You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Dental implants are considered a highly predictable and safe solution for restoring function and esthetics in partially or fully edentulous patients, offering long-term success. The global market for dental implants was valued at $4.12 billion in 2021, with more than 9 million implants placed that year alone.1 According to the Fifth International Team for Implantology (ITI) Consensus Conference, implant-supported restorations demonstrate an impressive 5-year survival rate of 97.1%.2
Despite this outstanding survival rate, implant restorations are still subject to esthetic, technical, and/or biological complications that may lead to early and/or late implant failure. Early implant failures are associated with several factors, including poor bone quality, insufficient bone volume, systemic disease, surgical technique, and prosthetic overload. Medical conditions that impair bone healing, such as uncontrolled diabetes or smoking, can hinder osseointegration and increase the likelihood of failure. Further, perioperative infection can also lead to early implant failure if not appropriately addressed.3
The use of antibiotics in implant dentistry has long been a subject of debate, with several controlled clinical trials showing inconsistent results.4,5 To reduce the risk of infections following dental implant placement, various prophylactic systemic antibiotic protocols have been recommended. Historically, antibiotics have been prescribed prior to surgery and continued for up to 10 days afterward. A widely followed regimen included administering 2 g of phenoxymethylpenicillin (penicillin-V) orally about 1 hour before the procedure, followed by 2 g twice daily for 10 days.6 Concerns with such protocols, particularly in the absence of clinical infection, include adverse side effects and allergic reactions. Antibiotic use can lead to a range of adverse effects, from mild gastrointestinal symptoms, such as nausea, diarrhea, and abdominal pain, to more serious conditions. Disruption of gut microflora can result in overgrowth of Candida and Clostridioides difficile, which is particularly common with broad-spectrum antibiotics. Allergic reactions to antibiotics are common, including dermatologic responses such as rashes and, less commonly, anaphylactic reactions. Penicillin is believed to account for 0.7% to 10% of all anaphylaxis cases. Another significant issue linked to the extensive use of antibiotics is the development of antibiotic-resistant bacteria and its impact on public health. Such adverse consequences of antibiotic therapy require thoughtful consideration of risks and benefits to evaluate the use of prophylactic antibiotic therapy.
Given these concerns and the risk–benefit analysis associated with them, updated guidelines involve administering 2 g of penicillin-V (or amoxicillin or amoxicillin/clavulanate) 1 hour before surgery, followed by 500 mg of penicillin-V four times a day for 1 day during dental implant placement surgery if postoperative antibiotic coverage is determined to be optimal.7 It should be noted that a single prophylactic dose of antibiotics has demonstrated efficacy in reducing implant failure rates,8 and, therefore, antibiotic treatment should be considered in the context of risks versus benefits and applied on an individualized basis. While current protocols suggest penicillin and/or amoxicillin as a first-line antibiotic in patients who may benefit from perioperative antibiotic prescription, approximately 10% of the US population reports having a penicillin allergy. Research suggests that the prevalence of penicillin allergy may be overestimated, and it is estimated that less than 1% of the population is truly allergic.9 Emerging evidence suggests that self-reported penicillin allergies may influence dental implant failure rates, raising concerns about the efficacy of alternative prophylactic antibiotics, such as clindamycin, or the impact of omission of antibiotic use altogether.10
This article examines how and why these self-reported allergies could affect implant outcomes, focusing particularly on whether patients with penicillin allergies are at a higher risk for implant failure and post-surgical complications compared to those without such allergies. By analyzing existing findings, this review aims to provide increased understanding of the clinical implications of these allergies on implant success and patient care.
Weighing Risks and Benefits for Perioperative Use of Antibiotic Therapy
While perioperative antibiotic use has been associated with decreased early implant failure rates,8,11 the routine use of extended perioperative antibiotic prescriptions may not be warranted due to other risks, including adverse events such as gastrointestinal distress, allergic reactions, and antibiotic resistance.12 For uncomplicated dental implant surgery, postoperative infection rates are low (generally less than 1%) with proper technique and strict patient selection; real-world infection rates range from 2.8% to 6.5% of patients.13 Factors associated with postoperative dental implant infections include uncontrolled diabetes mellitus, smoking, poorly executed surgical technique, implant surface characteristics, and bone quality.14 Clinicians should carefully assess individual patients and dental implant sites and perform risk–benefit analysis to determine if antibiotic use is warranted and the optimal course of perioperative implant therapy.
Penicillin Allergy and Implant Failure Rates:
The Potential Link
The effectiveness of preoperative antibiotics in preventing dental implant failure has been a topic of ongoing debate. A Cochrane meta-analysis that included six randomized controlled trials (RCTs) with 1,162 participants examined this issue and found that prophylactic delivery of 2 g or 3 g of amoxicillin immediately before dental implant placement surgery significantly reduced implant failure rates (risk ratio [RR] 0.33; 95% confidence interval [CI] 0.16 to 0.67; P = .002).15 The number needed to treat (NNT) to prevent one implant failure was 25 (95% CI 14 to 100), assuming an implant failure rate of 6% in participants not receiving antibiotics. There was borderline significance for prosthesis failures (RR 0.44; 95% CI 0.19 to 1.00), but no significant differences were observed for postoperative infections or other adverse events.
This evidence has been used to support the use of a single 2 g dose of preoperative amoxicillin to reduce implant failure rates, while the effectiveness of postoperative antibiotics remains unclear.15 A prospective double-blind RCT tested the effect of prophylactic antibiotics on postoperative morbidity and osseointegration of dental implants.16 While no failures occurred in the antibiotic group, five failures were recorded in the placebo group, although this was not statistically significant. The placebo group also experienced more swelling, bruising, pain, and interference with daily activities compared to the group that received antibiotics. The findings of this investigation suggest that prophylactic antibiotics may improve implant survival and reduce postoperative discomfort, although other factors such as surgical technique also play a role.15
Most of the evidence regarding the association between penicillin allergy and dental implant failure rates is derived from retrospective studies. Several studies have demonstrated that penicillin-allergic patients had a significantly higher risk of dental implant failure. In one retrospective analysis, 24.68% of patients reporting penicillin allergy experienced implant failure compared to 8.03% of non-allergic patients.10 Additionally, the fixture failure rate was higher in the penicillin-allergic group (18.86% vs 5.17%). In the investigation, penicillin-allergic patients had a 3.84 times higher risk of failure at the patient level and 3.64 times at the implant level compared to non-allergic patients.
Similarly, another retrospective study demonstrated higher implant failure rates in penicillin-allergic patients (17.1%) compared to non-allergic patients (8.4%).17 Pretreatment use of clindamycin or azithromycin, commonly prescribed to penicillin-allergic patients, was associated with elevated failure rates, with clindamycin use increasing the risk of failure by 2.9-fold compared to amoxicillin.
In a third retrospective analysis, it was found that the odds of failure for penicillin-allergic patients were 3.1 times higher, with a 10 times higher failure rate for immediate implant placements. Additionally, penicillin-allergic patients had a higher infection rate (3.4%) compared to non-allergic patients (0.6%) (P < .05).18
These and other studies as well as subsequent systematic reviews19,20 suggest that penicillin-allergic patients are at increased risk for implant failure and that this may be related to altered immune function associated with penicillin allergy and/or the impact of alternative antibiotics on wound healing and osseointegration of dental implants. Because a significant number of individuals who report penicillin allergy do not have a true allergy when tested, it has been suggested that confirming penicillin allergy through allergy testing, often called penicillin allergy delabeling, could improve implant outcomes by allowing the use of amoxicillin in those individuals who do not present with true allergy.
The Role of Penicillin Allergy in Implant Failure: Possible Mechanisms
To reduce the risk of early implant failure following dental implant placement in patients with a self-reported penicillin allergy, clindamycin has been used as the second-line antibiotic option over the years. Recently, dental prescribers have shifted from clindamycin to azithromycin based on recommendations from the American Dental Association, as risk–benefit analyses related to clindamycin-related pseudomembranous colitis suggest that clindamycin is risky when used as a second-line antibiotic choice.18,21 Additionally, recent concerns have focused on the negative impact of clindamycin on implant success.20
What is unknown is the potential mechanism of action that explains the link between clindamycin use and implant failure. Potential mechanisms include a lower antimicrobial efficacy and direct impacts on bone healing. According to current evidence, the underlying biological mechanisms responsible for the higher failure rates in patients with a self-reported penicillin allergy who are treated with clindamycin remain unclear. Several studies have reported increased postoperative infection rates in patients treated with clindamycin, with infection being a significant contributor to implant failure.18 Additionally, another retrospective study established a link between penicillin allergy and clindamycin use following socket grafting and ridge augmentation procedures, suggesting that clindamycin use may increase the risk of infection and act as a potential risk factor for complications in bone augmentation.22
An in vitro study revealed that several pathogenic species, such as Prevotella intermedia, Tannerella forsythia, and Aggregatibacter actinomycetemcomitans, exhibit resistance to clindamycin at therapeutic concentrations.23 Furthermore, several studies have linked the use of antibiotics other than beta-lactams in prescriptions to an increase in methicillin-resistant Staphylococcus aureus.24 Clindamycin-resistant bacteria have been found in elevated concentrations in implants affected by peri-implantitis.25 Finally, in vitro studies suggest that clindamycin may affect osteoblast viability, potentially disrupting osteointegration.26 These findings underscore the need for further research to better understand the mechanisms underlying the impact of clindamycin on implant success and to identify safer alternative treatments for patients with penicillin allergies.
Clinical Management of Penicillin-Allergic Patients Seeking Implant Treatment
To manage penicillin-allergic patients, especially those requiring dental implants, a protocol for penicillin allergy delabeling has been proposed.20 This process involves removing the penicillin allergy label, or designation, from the patient’s medical records after appropriate testing. Delabeling is particularly important, as studies show that penicillin allergies are often misreported. While approximately 10% of the US population claims to have a penicillin allergy, based on skin testing, less than 1% of individuals are actually allergic.9 Additionally, allergic reactivity may decline over time, and if a reaction to penicillin occurred more than 10 years ago, the likelihood of a true allergy is significantly reduced.27 Identifying patients with a genuine penicillin allergy can help prevent unnecessary use of alternative antibiotics that may be less effective or carry greater risks for dental implant procedures.
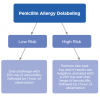
The assessment begins with a detailed allergy history, followed by classification into low- or high-risk profiles. Low-risk patients, who exhibit non-allergic symptoms such as mild rashes or gastrointestinal issues, may be delabeled after an oral challenge with a 250 mg dose of amoxicillin, followed by 1 hour of observation. If no reaction occurs, the patient can be safely delabeled. However, patients should be informed of the small possibility (2% to 3%) of a delayed rash. In cases of a severe rash or systemic symptoms, the antibiotic should be discontinued immediately. For higher-risk patients, skin testing is required, using an approved method like benzylpenicilloyl polylysine injection. A negative result allows for an oral amoxicillin challenge. Those with a history of immediate, severe reactions, such as anaphylaxis, or severe cutaneous adverse reactions, such as Stevens-Johnson syndrome or toxic epidermal necrolysis, should not undergo skin testing or challenges outside a research setting. Although these higher-risk individuals are rare, they benefit most from skin testing to confirm current tolerance to penicillin before any oral challenge.
Overall, most patients fall into the low-risk category and can be safely delabeled with a simple oral challenge. Table 1 and Figure 1 review the process of delabeling for high- and low-risk individuals.
The mechanisms of action, spectrum of microbial targets, and potential adverse reactions associated with common antibiotics used in dentistry are presented in Table 2.
Conclusion
Antibiotics are frequently used medications in dentistry. Dental healthcare professionals should be familiar with common antibiotics and aware of their mechanisms of action, microbial targets, and potential adverse reactions. Self-reported penicillin allergies have been linked to an increased risk of dental implant failure, particularly when clindamycin is used as an alternative prophylactic antibiotic. While the evidence is still limited, these findings highlight the importance of closely monitoring penicillin-allergic patients during and after dental implant procedures. Moreover, the potential for delabeling penicillin allergies in clinical practice to avoid the use of less effective antibiotics, such as clindamycin, warrants further consideration. Penicillin-allergic patients treated with clindamycin have shown up to a four-fold increased risk of implant failure, although factors like implant geometry and surface characteristics, surgical technique, and operator experience also have been associated with poor outcomes. While prophylactic antibiotics, specifically 2 g or 3 g of amoxicillin administered orally 1 hour before surgery, have proven effective in improving implant survival, reducing postoperative pain, and minimizing interference with daily activities, the benefit may be smaller and/or even inverse in individuals who cannot take penicillin drugs.
Dental healthcare professionals should use their clinical judgment and assess the risk–benefit analysis to determine if prophylactic antibiotics are warranted in a patient with a true penicillin allergy. Given that much of the current evidence linking penicillin allergy and implant failure is based on retrospective studies, further prospective research is needed to clarify the relationship between penicillin allergies, antibiotic choice, and dental implant success.
ABOUT THE AUTHORS
Efstratios Choreftakis, DDS, MS
Private Practice limited to Periodontology, Minneapolis, Minnesota
Farahnaz Fahimipour, DDS, MS, PhD
Assistant Professor, Department of Periodontology, University of Alabama at Birmingham School of Dentistry, Birmingham, Alabama; Diplomate, American Board of Periodontology
Maria L. Geisinger, DDS, MS
Professor, Acting Chair, Program Director, Advanced Education in Periodontology, Department of Periodontology, University of Alabama at Birmingham School of Dentistry, Birmingham, Alabama; Diplomate, American Board of Periodontology
Queries to the author regarding this course may be submitted to authorqueries@conexiant.com.
REFERENCES
1. Duong HY, Roccuzzo A, Stähli A, et al. Oral health–related quality of life of patients rehabilitated with fixed and removable implant-supported dental prostheses. Periodontol 2000. 2022;88(1):201-237.
2. Pjetursson BE, Asgeirsson AG, Zwahlen M, Sailer I. Improvements in implant dentistry over the last decade: comparison of survival and complication rates in older and newer publications. Int J Oral Maxillofac Implants. 2014;29 suppl:308-324.
3. Sakka S, Baroudi K, Nassani MZ. Factors associated with early and late failure of dental implants. J Investig Clin Dent. 2012;3(4):258-261.
4. Laskin DM, Dent CD, Morris HF, et al. The influence of preoperative antibiotics on success of endosseous implants at 36 months. Ann Periodontol. 2000;5(1):166-174.
5. Binahmed A, Stoykewych A, Peterson L. Single preoperative dose versus long-term prophylactic antibiotic regimens in dental implant surgery. Int J Oral Maxillofac Implants. 2005;20(1):115-117.
6. Adell R. Tissue integrated prostheses in clinical dentistry. Int Dent J. 1985;35(4):259-265.
7. Newman MG, Van Winkelhoff AJ. Antibiotic and Antimicrobial Use in Dental Practice. Chicago, IL: Quintessence Publishing; 1990:187-200.
8. Lund B, Hultin M, Tranaeus S, et al. Complex systematic review – perioperative antibiotics in conjunction with dental implant placement. Clin Oral Implants Res. 2015;26 suppl 11:1-14.
9. Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105(4):259-273.
10. Salomó-Coll O, Lozano-Carrascal N, Lázaro-Abdulkarim A, et al. Do penicillin-allergic patients present a higher rate of implant failure? Int J Oral Maxillofac Implants. 2018;33(6):1390-1395.
11. Torof E, Morrissey H, Ball PA. Antibiotic use in dental implant procedures: a systematic review and meta-analysis. Medicina (Kaunas). 2023;59(4):713.
12. Edibam NR, Prasanna R, Le Bach. Promoting responsible antibiotic usage in dental implant procedures. J Oral Maxillofac Surg. 2024;82(7):741-742.
13. Camps-Font O, Martín-Fatás P, Clé-Overjero A, et al. Postoperative infections after dental implant placement: variables associated with increased risk of failure. J Periodontol. 2018;89(10):1165-1173.
14. Schwartzenberg AV, Liu CC, Sahrmann P, et al. Risk characteristics of peri-implant infections: a retrospective evaluation in a university consultation setting. Dent J (Basel). 2022;10(9):159.
15. Esposito M, Worthington HV, Loli V, et al. Interventions for replacing missing teeth: antibiotics at dental implant placement to prevent complications [update in: Esposito M, Grusovin MG, Worthington HV. Cochrane Database Syst Rev. 2013;(7):CD004152]. Cochrane Database Syst Rev. 2010;(7):CD004152.
16. Nolan R, Kemmoona M, Polyzois I, Claffey N. The influence of prophylactic antibiotic administration on post-operative morbidity in dental implant surgery. A prospective double-blind randomized controlled clinical trial. Clin Oral Implants Res. 2014;25(2):252-259.
17. Zahra B, Nicholas B, Geoffrey R, et al. Dental implant failure rates in patients with self-reported allergy to penicillin. Clin Implant Dent Relat Res. 2022;24(3):301-306.
18. French D, Noroozi M, Shariati B, Larjava H. Clinical retrospective study of self-reported penicillin allergy on dental implant failures and infections. Quintessence Int. 2016;47(10):861-870.
19. Salgado-Peralvo AO, Peña-Cardelles JF, Kewalramani N, et al. Is penicillin allergy a risk factor for early dental implant failure? A systematic review. Antibiotics (Basel). 2021;10(10):1227.
20. Edibam NR, Lorenzo-Pouso AI, Caponio VCA. Self-reported allergy to penicillin and clindamycin administration may be risk factors for dental implant failure: a systematic review, meta-analysis and delabeling protocol. Clin Oral Implants Res. 2023;34(7):651-661.
21. American Dental Association. Antibiotic prophylaxis prior to dental procedures. ADA website. Updated December 30, 2025. Accessed January 6, 2026.
22. Basma HS, Misch CM. Extraction socket grafting and ridge augmentation failures associated with clindamycin antibiotic therapy: a retrospective study. Int J Oral Maxillofac Implants. 2021;36(1):122-125.
23. Rams TE, Balkin BE, Roberts TW, Molzan AK. Microbiological aspects of human mandibular subperiosteal dental implants. J Oral Implantol. 2013;39(6):714-722.
24. Wyles CC, Hevesi M, Osmon DR, et al. 2019 John Charnley Award: increased risk of prosthetic joint infection following primary total knee and hip arthroplasty with the use of alternative antibiotics to cefazolin: the value of allergy testing for antibiotic prophylaxis. Bone Joint J. 2019;101-B(6_supple_B):9-15.
25. Persson GR, Renvert S. Cluster of bacteria associated with peri-implantitis. Clin Implant Dent Relat Res. 2014;16(6):783-793.
26. Duewelhenke N, Krut O, Eysel P. Influence on mitochondria and cytotoxicity of different antibiotics administered in high concentrations on primary human osteoblasts and cell lines. Antimicrob Agents Chemother. 2007;51(1):54-63.
27. Centers for Disease Control and Prevention. Sexually Transmitted Infections Treatment Guidelines, 2021. Managing persons who have a history of penicillin allergy. CDC website. Reviewed September 21, 2022. https://www.cdc.gov/std/treatment-guidelines/penicillin-allergy.htm. Accessed January 6, 2026.