You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Implant related complications and failures fall into several main categories: surgical, biological, prosthetic, mechanical, esthetic, and those related augmentation procedures, placement, and loading.1 While complications concern all members of the dental team, periodontists are trained to focus on biological complications, while their restorative colleagues are most familiar with prosthetic and mechanical complications. Therefore, the focus of this discussion from a periodontal point of view will be on implant loss or failure, peri-implantitis, dehiscence, and recession, as well as implant malposition. It also will stress the importance of working together as a team to recognize and manage dental implant complications.
Classification of Complications
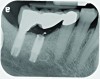
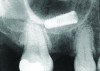
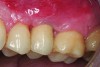
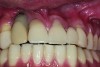
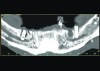
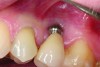
Implant related complications may be classified as minor, intermediate, major reversible/nonreversible, or major nonreversible. For example, a simple complication like a fractured abutment screw is usually considered a minor complication. It usually can be corrected with minimal cost, time, pain, and inconvenience. Treatment may be more involved for an intermediate complication such as a fractured implant (Figure 1). Up the severity scale are major complications, such as when an implant migrates into the sinus2 (Figure 2), or is exposed by mucosa and bone loss and visible to the patient a year after restoration (Figure 3 and Figure 4). Major complications may cause irreversible damage and/or require multiple procedures to try to make the patient “whole,” adding cost, time, pain, and surgeries (Figure 5 and Figure 6), and the patient may or may not receive the original restoration.
The literature describes a major nonreversible complication of numbness after implant placement due to damage to the alveolar nerve (Figure 7). Articles by Misch and Resnik,3 Alhassani and AlGhamdi,4 and most recently by Renton and Yilmaz5 discuss treatment of neurosensory complications. A case by Kan and others6 published in 1997 described another major complication of fracture of the mandible that occurred after an endosseous implant was placed in conjunction with an inferior nerve transposition.
Incidence/Prevalence of Complications
Doctors who have never experienced implant failures or complications likely either have not performed enough of them or their patients are seeing another doctor for treatment of the problems they experience. In 2010, Steve Eckert, editor-in-chief of International Journal of Oral & Maxillofacial Facial Implants, wrote: “I believe that the study and elimination of complications is the most fertile field in implant dentistry at this time.”7 The concern was also reflected in the 2010 guidelines published by the Academy of Osseointegration for those placing or restoring an implant: “In particular, the ability of a practitioner to predict, recognize, and treat complications arising from treatment is of paramount importance.”8 The latter was cited as the “legal standard.”
While many are concerned about the incidence of implant complications (that is, the number of new cases that occur during a given period of time), it is its prevalence—ie, the total number of cases of a disease that exist—that is a better reflection of the problem, mainly because complications may take many years to become apparent.
Studies by Pjertursson et al9,10 found that 38.7% of patients had complications in the first 5 years after implantation; and that implant-supported complications occurred at more than double the rate of the tooth-supported complications. After a total of 10 years in function, Lang et al11 found biological and technical complications occurred in about 50% of the cases.11 These findings suggest that one out of every two patients who receive implant restorations will eventually experience complications.
Biologic Complications: Implant Failure
A biologic complication of great concern is implant failure. Examining 14 trials over a period of 2 to 16 years, Paquette found an overall mean implant survival rate of 94.4% with a range between 76% and 98.7%, meaning that 1% to 24% of the implants failed.12 No differences were observed between the various types of implants for implant failure, but Esposito et al concluded that machined surface implants appeared to be less prone to peri-implantitis, compared to rough surface implants.13 Different surgical techniques (ie, early, immediate, or delayed) in placing dental implants do not appear to be associated with different survival rates, according to Coulthard et al.14
Perhaps most worrisome about implant failure is that most practitioners agree that replacement implants and replacement restorations rarely measure up to the esthetics achieved with the original implant restorations. Whether it is the reasons for the failure or its aftermath—loss of bone, tissue, and blood supply—research shows that the survival rate of the replacement implants drops progressively with each replacement. A study by Grossman and Levin of failed implants replaced by a similar implants in the same location reported a 71% survival rate.15 As for third attempts to place implants in sites where previous surgeries have failed, Machtei et al reported a 60% survival.16
A so-called “cluster effect”—ie, more than one implant failure per patient, which was not necessarily in the same area or quadrant—was discussed in the work of Schwartz-Arad et al,17 who reported that one-third (32.8%) of the patients in which 56.6% of all failures were found exhibited this cluster pattern in failures that occurred both in the surgical and prosthetic phases.
Causes of Failure
Signs of early failure requiring implant removal include mobility, pain, pathology, and/or radiolucency. Early (preprosthetic) implant failure has five possible primary causes: overheating of the bone, lack of initial stability, underpreparation of the osteotomy, overpreparation of the osteotomy, or contamination of the implant surface. Late (post-loading) failure is caused primarily by implant overload, parafunction issues, peri-implantitis, and implant malposition. It follows that strategies for avoiding these complications involve avoidance of the foreseeable problems, such as by limiting micromotion to <50 um, observing surgical guidelines related to site preparation and hygiene, taking care to keep the drill and bone temperature cool during implant insertion, avoiding overload—especially in poor quality bone and with early loading protocols—and considering issues of parafunction/temporization.
Post-loading Failure
The four categories of post-loading failure include: occlusal overload, dehiscence and recession, implant malposition, and peri-implantitis.
Occlusal Overload
The occlusal overload topic remains controversial. In 2006, Isidor18 reported an association between the loading conditions and marginal bone loss around implants, but a causal relationship has not been shown. However, it seems obvious to this author that overload means a case is under-engineered and does not offer sufficient support for the implant in function, particularly in cases where parafunctional habits (ie, bruxing, clenching) exist.
Dehiscence, Recession, Poor Implant Position
The biological problems of dehiscence and recession usually occur because of poor implant position. Poor implant positioning is not just an esthetic problem, it can damage adjacent teeth or vital structures. Its causes are generally due to poor planning and poor surgical technique. Prevention of this complication requires proper collaborative surgical/prosthetic planning—ie, mounted study models; surgical guides prepared from ideal wax-ups; use of properly designed implants (tapered, short, narrow diameter)—and proper surgical and restorative experience and execution.
The implant of the patient shown in Figure 8 demonstrated all three of the red flag situations: it was placed too far buccally, had inadequate bone and soft tissue at the time of implant placement, and lost bone and soft tissue post implant placement. Correcting this required increased time, cost, and surgeries (Figure 9).
Prevention of this type of complication requires collaboration between the periodontist and the restorative dentist in the planning phase, and proper collaboration in the surgical placement, and in the restoration of the implants. No one member of the dental team can do it alone.
Peri-implantitis
The topic of peri-implantitis is often fraught with confusion, mainly due the definitions and overlapping symptoms it shares with peri-implant mucositis.19 According to the First European Workshop on Periodontology—1993, peri-implant mucositis is a term used to describe a reversible inflammatory reaction in the mucosa adjacent to an implant, whereas the definition of peri-implantitis is an inflammatory process that affects the tissues around an osseointegrated implant in function and results in loss of supporting bone.20 In 2008, at the Sixth European Workshop on Periodontology, Lindhe and Meyle revised these definitions to determine that a diagnosis of peri-implant mucositis should be based on the parameters of bleeding upon probing without loss of supporting bone, while the diagnosis of peri-implantitis should include both bleeding upon probing and detectable bone loss after 1 year in function.21
Ultimately, the consequences of peri-implantitis, as defined above—with both inflammation in the soft tissue and bone loss—are far more serious. Moreover, if the bone loss is not treated, the implant will fail. This was discussed in 2012 by Tomasi and Derks, who used the revised definition by Lindhe and Meyle.21,22
David Cochran, DDS, has referred to peri-implant mucositis and peri-implantitis as “convenient diseases” while questioning whether they are diseases at all. He wrote, “While these might be catchy and convenient terms, their use and implication are fraught with huge assumptions. Are these really diseases, and if so, why aren’t their definitions so obvious? They certainly imply that they are similar to gingivitis and periodontitis but is this misleading and or convenient. Why can’t we simply and more accurately call it like we see it? The fact is, we are observing conditions where there is peri-implant inflammation with and without bone loss.”23
In support of Cochran’s observations, dog studies by Lindhe et al in 1992,24 Marinello et al in 1995,25 Ericsson et al in 1996, 26 and Persson et al in 199627 found that peri-implantitis may not develop in all peri-implant sites with mucositis, just as periodontitis may not develop in all sites with gingivitis. As demonstrated by the lesions studied by Lindhe et al24 in plaque-associated peri-implant tissues in dogs, if allowed to progress, the lesions lead to loss of the implant.
Therefore, for purposes of simplification, and in line with previous published literature, the author will refer to peri-implant inflammation with bone loss (PII w/BL) as peri-implantitis and peri-implant inflammation without bone loss (PII w/o BL) as mucositis.
The proven principle behind mucositis (PII w/o BL)—that is, the existence of a cause-and-effect relationship between bacterial plaque and developing mucositis—was demonstrated for oral implants in separate studies by Pontoriero et al28 and Salvi et al,29 who had patients refrain from oral hygiene for a period of 3 weeks, after which time both the teeth and the implants developed gingivitis and mucositis. That the condition is reversible was also demonstrated in those same studies in which the tissues were returned to health after regular oral hygiene was resumed.28,29
Prevalence of peri-implant mucositis (which is reversible through removal of the biofilm and accretions) ranges from 33% to 66%, according to several studies, cited in the work of Tomasi and Derks,22 which also reported the prevalence of peri-implantitis to range from 8.9% to 47% of the subjects studied.
Koldsland et al30 suggested that a clearer picture of the prevalence of peri-implantitis can be gained by establishing a threshold for bone loss. While defining peri-implantitis using the Albrektsson criteria31—that is, greater than 0.2 mm bone loss per year after the first year—places the prevalence of patients who will develop peri-implantitis at 47.1%. Changing the definitive amount of bone loss to 3 mm or more after the first year reduces the prevalence to 11.3%.30
Given the amount of confusion that surrounds definitions of these oral conditions and confounds useful characterization of their prevalence, the author and Paul Rosen, in 2012,32 sought to establish a standard peri-implantitis classification system differentiating early, moderate, and severe disease based on bone loss. In all cases, these classifications assume pocket depths of 4 mm or greater and bleeding on probing or suppuration. When bone loss as shown on x-rays is present up to 25% of implant length, the disease is classified as early peri-implantitis; 25% to 50% is moderate peri-implantitis; and greater than 50% of implant length is classified as severe/advanced peri-implantitis. These delineations are not only useful for determining prevalence, they also have implications for treatment, which should be rendered without delay when caused by bacterial contamination combined with a compromised host response. An approach involving regenerative therapy that is described by the author, Dr. Stuart Froum, Dr. Scott Froum, and Dr. Paul Rosen, seeks to resolve the inflammation; promote soft tissue and bone formation; and promote re-osseointegration.33
The Importance of Good Hygiene
Among the seven risk factors for peri-implantitis identified by the Sixth European Workshop, the one deemed most important by the author is poor oral hygiene. Noting that both peri-implant mucositis and peri-implantitis are caused by bacteria, according to two reports that studied disease prevalence, peri-implant mucositis occurred in about 80% of subjects (50% of sites) restored with implants, and peri-implantitis in 28% and 56% of subjects (corresponding to 12% and 40% of sites).21
Because oral hygiene is so important, treatment planning should take into consideration restorative design that can impact hygiene. A case controlled study by Serino and Strom found that 48% of implants that had no accessibility or capability for proper oral hygiene were affected by peri-implantitis, compared to only 4% with accessibility.34
It is essential to consider hygiene both in terms of in office professional care and at-home preventive maintenance. A 5-year follow-up study by Costa et al found that without preventive maintenance, patients with pre-existing peri-implant mucositis had a 44% higher incidence of peri-implantitis.35
From these studies, it can be concluded that to maximize oral hygiene and thereby minimize the risk of developing peri-implant mucositis and peri-implantitis, patients must be able to clean the implant-supported restorations placed and these patients should be urged to comply with recommended recall visits during which hygiene and peri-implant health can be professionally maintained and monitored.
Conclusion
The growing prevalence of dental implant complications stands to create a crisis of confidence that could set back the significant strides made in the field of implantology and hinder patients’ access to this treatment option. Dentists who lose confidence in the implant option could make the mistake of returning to limiting restorations to teeth rather than utilizing an implant option; patients who lose confidence could turn to lawyers, rather than doctors; and third-party control of treatment plans that include implant restorations could result in denial of implant options that may be considered risky and expensive by non-dentists or dentists with limited implant experience.
To prevent this from happening, dentists should make every effort to prevent complications from occurring. This requires knowledge of the causes of complications together with proper collaboration between the periodontist and the restorative dentist, not only on planning, but also in execution of proper treatment. Dentists should adhere to complete planning from top down utilizing surgical and radiographic guides, wax-ups, and scans. They should ensure proper implant spacing, with adequate embrasures. They should prevent and/or treat peri-implant mucositis by removing biofilm and accretions and reinforcing proper hygiene procedures. Finally, to prevent complications from becoming more complicated, they should know what to do when things go wrong. This includes removing excess cement (with lidocaine); when necessary redoing the prosthesis with adequate embrasures; avoiding splints when feasible; re-preparing abutments with adequate room for maintenance between them; and redoing ill-fitting implant-supported restorations. Most importantly, working together to optimize implant placement and restoration helps achieve successful results while minimizing potential complications. When complications do occur, their resolution is again best accomplished by collaborative treatment between the periodontist and restorative dentist.
REFERENCES
1. Froum SJ, Klokkevold PR, Sang Choon Cho, Froum SH. Implant related complications and failures. In: Newman MG, Takei H, Klokkevold PR, Carranza FA, eds. Carranza’s Clinical Periodontology. 11th ed. St. Louis, Mo; Saunders: 2012.
2. Galindo-Moreno P, Padial-Molina M, Sánchez-Fernández E, et al. Dental implant migration in grafted maxillary sinus. Implant Dent. 2011;20(6):400-405.
3. Misch CE, Resnik R. Mandibular nerve neurosensory impairment after dental implant surgery: management and protocol. Implant Dent. 2010;19(5);378-386.
4. Alhassani AA, AlGhamdi AS. Inferior alveolar nerve injury in implant dentistry: diagnosis, causes, prevention, and management. J Oral Implantol. 2010;36(5):401-407.
5. Renton T, Yilmaz Z. Managing iatrogenic trigeminal nerve injury: a case series and review of the literature. Int J Oral Maxillofac Surg. 2012;41(5):629-637.
6. Kan JY, Lozada JL, Boyne PJ, et al. Mandibular fracture after endosseous implant placement in conjunction with inferior alveolar nerve transposition: a patient treatment report. Int J Oral Maxillofac Implants. 1997;12(5):655-659.
7. Eckert SE. The greatest complication of all. Int J Oral Maxillofac Implants. 2010;(25)1. http://jomi.quintessenz.de/jomi_2010_01_s0017.pdf. Accessed June 23, 2013.
8. Academy of Osseointegration. 2010 Guidelines of the Academy of Osseointegration for the provision of dental implants and associated patient care. Int J Oral Maxillofac Implants. 2010;25(3):620-627.
9. Pjetursson BE, Tan K, Lang NP, et al. A systematic review of the survival and complication rates of fixed partial dentures (FPDs) after an observation period of at least 5 years. Clin Oral Implants Res. 2004;15(6):625-642.
10. Pjetursson BE, Brägger U, Lang NP, Zwahlen M. Comparison of survival and complication rates of tooth-supported fixed dental prostheses (FDPs) and implant-supported FDPs and single crowns (SCs). Clin Oral Implants Res. 2007;18(suppl 3):97-113.
11. Lang NP, Pjetursson BE, Tan K, et al. A systematic review of the survival and complication rates of fixed partial dentures (FPDs) after an observation period of at least 5 years. II. Combined tooth—implant-supported FPDs. Clin Oral Implants Res. 2004;15(6):643-53.
12. Paquette DW, Brodala N, Williams RC. Risk factors for endosseous dental implant failure. Dent Clin North Am. 2006;50(3):361-374.
13. Esposito M, Coulthard P, Thomsen P, Worthington HV. Interventions for replacing missing teeth: different types of dental implants. Cochrane Database Syst Rev. 2005;(1):CD003815.
14. Coulthard P, Esposito M, Jokstad A, Worthington HV. Interventions for replacing missing teeth: surgical techniques for placing dental implants. Cochrane Database Syst Rev. 2003;(1):CD003606.
15. Grossmann Y, Levin L. Success and survival of single dental implants placed in sites of previously failed implants. J Periodontol. 2007;78(9):1670-1674.
16. Machtei EE, Horwitz J, Mahler D, et al. Third attempt to place implants in sites where previous surgeries have failed. J Clin Periodontol. 2011;38(2):195-198.
17. Schwartz-Arad D, Laviv A, Levin L. Failure causes, timing, and cluster behavior: an 8-year study of dental implants. Implant Dent. 2008;17(2):200-207.
18. Isidor F. Influence of forces on peri-implant bone. Clin Oral Implants Res. 2006;17(suppl 2):8-18.
19. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. J Periodontol. 2013;84
(4):436-443.
20. Lang NP, Karring T. Proceedings of the 1st European Workshop on Periodontology. London; Quintessence Publishing: 1994:365-369.
21. Lindhe J, Meyle J, Group D of European Workshop on Periodontology. Peri-implant diseases: Consensus Report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008;35(suppl 8):282-285.
22. Tomasi C, Derks J. Clinical research of peri-implant diseases—quality of reporting, case definitions and methods to study incidence, prevalence and risk factors of peri-implant diseases. J Clin Periodontol. 2012;39(suppl 12):207-223.
23. Cochran DL. Tampa and convenient diseases. Acad Osseointegrat N. 2012;23(4):1, 13.
24. Lindhe J, Berglundh T, Ericsson I, et al. Experimental breakdown of peri-implant and periodontal tissues. A study in the beagle dog. Clin Oral Implants Res. 1992;3(1):9-16.
25. Marinello CP, Berglundh T, Ericsson I, et al. Resolution of ligature-induced peri-implantitis lesions in the dog. J Clin Periodontol. 1995;22(6):475-479.
26. Ericsson I, Persson LG, Berglundh T, et al. The effect of antimicrobial therapy on periimplantitis lesions. An experimental study in the dog. Clin Oral Implants Res. 1996;7(4):320-328.
27. Persson LG, Ericsson I, Berglundh T, et al. Guided bone regeneration in the treatment of periimplantitis. Clin Oral Implants Res. 1996;7(4):366-372.
28. Pontoriero R, Tonelli MP, Carnevale G, et al. Experimentally induced peri-implant mucositis. A clinical study in humans. Clin Oral Implants Res. 1994;5(4):254-259.
29. Salvi GE, Aglietta M, Eick S, et al. Reversibility of experimental peri-implant mucositis compared with experimental gingivitis in humans. Clin Oral Implants Res. 2012;23(2):182-190.
30. Koldsland OC, Scheie AA, Aass AM. Prevalence of peri-implantitis related to severity of the disease with different degrees of bone loss. J Periodontol. 2010;81(2):231-238.
31. Albrektsson T, Zarb G, Worthington P, Eriksson AR. The long-term efficacy of currently used dental implants: a review and proposed criteria of success. Int J Oral Maxillofac Implants. 1986;1(1):11-25.
32. Froum SJ, Rosen PS. A proposed classification for peri-implantitis. Int J Periodontics Restorative Dent. 2012;32(5):533-540.
33. Froum SJ, Froum SH, Rosen PS. Successful management of peri-implantitis with a regenerative approach: a consecutive series of 51 treated implants with 3- to 7.5-year follow-up. Int J Periodontics Restorative Dent. 2012;32(1):11-20.
34. Serino G, Ström C. Peri-implantitis in partially edentulous patients: association with inadequate plaque control. Clin Oral Implants Res. 2009;20(2):169-174.
35. Costa FO, Takenaka-Martinez S, Cota LO, et al. Peri-implant disease in subjects with and without preventive maintenance: a 5-year follow-up. J Clin Periodontol. 2012;39(2):173-181.
ABOUT THE AUTHOR
Stuart J. Froum, DDS
Director of Clinical Research, Clinical Professor, Department of Periodontology and Implant Dentistry,
New York University College of Dentistry,
New York, New York;
Private Practice,
New York, New York
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com