You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
In the realm of "bioactive" materials, most clinicians are familiar with liners that have the ability to induce the formation of reparative dentin for pulp capping. A less understood and less frequently used class consists of bioactive materials for direct restorations. A recent American Dental Association (ADA) Clinical Evaluators Panel Report indicated that 56% of dentists use bioactive liners daily/weekly, whereas only 15% of dentists use bioactive restoratives daily/weekly.1 Part of the hesitance for clinicians to use bioactive restoratives may be the lack of knowledge of the potential benefits. Furthermore, scientific evidence to support these potential benefits should be provided to convince the clinician to adopt this class of materials. This review will focus on potential benefits of materials used for direct restorations with claims of bioactivity.
Definition of Bioactive Material
In July 2018, a group of 50 key opinion leaders met to develop a consensus statement on the definition of a bioactive restorative dental material.2 From that consensus meeting, "bioactivity" describes "an active beneficial biological process." A bioactive restorative material would restore missing tooth structure and additionally "stimulate or direct specific cellular or tissue responses, or both, or they can control interactions with microbiological species." Several examples of bioactive effects were mentioned, including the following:
• Components that dissolve that can be identified with
normal physiological species that are involved in a biologi-
cal process
• A surface that may nucleate the formation of biological-like
calcium phosphates, including bioapatite-like material, when
in contact with saliva or tissue fluids
• Components that dissolve and thereby cause local precipi-
tation of biological-like calcium phosphates, including bio-
apatite-like materials, in a purely passive chemical process
• Components that dissolve and happen to have antimicrobial
activity
Several of these proposed bioactive effects correspond with the claims of commercially available bioactive materials marketed for use as direct filling materials. First, several restorative materials have claimed to release fluoride, calcium, or phosphate, which may be used to help remineralize tooth structure at the margins of a restoration. Second, several restorative materials have been shown to precipitate hydroxyapatite on their surface for the purpose of sealing the interface between the tooth and the restorative material. Finally, some new materials aim to prevent biofilm attachment by either changing the local pH or producing an electric charge on the surface of the material.
Ion Releasing
The mineral content of enamel and dentin is hydroxyapatite. This mineral is composed of 5 parts calcium, 3 parts phosphate, and 1 part hydroxyl group. Enamel may also be mineralized with fluorapatite, in which the hydroxyl group is substituted for a fluoride ion.3During the process of recurrent caries, hydroxyapatite is removed from the surface of the tooth surrounding a dental restoration. A restoration that can release the ions present in the mineral content of the tooth would, in theory, be able to assist the process of remineralization in the tooth structure surrounding the restoration.3
Glass-ionomer materials, developed in the 1960s, have been reported to reduce the incidence of recurrent caries. A 2011 systematic review and meta-analysis summarized the data from six clinical trials and compared the presence of carious lesions around glass-ionomer and resin composite restorations within 24 data sets.4 Seventeen of the data sets showed no difference in the incidence of caries between glass-ionomers and resin composites. In seven of the data sets, there was a lower incidence of secondary caries around the restorations restored with glass-ionomer materials.
The evidence for the preventive effect of glass-ionomer materials has been credited to the ability of the material to prevent demineralization in the tooth structure surrounding the restoration. In a study by Donly et al,5 primary teeth were restored with either amalgam or glass-ionomer restorations. The exfoliated teeth were then collected on average 26 months after placement. The teeth were sectioned and viewed with polarized light microscopy. The sections revealed that there was less demineralization on the tooth structure adjacent to glass-ionomer restorations than amalgam restorations.
Fluoride release has been credited with preventing demineralization with glass-ionomer materials; consequently, there have been several iterations of fluoride-releasing materials.6 The basic composition of a glass-ionomer is a polyacrylic acid monomer and aluminofluorsilicate glass filler.6 Resin-modified glass-ionomer (RMGI) materials are a modification of the formulation of glass-ionomer to include light-polymerizable additions to the polyacrylic acid monomers.6 Another fluoride-releasing class of material is compomer. Compomers are composed of methacrylate monomer (similar to resin composites) modified with the addition of polyacrylic acid and mixed with fluoride-releasing filler particles.7Compomers are similar in composition to resin composites, so they have better mechanical and esthetic properties than glass-ionomers.7 Another distinction of compomer materials is that their hydrophilic components allow water uptake and fluoride release from the material.7
Although compomers allow fluoride release, they do not have the same fluoride release properties as glass-ionomers and RMGI materials. A recent study reported that several compomer materials released 2 to 12 times less fluoride than a reference RMGI material within the first 5 days after placement in water.8After 6 days, however, the fluoride release from the RMGI began to diminish. By 10 days, the fluoride release from the RMGI material was nearly equivalent to the compomer materials. Perhaps more important than the fluoride release of restorative materials is their ability to recharge fluoride. In another study, a compomer material was recharged with fluoride from a 2% neutral sodium fluoride gel.9The compomer was able to rerelease fluoride up to 30 days after the recharge; however, the amount of fluoride released after recharging was significantly less than that released by the RMGI reference material after recharging.
Because calcium is a component of the mineral content in tooth structure, several experimental dental restorative materials have been produced with calcium release. Calcium fluoride nanoparticles have been used to reinforce experimental composite resins. These materials have shown high fluoride release and good mechanical properties; however, their opacity has limited their use as dental restoratives.10 Calcium phosphate (CaP) particles have also been used as filler particles for experimental resin composites. The initial formulations of CaP-filled composites contained filler particles from 1 to 55 µm in size. These CaP composites showed release of calcium and phosphate ions and the ability to cause in vitro tooth remineralization; however, the mechanical properties of these composites were poor.3,11,12 More recently, nanoparticles of CaP have been developed with sizes around 100 nm. Incorporation of nano-sized CaP into a dental composite allowed improvement in the mechanical properties of the material so that it was reported to be equivalent to a reference composite.3,13 The nano-sized CaP composite was able to promote tooth remineralization in both in vitro and in vivo studies.14,15
Several calcium-releasing restorative materials have been made commercially available. Some of the bioactive restorative materials currently on the market, such as Predicta™ Bioactive Bulk (Parkell, parkell.com) and Activa™ BioActive-Restorative™ (Pulpdent, Pulpdent.com), could be considered modified compomers because their polymer matrix is predominantly composed of a dual-cured resin methacrylate, with polyacid monomers added to at least one of them (Activa BioActive-Restorative).16 The mechanical properties of Activa BioActive-Restorative have been shown to be similar to a reference resin composite.17,18 Another commercially available calcium-releasing restorative material, Cention N® (Ivoclar Vivadent, ivoclarvivadent.com), contains methacrylate monomers as well as calcium fluorosilicate glass fillers.19 All these materials have been shown to release some calcium and fluoride.17,20 Additionally, Cention N has been shown to inhibit demineralization at restoration margins in vitro.21
Hydroxyapatite Precipitation
Another claim of some bioactive restorative materials is the ability to form hydroxyapatite precipitates on their surface. Predicta Bioactive Bulk, Activa BioActive-Restorative, and Cention N have all been shown to grow a layer of hydroxyapatite on their surface.19,20,22An important question is, what is the clinical applicability of this hydroxyapatite growth? A calcium aluminate material, Doxadent (Doxa), had previously been released with claims of bioactivity. In an in vitro experiment, the interface between the calcium aluminate material was shown to closely adapt to tooth structure with high magnification scanning electron microscopy. The close adaption of the restorative material to the tooth was speculated to occur due to deposition of an apatite precipitate.23 In another experiment, premolars were restored with Doxadent, extracted for orthodontic reasons, and evaluated for adaption between tooth and restorative material. The calcium aluminate material showed better adaption to dentin than a reference composite but worse adaptation to enamel.24 Ultimately, Doxadent did not succeed as a restorative material due to the high incidence of material and tooth fracture.25
Due to the claims of hydroxyapatite formation, some bioactive restorative materials have been evaluated for use without a bonding agent. A 1-year clinical trial of Activa BioActive-Restorative used without a bonding agent showed an unacceptable clinical failure rate (24.1%).26A 2-year clinical trial reported similar clinical survival rates of Cention N used with and without an adhesive; however, less marginal staining was observed when an adhesive was used.27 The use of a bonding agent has been shown to significantly reduce the amount of fluoride released from compomer materials; however, the bonding agent did not completely prevent the release of fluoride.28Therefore, the use of a bonding agent with Activa BioActive-Restorative and Predicta Bioactive Bulk is prudent.
New Concepts in Bioactive Restorative Materials
New concepts in bioactive restorative materials are emerging. Cention N is an alkasite material, implying that it is able to raise the local pH. Higher pH can be beneficial for remineralizing tooth structure and possibly bacteriostatic.3 Some novel composites have been modified with the addition of quaternary ammonium compounds (QAC). QACs produce an electric charge that may be used to repel bacterial attachment.29 Finally, new materials have been fabricated with poly(amido amine) (PAMAM) additions, which are polymers that can serve as nucleation sites for hydroxyapatite.3PAMAMs bind to collagen in demineralized tooth structure and attract calcium and phosphate ions.
Clinical Application
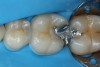
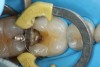
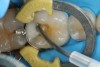
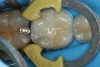
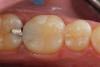
Predicta Bioactive Bulk restorative material was used to restore a fractured Class II amalgam restoration in a mandibular first molar (Figure 1). A selective-etch technique was used in which phosphoric acid was applied only to the enamel. A universal adhesive was applied to the tooth and light polymerized (Figure 2). Predicta Bioactive Bulk is a dual-cure bulk-fill material. The material was syringed into the preparation starting in the depth of the Class II box (Figure 3). A waiting period of 1 minute followed in order to allow the material to begin chemical polymerization within the depth of the Class II box (Figure 4). The theory for waiting before light polymerization is to allow the material to polymerize toward the area in which the material was first injected (the floor of the box). The material was then light polymerized, occlusion was adjusted, and the restoration was polished (Figure 5).
Conclusions
The new class of bioactive materials differentiates itself by the ability to release calcium and precipitate hydroxyapatite on its surface. The release of calcium and fluoride from these materials may help prevent the incidence of recurrent caries. Precipitation of hydroxyapatite on the surface of these materials may occur; however, a bonding agent should still be used. The potential clinical benefits of these materials require further evidence to substantiate the claims.
ABOUT THE AUTHOR
Nathaniel Lawson, DMD, PhD
Assistant Professor, Department of Clinical and Community Sciences, Division of Biomaterials, University of Alabama at Birmingham School of Dentistry, Birmingham, Alabama
References
1. ACE panel report: bioactive materials. American Dental Association. https://www.ada.org/~/media/ADA/Science%20and%20Research/Files/ACE_Panel_Report_Bioactive_Materials_Q2_2018.pdf?la=en. Published 2018. Accessed May 14, 2019.
2. Price RB, Roulet JF. The value of consensus conferences: peer review by 50 key opinion leaders! Stoma Edu J. 2018;5(4):202-204.
3. Liang K, Wang S, Tao S, et al. Dental remineralization via poly(amido amine) and restorative materials containing calcium phosphate nanoparticles. Int J Oral Sci. 2019;11(2):15.
4. Yengopal V, Mickenautsch S. Caries-preventive effect of resin-modified glass-ionomer cement (RM-GIC) versus composite resin: a quantitative systematic review. Eur Arch Paediatr Dent.2011;12(1):5-14.
5. Donly KJ, Segura A, Kanellis M, Erickson RL. Clinical performance and caries inhibition of resin-modified glass ionomer cement and amalgam restorations. J Am Dent Assoc.1999;130(10):1459-1466.
6. Baig MS, Fleming GJ. Conventional glass-ionomer materials: a review of the developments in glass powder, polyacid liquid and the strategies of reinforcement. J Dent. 2015;43(8):897-912.
7. Nicholson JW. Polyacid-modified composite resins ("compomers") and their use in clinical dentistry. Dent Mater. 2007;23(5):615-622.
8. Garoushi S, Vallittu PK, Lassila L. Characterization of fluoride releasing restorative dental materials. Dent Mater J. 2018;37(2):293-300.
9. May E, Donly KJ. Fluoride release and re-release from a bioactive restorative material. Am J Dent. 2017;30(6):305-308.
10. Xu HH, Weir MD, Sun L, et al. Strong nanocomposites with Ca, PO4, and F release for caries inhibition. J Dent Res.2010;89(1):19-28.
11. Skrtic D, Antonucci JM, Eanes ED, et al. Physicochemical evaluation of bioactive polymeric composites based on hybrid amorphous calcium phosphates. J Biomed Mater Res.2000;53(4):381-391.
12. Langhorst S, O'Donnell J, Skrtic D. In vitro remineralization of enamel by polymeric amorphous calcium phosphate composite: quantitative microradiographic study. Dent Mater. 2009;25(7):884-891.
13. Xu HH, Moreau JL, Sun L, Chow LC. Nanocomposite containing amorphous calcium phosphate nanoparticles for caries inhibition. Dent Mater.2011;27(8):762-769.
14. Weir MD, Chow LC, Xu HH. Remineralization of demineralized enamel via calcium phosphate nanocomposite. J Dent Res. 2012;91(10):979-984.
15. Melo MA, Weir MD, Rodrigues LK, Xu HH. Novel calcium phosphate nanocomposite with caries-inhibition in a human in situ model. Dent Mater. 2013;29(2):231-240.
16. RMGI Low Viscosity 510(k) premarket notification 2012. US Food and Drug Administration. https://www.accessdata.fda.gov/cdrh_docs/pdf12/K123265.pdf. Accessed July 31, 2019.
17. Bansal R, Burgess J, Lawson NC. Wear of an enhanced resin-modified glass-ionomer restorative material. Am J Dent.2016;29(3):171-174.
18. Kulkarni P, Farheen F, Lawson NC, Burgess JO. Ion release and flexural strength of bioactive materials. J Dent Res. 2018;97(spec iss A):1160.
19. Cention N. Scientific documentation. Ivoclar Vivadent. https://www.ivoclarvivadent.in/p/all/cention-n. Accessed July 31, 2019.
20. Predicta Bioactive Bulk. Scientific documentation. Parkell.
21. Donly KJ, Liu JA. Dentin and enamel demineralization inhibition at restoration margins of Vitremer, Z 100 and Cention N. Am J Dent.2018;31(3):166-168.
22. Chao W, Perry R, Kugel G. Surface deposition analysis of bioactive restorative material and cement. J Dent Res. 2016;95(spec iss A):S1313.
23. Engqvist H, Schultz-Walz JE, Loof J, et al. Chemical and biological integration of a mouldable bioactive ceramic material capable of forming apatite in vivo in teeth. Biomaterials. 2004;25(14):2781-2787.
24. Sunnegårdh-Grönberg K, van Dijken JW, Lindberg A, Hörstedt P. Interfacial adaptation of a calcium aluminate cement used in class II cavities, in vivo. Clin Oral Investig. 2004;8(2):75-80.
25. Van Dijken JW, Sunnegårdh-Grönberg K. A three year follow-up of posterior Doxadent restorations. Swed Dent J.2005;29(2):45-51.
26. van Dijken JWV, Pallesen U, Benetti A. A randomized controlled evaluation of posterior resin restorations of an altered resin modified glass-ionomer cement with claimed bioactivity. Dent Mater. 2019;35(2):335-343.
27. Robles A, Fu C, Lawson NC, Burgess JO. Clinical evaluation of an amalgam replacement restorative material: 24-month recall. J Dent Res. 2019;98(spec iss A):736.
28. Vercruysse CW, De Maeyer EA, Verbeeck RM. Fluoride release of polyacid-modified composite resins with and without bonding agents. Dent Mater. 2001;17(4):354-358.
29. Cheng L, Zhang K, Zhang N, et al. Developing a new generation of antimicrobial and bioactive dental resins. J Dent Res. 2017;96(8):855-863.