit crowns: a laboratory study. Clin Oral Investig. 2022;26(3):3189-3201.
10. Hamza B, Eliades T, Attin T, et al. Initial bacterial adherence and biofilm formation on novel restorative materials used in paediatric dentistry. Dent Mater. 2024;40(3):573-579.
11. Spitznagel FA, Boldt J, Gierthmuehlen PC. CAD/CAM ceramic restorative materials for natural teeth. J Dent Res. 2018;97(10):1082-1091.
12. Sarici T, Dayi B. Evaluation of the effects of different polishing systems on surface roughness and surface discoloration of various restorative materials. BMC Oral Health. 2025;25(1):978.
13. Eggmann F, Orta AL, Abdulmajeed A, et al. Interdisciplinary Delphi study by PROSEC North America: recommendations on single indirect restorations made from ceramic and nonmetallic biomaterials for posterior teeth. J Esthet Restor Dent. 2025;37(3):809-820.
14. Furrer C, Battig R, Votta I, et al. Patient acceptance of “guided biofilm therapy” [in German]. Swiss Dent J. 2021;131(3):229-234.
15. Cyris M, Festerling J, Kahl M, et al. Guided biofilm therapy versus conventional protocol – clinical outcomes in non-surgical periodontal therapy. BMC Oral Health. 2024;24(1):1105.