You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Penicillin allergy is an important consideration in dental practice because of the widespread use of penicillin and its derivatives in antibiotic prophylaxis and treatment of bacterial infections. As penicillin allergy is reported by about 10% of all patients—with allergy reporting having a female predilection—it is one of the most commonly reported drug allergies.1,2 However, about 95% of reported allergies are false.3,4 True immunoglobulin E (IgE)–mediated hypersensitivity reactions have been found to be much less common, with data indicating that only about 1% of the population has a true penicillin allergy confirmed by skin testing.5 IgE-mediated penicillin allergies are said to be decreasing because of less intravenous administration of penicillin and the fact that oral amoxicillin is less likely than other penicillin derivatives to cause a severe anaphylactic reaction.1 In addition, patients with immune-mediated allergy show decreased response to penicillin as they get older, with about 80% of patients tolerant after 10 years.1,3
Mechanisms and Clinical Manifestations of Penicillin Allergies
Gell and Coombs classified hypersensitivity into four main types: I, II, III, and IV (Table 1). The prevalence of life-threatening anaphylactic reactions to penicillin ranges between 0.02% and 0.04%.6 Clinical manifestations of penicillin allergies can be immediate (IgE-mediated via Type I hypersensitivity reaction) or delayed (non-IgE mediated via complement system) based on the timing and type of immune response (Table 2). Most delayed reactions are via Type IV hypersensitivity reaction, but Types II and III can also play a role.1 Such delayed reactions can range from mild to severe and manifest across various organ systems. The most common clinical manifestation, however, is cutaneous.6,7
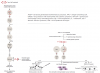
The mechanism of action of Types I and IV hypersensitivity reactions in penicillin allergy is depicted in Figure 1.
Mechanisms
IgE-mediated Type I hypersensitivity: Type I hypersensitivity reactions are immediate and IgE-mediated. They occur within minutes to hours after exposure to penicillin. Upon initial exposure, penicillin acts as a hapten, binding to proteins and forming a complex that is recognized as foreign by the immune system. This complex stimulates B cells to produce IgE antibodies specific to the penicillin–protein complex. These IgE antibodies bind to Fc receptors on mast cells and basophils. Upon subsequent exposure to penicillin, crosslinking of the bound IgE occurs on these cells, leading to degranulation and release of histamine and other inflammatory mediators. This results in symptoms ranging from urticaria and angioedema to anaphylaxis.8
Type IV hypersensitivity: Type IV hypersensitivity reactions, also known as delayed-type hypersensitivity reactions, play a significant role in penicillin allergies. These reactions are mediated by T cells rather than antibodies, which distinguishes them from other hypersensitivity types. The pathophysiology involves various T-cell subsets and mechanisms that lead to tissue damage and inflammation.9 When penicillin or its metabolites bind to proteins in the body, they form a new antigenic complex that can be recognized by T cells, leading to their activation and proliferation. These T cells release cytokines that recruit and activate macrophages and other inflammatory cells. Three models can explain this process:
1. Hapten/pro-hapten model: Penicillin acts as a hapten, a small molecule that binds covalently to a larger protein, creating a new antigenic determinant. This complex is then processed by antigen-presenting cells and presented to T cells, leading to their activation and proliferation. Upon re-exposure, memory T cells respond quickly, leading to a hypersensitivity reaction.9
2. Pharmacologic interaction with immune receptors (P-I) concept: This model suggests that penicillin can directly bind to the T-cell receptor or major histocompatibility complex (MHC) molecules without the need for antigen processing. This non-covalent binding can rapidly activate T cells and trigger an immune response.9
3. Altered peptide repertoire model: In this scenario, penicillin alters the peptide-binding groove of MHC molecules, changing the peptides that are presented to T cells. This alteration can lead to the activation of T cells that were not previously reactive to the native peptide–MHC complex.
Clinical Manifestations
Immediate reactions: Immediate reactions occur within an hour of drug administration and are mediated by IgE antibodies. Symptoms include urticaria, which is the most common; angioedema; bronchospasm, which can present as wheezing or shortness of breath; anaphylaxis, which is life-threatening and requires immediate intervention10; laryngeal edema; syncope; hypotension; and cardiac arrest, which can be fatal without timely intervention.6
Delayed reactions: Delayed reactions occur hours to days after drug exposure and are typically non-IgE-mediated. The clinical manifestations of Type IV hypersensitivity to penicillin can vary. They include maculopapular exanthems, drug reaction with eosinophilia and systemic symptoms (DRESS), and serum sickness-like reactions.8 Others include exfoliative dermatitis, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), acute interstitial nephritis, drug-induced hemolytic anemia, and non-pruritic maculopapular eruption.6,9 Severe conditions such as SJS or TEN involve extensive skin and mucosal damage due to widespread T-cell activation and cytokine release.11
Penicillin Allergy Reporting
Patients may report penicillin allergies in response to specific drug allergy questions during history taking.12 While patient history is important in allergy evaluation,1 it can be inaccurate and unspecific, especially when the incidence being reported occurred a long time ago and multiple drugs may have been ingested.12 False reporting may overestimate the prevalence of true allergy, and, moreover, it can expose patients to negative impacts, such as increased cost, toxicity, reduced effectiveness of alternative medications, and longer duration of morbidity.
The discrepancy between reported and actual allergy rates highlights the importance of accurate diagnosis and patient education. Correct diagnosis of penicillin allergy is needed to optimize antibiotic selection in dental patients. Therefore, confirmatory testing is necessary when individuals report a history of penicillin allergy.
Diagnosis
Accurate diagnosis of penicillin allergy is essential to avoid unnecessary circumvention of this important class of antibiotics and maintain antibiotic stewardship. The following diagnostic tests may be used:
Skin test: Penicillin skin testing is considered the benchmark for diagnosing IgE-mediated penicillin allergy. It involves intradermal and prick testing with penicillin determinants and a saline control. A positive test indicates a high likelihood of allergy.13 Reported penicillin allergy is confirmed through skin prick tests (SPTs) and/or intradermal tests (IDTs), followed by an oral challenge in selected cases. Testing is recommended to accurately identify penicillin-allergic individuals.4,12
SPT and IDT are critical in diagnosing immediate drug allergies that are IgE mediated. SPT is recommended first for screening; if the test is negative, an IDT is done. The IDT is more sensitive for drug-specific IgE.12 Skin tests can be used to demonstrate both immediate and delayed hypersensitivity to penicillin. In immediate reactions, a positive SPT or IDT can be seen within 20 minutes of administration, whereas in delayed reactions, a positive patch test and/or late-reading intradermal test takes several hours to days after administration to appear.14 These tests should be conducted by trained allergists or immunologists in a controlled setting to mitigate the risk of adverse reactions.
Drug provocation test/drug challenge: This diagnostic test involves exposing the patient to graded oral doses of the medication. Gradually increased doses of penicillin are administered under close medical supervision. A negative challenge indicates tolerance to penicillin.15 This approach is the benchmark used to confirm that a patient is not allergic after skin tests are negative, as the negative predictive value of skin tests is less than 100%.4,12 Drug challenge should be carried out with close surveillance of the patient and readiness to respond to any adverse events. Oral tolerance tests can be used alone in the delabeling (ie, removal of penicillin allergy from the medical record) of low-risk patients.1,4
In vitro tests: While in vitro tests may be utilized, their actual usefulness is uncertain given insufficient data.14 As such, in vitro tests cannot serve as replacements for skin testing.6 Such tests include specific IgE testing, which measures IgE antibodies against penicillin but has lower sensitivity compared to skin testing,16 and basophil activation testing, which is a newer diagnostic tool that measures the activation of basophils in response to penicillin and can be useful in cases where skin testing is contraindicated.15,17,18
Risk Stratification
When a patient presents with a self-reported penicillin allergy, risk stratification aims to classify the patient based on their likelihood of having a true penicillin allergic reaction. Risk stratification is crucial in evaluating patients with a reported penicillin allergy and may aid in antibiotic selection in cases where penicillin may be the best drug therapy option. Factors such as the nature of the reported reaction, the time elapsed since the reaction, and the patient’s clinical history help determine the likelihood of a true allergy.19 Stratification helps in deciding whether to proceed with further testing or consider alternative antibiotics and is essential for identifying patients who may safely receive penicillin despite a reported allergy.
Risk stratification based on allergy history is reported to reduce treatment failure and increase clinical outcomes in patients that had methicillin-sensitive bacteremia and reported penicillin allergy when compared to administering alternative antibiotics.1 Stratification involves detailed patient history and categorizing reactions into immediate (IgE-mediated) and non-immediate (T-cell mediated) hypersensitivities. Immediate reactions occur within 1 hour of exposure and include symptoms such as urticaria, angioedema, and anaphylaxis. Non-immediate reactions manifest hours to days later and include morbilliform rashes and SJS.6
Thorough history taking is important,2 and patient allergy history can be used to place patients into high-, moderate-, or low-risk categories. Generally, high-risk factors include a history of anaphylaxis, angioedema, or urticaria occurring within hours of penicillin administration, severe cutaneous adverse reactions such as SJS or TEN, and/or documented positive penicillin skin test or specific IgE test. Low-risk factors include isolated gastrointestinal symptoms, nonspecific rash occurring days after administration, and family history of penicillin allergy without personal history of reaction.
The penicillin allergy decision rule (PEN-FAST) provides a clinical decision-making framework based on factors like the nature of the allergic reaction, time elapsed since the reaction, and whether treatment was required during the initial reaction.20 PEN-FAST aids clinicians in identifying low-risk patients and stratifying patients to determine their risk of true penicillin allergy and guide further testing and treatment decisions.20,21 The degree of penicillin allergy risk is ascertained using a point-scoring system with values ranging from 0 to 5 based on the total number of points (Table 3).4 A total score of 0 indicates very low risk of positive penicillin allergy, a total score of 1–2 signifies low risk, a total score of 3 denotes moderate risk, and a total score of 4–5 indicates high risk for positive penicillin allergy. It should be noted that screening patients who have not reported a previous allergic reaction is not recommended.2,12
Drug Choices in Penicillin-Allergic Patients
Selecting alternative antibiotic drugs for patients who report penicillin allergy is a major consideration for clinicians. Cephalosporins seem to be generally avoided, as cross-reactivity with penicillin allergy has been reported, although this concern appears to be lessening as data increasingly shows a lower risk of cross-reactivity, from previously reported 8% to 2%.1,2 Cross-reactivity is reported mainly with amino cephalosporins (Table 4). Cefazolin has a different side chain and is less likely to have a cross-reactivity.1 Cross-reactivity between penicillin and other medications like carbapenems has been reported to be 1%, and no known cross-reactivity has been reported with monobactams.1
Alternative treatment options in allergic patients can be categorized into beta-lactam alternatives and non-beta-lactam alternatives. For dental patients with penicillin allergy, alternative antibiotics should be considered based on the type of infection and the patient’s allergy profile.
Beta-lactam alternatives include cephalosporins, namely cephalexin and cefazolin; carbapenems, including meropenem and imipenem; and monobactam, namely aztreonam.1 Cephalosporins should be used with caution in patients with penicillin allergy, and only when the allergy is not severe. As noted earlier, while cross-reactivity between penicillins and cephalosporins reportedly has been lessening, the highest risk is associated with first-generation cephalosporins.22 Carbapenems, having very low cross-reactivity with penicillin, are a safe alternative for patients with penicillin allergies,15 and monobactams are safe in both immediate and delayed penicillin allergies because there is no known cross-reactivity between penicillin and aztreonam.15,23
Non-beta-lactam alternative options include fluoroquinolones, such as ciprofloxacin; nitroimidazoles like metronidazole; macrolides, such as azithromycin; glycopeptides like vancomycin; tetracyclines, such as doxycycline; lincosamides, including clindamycin; oxazolidinones, such as linezolid; sulfonamides, including trimethoprim-sulfamethoxazole; aminoglycosides like gentamicin; and lipopeptides, such as daptomycin.1
Macrolides such as erythromycin and azithromycin are commonly used alternatives in penicillin allergy cases, especially for orofacial infections. They are generally well-tolerated and effective against common oral pathogens24; however, interaction with drugs like digoxin is an important consideration when deciding to prescribe macrolides.25 Clindamycin should be avoided as it may be associated with Clostridioides difficile pseudomembranous colitis.25,26 Muller and Ouanounou recently documented the advantages of prescribing azithromycin over clindamycin for patients allergic to penicillin.27
Tetracyclines, including doxycycline, are broad-spectrum antibiotics useful in treating periodontal disease. However, their use is limited by potential side effects on fetuses in pregnant patients, young children, and the skeletal system28; such side effects may include inhibited bone growth, skeletal malformations, and teeth discoloration. Metronidazole is effective against anaerobic bacteria and is often used in combination with other antibiotics to treat severe dental infections. It is a viable option for patients with penicillin allergy.26
Some alternative drugs can cause adverse reactions. Vancomycin, for example, can cause “red man syndrome,” DRESS syndrome, and nephrotoxicity among others.1,15 Vancomycin-resistant enterococci may also be a resultant downside to the use of this medication.1 Clindamycin, as stated earlier, and fluroquinolones are associated with C difficile infections.
For antibiotic prophylaxis, the American Heart Association’s updated guideline is recommended (Table 5),29 along with American Dental Association guidelines.
Management of a Penicillin Allergic Reaction in a Dental Setting
While several strategies can be employed to manage a penicillin allergic reaction in the dental setting, there are a number of steps that can be taken to try to avoid such situations. These include performing a preoperative assessment,30 in which detailed allergy history and potential testing is conducted before carrying out any dental procedures that require antibiotics. Also, for patients with a confirmed penicillin allergy, penicillin and related beta-lactam antibiotics should be avoided. Cross-reactivity with cephalosporins, carbapenems, and monobactams varies, and caution is advised.13
For patients requiring penicillin but who have a confirmed allergy, desensitization can be performed. This involves administering gradually increased doses of penicillin to induce temporary tolerance. Desensitization should only be done in a controlled setting with medical oversight.15 It is important to note that desensitization is contraindicated in severe Type IV hypersensitivity reactions like SJS, DRESS, and TEN.15
In the event of an actual penicillin allergic reaction, management strategies that can be used to ensure patient safety while optimizing treatment outcomes include:
Training in emergency protocols: Management of medical emergencies in a dental setting begins with the staff being prepared and properly trained. Prompt recognition of allergy signs and symptoms, immediate response, and availability of emergency equipment and medications are critical.2,12 The primary aim in the management of a medical emergency is to ensure that vital organs (eg, brain and heart) are not deprived of oxygenated blood while underlying pathophysiologies are addressed and remedied.2
Epinephrine administration: Anaphylaxis is an acute reaction to penicillin that can be fatal. Once it is noticed, the dental procedure being conducted should be stopped, the patient’s airway, breathing, and circulation assessed, and appropriate medications administered. Epinephrine is the drug of choice in anaphylactic reactions,2,30 with 0.3 mg to 0.5 mg given intramuscularly or 0.1 mg given intravenously. Attention should be paid to the risk-benefit ratio when administering epinephrine in patients with ischemic heart diseases.2
Use of antihistamines: Antihistamines also can be useful in the management of allergic reactions. Oral antihistamines may be used in mild reactions that are not life-threatening.30 In life-threatening allergic reactions, injectable antihistamines are administered alone (when the signs are mainly on the skin) or in combination with other agents as part of the management.2 Diphenhydramine (25 mg to 50 mg) or chlorpheniramine (10 mg to 20 mg) can be administered.
Finally, educating patients about their allergy, including the importance of accurate reporting and understanding the potential risks of cross-reactivity, is crucial. This education should include information on recognizing signs of an allergic reaction and seeking medical help promptly.
Conclusion
Penicillin allergy can present a significant challenge in dental practice that affects antibiotic selection for prophylaxis and treatment of dentofacial infections. Use of penicillin antibiotics in a dental setting, therefore, requires careful risk assessment, accurate diagnosis, and appropriate management strategies. Practitioners should understand the epidemiology, clinical manifestations, and diagnostic tools for penicillin allergy. Patient evaluation through detailed history taking and risk stratification of reported allergies is needed to differentiate true allergy from non-allergic reactions, and this helps optimize antibiotic therapy. Additionally, being knowledgeable about alternative treatment options helps facilitate safe and effective care to patients. Collaboration with allergists or immunologists to properly confirm penicillin allergies aids in more accurate labeling of allergies. The management of penicillin allergic reactions entails proper patient evaluation, consideration of alternative antibiotics, and medical emergency preparedness. Dental professionals must adopt evidence-based approaches to penicillin allergy labeling and management to ensure safe and effective care for patients while minimizing avoidable alternative antibiotic use and their associated disadvantages. Continued research and education are necessary to improve the management of penicillin allergy in dental patients, with the goal of enhancing patient outcomes and safety.
ABOUT THE AUTHORS
Ifeanyichukwu Ezeliorah, BDS, DDS
Resident, Dental Anesthesia Specialty Program, Faculty of Dentistry, University of Toronto, Toronto, Ontario, Canada
Aviv Ouanounou, BSc, MSc, DDS
Associate Professor, Department of Clinical Sciences, Pharmacology and Preventive Dentistry, Faculty of Dentistry, University of Toronto, Toronto, Ontario, Canada; Fellow, International College of Dentists; Fellow, American College of Dentists; Fellow, International Congress of Oral Implantologists
Queries to the author regarding this course may be submitted to authorqueries@conexiant.com.
REFERENCES
1. Shenoy ES, Macy E, Rowe T, Blumenthal KG. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321(2):188-199.
2. Ramsey A. Penicillin allergy and perioperative anaphylaxis. Front Allergy. 2022;3:903161.
3. Turner P. Delabelling penicillin allergy is not rocket science. Arch Dis Child. 2023;108(5):329.
4. Copaescu AM, Vogrin S, James F, et al. Efficacy of a clinical decision rule to enable direct oral challenge in patients with low-risk penicillin allergy: the PALACE randomized clinical trial. JAMA Intern Med. 2023;183(9):944-952.
5. Macy E. Penicillin and beta-lactam allergy: epidemiology and diagnosis. Curr Allergy Asthma Rep. 2014;14(11):476.
6. Gonzalez-Estrada A, Radojicic C. Penicillin allergy: a practical guide for clinicians. Cleve Clin J Med. 2015;82(5):295-300.
7. Patterson RA, Stankewicz HA. Penicillin allergy. June 20, 2023. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; January 2025. PMID: 29083777.
8. Pichler WJ. Immune pathomechanism and classification of drug hypersensitivity. Allergy. 2019;74(8):1457-1471.
9. Copaescu A, Gibson A, Li Y, et al. An updated review of the diagnostic methods in delayed drug hypersensitivity. Front Pharmacol. 2021;11:573573.
10. Khan DA, Solensky R. Drug allergy. J Allergy Clin Immunol. 2010;125(2 suppl 2):S126-S137.
11. Jourdan A, Sangha B, Kim E, et al. Antibiotic hypersensitivity and adverse reactions: management and implications in clinical practice. Allergy Asthma Clin Immunol. 2020;16:6.
12. Demoly P, Adkinson NF, Brockow K, et al. International Consensus on drug allergy. Allergy. 2014;69(4):420-437.
13. Solensky R. Penicillin allergy as a public health measure. J Allergy Clin Immunol. 2014;133(3):797-798.
14. Torres MJ, Romano A, Celik G, et al. Approach to the diagnosis of drug hypersensitivity reactions: similarities and differences between Europe and North America. Clin Transl Allergy. 2017;7:7.
15. Broyles AD, Banerji A, Barmettler S, et al. Practical guidance for the evaluation and management of drug hypersensitivity: specific drugs. J Allergy Clin Immunol Pract. 2020;8(9S):S16-S116.
16. Confino-Cohen R, Rosman Y, Meir-Shafrir K, et al. Oral challenge without skin testing safely excludes clinically significant delayed-onset penicillin hypersensitivity. J Allergy Clin Immunol Pract. 2017;5(3):669-675.
17. Bennett MR, Mathioudakis AG, Wu J, et al. Performance characteristics of basophil activation tests for diagnosing penicillin allergy: a meta-analysis. J Allergy Clin Immunol Pract. 2024;12(3):714-723.e5.
18. Koumaki D, Gregoriou S, Evangelou G, et al. Real-life utility of basophil activation test in the diagnosis of immediate hypersensitivity drug reactions. Dermatol Ther (Heidelb). 2023;13(12):3229-3239.
19. Bhattacharya S. The facts about penicillin allergy: a review. J Adv Pharm Technol Res. 2010;1(1):11-17.
20. Trubiano JA, Vogrin S, Chua KYL, et al. Development and validation of a penicillin allergy clinical decision rule. JAMA Intern Med. 2020;180(5):745-752.
21. Whelan SO, Moriarty F. Predicting true penicillin allergy in adults. Am Fam Physician. 2021;103(12):760-761.
22. Pichichero ME. Cephalosporins can be prescribed safely for penicillin-allergic patients. J Fam Pract. 2006;55(2):106-112.
23. Gaeta F, Valluzzi RL, Alonzi C, et al. Tolerability of aztreonam and carbapenems in patients with IgE-mediated hypersensitivity to penicillins. J Allergy Clin Immunol. 2015;135(4):972-976.
24. Patel PH, Hashmi MF. Macrolides. May 16, 2023. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; January 2025. PMID: 31855339.
25. Ouanounou A, Ng K, Chaban P. Adverse drug reactions in dentistry. Int Dent J. 2020;70(2):79-84.
26. Sanders JL, Houck RC. Dental abscess. February 20, 2023. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; January 2025. PMID: 29630201.
27. Muller D, Ouanounou A. Should we start prescribing azithromycin over clindamycin? Ontario Dentist. 2024;99(10):20-23.
28. Warner AJ, Hathaway-Schrader JD, Lubker R, et al. Tetracyclines and bone: unclear actions with potentially lasting effects. Bone. 2022;159:116377.
29. Wilson WR, Gewitz M, Lockhart PB, et al; American Heart Association Young Hearts Rheumatic Fever, Endocarditis and Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; Council on Cardiovascular and Stroke Nursing; and the Council on Quality of Care and Outcomes Research. Prevention of Viridans group streptococcal infective endocarditis: a scientific statement from the American Heart Association [Erratum in: Circulation. 2021;144(9):e192. Erratum in: Circulation. 2022;145(17):e868]. Circulation. 2021;143(20):e963-e978.
30. Haas DA. Management of medical emergencies in the dental office: conditions in each country, the extent of treatment by the dentist. Anesth Prog. 2006;53(1):20-24.