You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
Marketed as Diprivan® by AstraZeneca (www.astrazeneca.com), propofol is a commonly used general anesthetic induction agent.1 This short-acting intravenous medication is labeled by the Food and Drug Administration (FDA) as a prescription drug for human and veterinary medicine.1 The US Drug Enforcement Agency (DEA) has not currently classified propofol as a controlled substance.1 However, because of the increasing rate of propofol abuse among healthcare professionals and the 2009 death of entertainer Michael Jackson, who was given the drug to treat his insomnia, the DEA considered changing the classification to DEA Schedule IV, which is the classification for the abuse potential of benzodiazepines.1 Only with Jackson’s death did the general public and many healthcare professionals become aware that propofol is a potential drug for abuse that carries an extremely narrow margin for error when in the wrong hands. Conversely, in the right hands, propofol can be a valuable tool to achieve deep sedation or general anesthesia and provides rapid onset and recovery. However, if administered by individuals not trained in anesthesia or by operator–sedationists who do not closely monitor vital signs and respiratory output, the drug’s use can lead to death.
An article in a leading anesthesiology journal stated that “propofol abuse has become a growing problem in anesthesiology.”2 Propofol’s abuse potential had been previously suggested in animal studies, which have demonstrated elevated endogenous cannabinoid anandamide levels (an endogenous marijuana-like compound), as well as effects on the mesocorticolimbic reward system.2,3 The mesocorticolimbic dopaminergic reward circuit in the brain displays increased activity following intake of drugs with abuse potential.2,3 Both subanesthetic and anesthetic doses of propofol increase dopamine concentrations, which reflect activation of the reward circuit.2,3 After receiving subanesthetic doses of propofol, individuals describe feeling high, sedated, coasting, “spaced out,” drunk, pleasant, euphoric, or relaxed.3-5
Propofol presents a definite risk for developing psychological dependence; however, definitive signs of physical dependence usually exhibited as a withdrawal syndrome on abrupt discontinuation of the drug have not been well characterized.3-5 Abusers report cravings and repeatedly self-injecting; this psychological dependence has only been observed in abusers but not in patients who are legitimately administered the drug for acute situations (general anesthetic induction or a one-time sedation procedure).3 Limited data regarding tolerance and withdrawal in propofol abusers are available.3-5
A survey by Wischmeyer et al2 assessing the rate of propofol abuse in academic anesthesia programs reported that propofol abuse is on the rise. In the last 30 years, a fivefold increase in the rate of propofol abuse has been reported, whereas the rate of opioid abuse has remained relatively constant.2 Although opioid abuse is still more common and is a growing problem in younger individuals with FDA-approved pain medications containing hydrocodone and oxycodone, the rising abuse rate for propofol is concerning because of its increased risk for fatality.2,6 In fact, findings from a survey of anesthesiology training programs showed that 18% of these programs reported propofol abuse.2 Although death was not the final outcome for all individuals caught abusing propofol, there was no prior evidence of abuse for those who did die until they were found dead with empty propofol vials and syringes nearby.2 In residents who were self-administering propofol, a 38% mortality rate was observed.2 The greatest risk for complications related to drug addiction occurred in individuals less than 5 years out from medical school.2 The retrospective report found that no attending anesthesiologist abusing the drug had died.2 However, the DEA has found a 28% mortality rate among anesthesiologists abusing the drug.1
In one case report, a 38-year-old female anesthesiologist was found dead in a locked room. The postmortem analysis showed that she had self-administered therapeutic levels of the drug, albeit for illegal purposes; it was not an overdose per se. This has been a recurrent theme in other reports of propofol-related fatalities.3,4 In the absence of medically supervised administration and airway intervention, the unmitigated effects of respiratory depression and apnea can lead to death.3,4
The remainder of this article will focus on the legitimate use of propofol as a deep sedation and general anesthetic agent, with special emphasis on pharmacology and toxicology.
Levels of Sedation
The Joint Commission’s Comprehensive Accreditation Manual for Ambulatory Care has defined four levels of sedation and anesthesia: minimal sedation (anxiolysis); moderate sedation (formerly known as conscious sedation); deep sedation; and general anesthesia.7 Table 1 illustrates that as a patient progresses from minimal to deeper levels of sedation, central nervous system and cardiovascular depression becomes more pronounced, and the clinician’s ability to maintain a patent airway becomes more impaired. The addition of therapeutic doses of propofol to conventional conscious sedation regimens (eg, fentanyl plus midazolam titrated to effect) will invariably lead to at least a deep level of sedation.
Clinical Pharmacology of Propofol
Propofol (2,6-diisopropylphenol) is a lipid-soluble alkylphenol compound8 consisting of a phenol ring with two isopropyl groups (Figure 1).9 Its mechanism of action is thought to involve an interaction with specific structures on the postsynaptic membrane, which results in a decreased dissociation of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) from the GABA receptor located in the central nervous system.8 In addition to potentiating the actions of GABA on the GABA receptor, propofol directly opens the chloride channel independent of GABA, which is an action similar to that of barbituric acids such as methohexital (Brevital®, JHP Pharmaceuticals, www.jhppharma.com) and could partially explain its much steeper dose response curve than benzodiazepines.10 No reversal agent is available for propofol.8,9
The pharmacokinetic profile makes propofol an attractive drug for outpatient general anesthetic and deep sedation cases. Onset is rapid due to the drug’s high lipophilicty.9 Propofol is metabolized by the liver where it is conjugated to inactive, water-soluble glucuronic acid and sulphate metabolites.8 Extrahepatic metabolism via the lungs has been implied due to the clearance of the drug exceeding hepatic blood flow.8,9 The metabolites are primarily excreted renally.9 Moderate cirrhosis and chronic renal failure do not significantly alter the pharmacokinetic profile of the drug.9 For sedation, drug doses can be titrated to effect in patients as well, because the drug is delivered intravenously. A propofol infusion can maintain the desired level of sedation or anesthesia during a surgical procedure or to keep the patient adequately sedated to remain intubated in an intensive care unit. In order for a patient to be awakened from anesthesia, the plasma propofol concentration needs to be reduced by half.8 The initial distribution half life, or clearance from the central component, is rapid and may be only 2 to 8 minutes for a single bolus dose.8,9 This explains the relatively rapid recovery from the drug. The context-sensitive half life following an infusion less than 8 hours in duration is still less than 40 minutes.8
While propofol has an attractive pharmacokinetic profile of rapid onset and offset, the pharmacodynamic profile, or its effects on the central nervous system and cardiovascular systems, must be respected and merits that its use be reserved for providers with anesthesia training. Propofol causes profound and dose-dependent respiratory depression.8 An induction dose will usually result in apnea. Hypoxic ventilatory drive is inhibited, and the ventilatory response to hypercarbia (increased CO2) is diminished.9 Furthermore, both of these respiratory effects occur in subanesthetic infusion doses administered for conscious sedation.9 The principal cardiovascular effect of propofol is hypotension, which results as a decrease in systemic vascular resistance (also by inhibiting sympathetic-mediated vasoconstriction), preload, and cardiac contractility.9 Hypotension is more pronounced when the drug is administered rapidly or in large doses, or when used in elderly patients.9 Also, the baroreceptor reflex to hypotension is drastically impaired.9 Therefore, propofol causes a decrease in heart rate and cardiac output. Such decreases can be transient and tolerated in the healthy patient but if sufficiently severe, can lead to asystole, especially in patients taking negative chronotropic medications, such as ß-blockers, and patients at the extremes of age.9 It is also notable that patients do not develop a tolerance following long-term infusions of the drug9 and, thus, can develop significant respiratory and cardiovascular depression without requiring higher propofol doses.
Propofol has several other unique properties. While histamine release may occur, wheezing is less common with propofol than with other short-acting intravenous induction agents such as barbiturates and etomidate; propofol is not contraindicated for use in patients with asthma.9 Propofol also has antipruritic and antiemetic properties.8,9 Patients undergoing propofol-induced general anesthesia may occasionally exhibit excitatory motor activity, also known as nonepileptic myoclonia, which may include muscle twitching or hiccupping.8,9 However, propofol has anticonvulsant properties; thus, it is not contraindicated in patients with seizures and has been used in the termination of status epilepticus.8,9
Propofol was initially formulated with a Cremophor EL surfactant; however, the surfactant was associated with a high incidence of anaphylaxis.8,11 The current formulation of propofol is available only for intravenous administration as an egg lecithin emulsion, which provides a lipid vehicle for the drug in the aqueous phase.8,9,11,12 Because propofol is formulated in an egg lecithin emulsion, it is pertinent to consider allergic reactions to the drug or its lipid vehicle. Warnings for propofol differ throughout the world regarding food allergies, specifically allergies to eggs and soy; however, the United States has no food allergy warnings for this drug. The emulsion consists of 10% soybean oil, 2.25% glycerol, and 1.2% egg phosphatide B. Typically, this oil-in-water emulsion can produce injection-site pain,8,9 which can be reduced by administration of intravenous lidocaine either immediately before propofol injection or by mixing the propofol and lidocaine in a syringe.8,9 However, one must carefully calculate the dose of intravenous lidocaine to avoid systemic local anesthetic toxicity, especially in pediatric patients.8,9 Egg lecithin, which is from egg yolk, is a stabilizer and emulsifier in many prepared food products.12 Of the nine yolk proteins, only two are known to be allergenic. It is not clear whether these two proteins are found in egg lecithin.12 One study used skin prick and intradermal testing with propofol and the lipid vehicle in patients with egg allergies and produced negative results.11 Peanut allergies are concerning due to possible cross reactivity with soy.12 Another study concluded that a peanut allergy independent of soy or severe egg allergy is not a contraindication for propofol. The authors of the latter study concluded propofol can be safely administered in most children with egg allergies, but not severe egg anaphylaxis. They suggested more data were needed for soy or peanut allergies and propofol.12 Because type 1 hypersensitivity reactions worsen upon subsequent exposure and are not dose-dependent, it is important to consider that an egg-allergic individual who may be allergic to propofol may be primed to have an even more severe reaction to the egg protein if given propofol.12 In addition, the two isopropyl groups and the phenol group may be sensitizing antigenic epitopes of the drug propofol itself.12 It is the authors’ opinion that patients with allergies to eggs and/or soy should not receive this drug.
Drug Interactions/Intentional Drug Combinations for Sedation
Because propofol sedation usually results in deep sedation, the provider should be trained to rescue the patient from the next level of sedation, which is general anesthesia.13-15 Interestingly, in December 2009, the Centers for Medicare and Medicaid Services (CMS) issued a statement that said an anesthesiologist, certified nurse anesthetist, anesthesiologist assistant, or anesthesia-trained provider as recognized by state law (including medical doctors, doctors of osteopathy, dentists, and podiatrists) could administer deep sedation but must not be involved in the performance of the procedure itself.14,16 The CMS policy “applied to hospitals but was never issued for ambulatory surgery centers, which logically would have fewer airway experts available to rescue patients who were inadvertently oversedated.”14 This is exceedingly relevant to office-based sedation in the dental office, where the dentist may be an operator–sedationist.
While propofol has uniquely attractive advantages, such as a rapid onset and offset followed by a clear sensorium, the FDA maintains that the unique risks of propofol and demands of managing those risks render it an unacceptable drug for administration by an operator–sedationist, especially one that has not been formally trained in general anesthesia.7 Propofol has a narrow therapeutic index, meaning that the dose range to safely reach the desired level of sedation is small. It is easy for a patient to enter a deeper-than-intended level of sedation with propofol.7 Propofol is a potent, quick-acting drug, and it is possible for a patient to enter general anesthesia with a single intravenous dose within only 30 seconds.13 Interindividual variability in the metabolism of the drug is approximately 20-fold, so one dose can be rapidly metabolized in an individual and yet that same dose may be metabolized slower in another patient, risking anesthetic depth deeper than the provider originally intended.13 There is no reversal agent (receptor antagonist) for the drug, unlike benzodiazepines and opioids, which have reversal agents (flumazenil and naloxone, respectively.)13,14 Serious risks of propofol include partial or complete airway obstruction, apnea, and profound decreases in blood pressure.7 The resulting hypotension and arterial oxygen desaturation can manifest suddenly and become life-threatening if the appropriate interventions are not taken to mitigate these effects.7
Although it is true that a combination method for sedation consisting of benzodiazepines, opioids, and other sedative hypnotics (such as propofol) may provide therapeutic advantages from each drug class while reducing the adverse effects of giving larger doses of any of these drugs individually, this approach does not prevent respiratory depression and provides no guarantee for an added layer of patient safety. Such drug combinations can be alluring, as benzodiazepines provide anxiolysis and amnesia and opioids provide analgesia. However, all three drug categories can cause respiratory depression. In fact, such a regimen may potentiate respiratory depression from all three drug categories, despite the use of a smaller dose of propofol.7
Benzodiazepines play a key role in the armamentarium of the operator–sedationist non-general anesthesia-trained dental provider. Benzodiazepines have anxiolytic and amnesic effects while having less cardiac, respiratory, and central nervous system depression than opioids of propofol.17 Benzodiazepines can be titrated to effect, and a reversal agent, flumazenil, exists. This method of sedation can be supplemented with opioids, which provide analgesia and potentiate other sedatives, although the addition of an opioid invariably increases the risk of respiratory depression.18 Opioid oversedation may be antagonized with naloxone, but its use should be reserved to clinicians who are skilled in airway management. This is because respiratory depression can reoccur as the reversal agents have shorter half-lives than the agonist medications, which is especially concerning if multiple doses of the agonists have been given.17
While mild-to-moderate sedation may provide sufficient anxiolysis, analgesia, and amnesia for many patients, other patients may require deeper sedation or general anesthesia. It may be attractive to achieve a greater depth of anesthesia with more potent agents, but it could also prove to be dangerous in the wrong hands. According to the American Dental Association, “the drugs and/or techniques used [for conscious sedation] should carry a margin of safety wide enough to render unintended loss of consciousness unlikely.”19,20 In some states, inhalation and intravenous induction agents such as sevoflurane, methohexital, ketamine, propofol, and remifentanil are restricted agents, only to be used by those who have general anesthesia permits.19 The rationale for this decision made on the state level, as well as that of the FDA when issuing the warning label on propofol, is that there are unique challenges when using such drugs. An operator–sedationist dentist administrating propofol would have two responsibilities—the procedure and the deep sedation.19 Deep sedation has greater demands with regards to monitoring, maintaining an airway, and sustaining hemodynamic stability. Furthermore, the operator would be prone to increasing drug delivery to blunt a patient’s reaction to acute painful stimuli. The patient may enter a state of general anesthesia if too much drug is administered or if the painful stimulus is removed.19
Others have contended that using ketamine in combination with propofol can mitigate some unwanted adverse effects such as airway obstruction.21 These claims may seem like an enticing magic bullet, but using two induction agents is not safer than using one.21 Ketamine provides analgesia without increased apnea and raises blood pressure and heart rates. These effects oppose the cardiovascular and respiratory depression of propofol. However, ketamine also increases airway secretions, causes nausea and emesis, and is associated with recovery agitation.21 In one of the largest randomized, double-blinded studies for emergency procedural sedation, the incidence of adverse respiratory events in patients sedated with propofol alone versus ketamine and propofol was the same.21 Propofol did mitigate emesis caused by ketamine; however, the efficacy, induction time, and sedation time were similar in both groups.21 Apnea can also result from rapid ketamine administration even in the absence of propofol.21 In fact, subclinical respiratory depression was noted in a large proportion of adults receiving ketamine alone.21 Furthermore, there was a slightly higher incidence of airway complications when ketamine was given in higher doses or when ketamine was given with anticholinergics and/or benzodiazepines to preempt secretions that may irritate the respiratory tract or emergence agitation, respectively.15 It would be prudent to reserve the administration of induction agents and combinations of induction agents for those trained in general anesthesia and are proficient in airway and hemodynamic management.
The FDA Warning, Gastroenterology, and How Dentistry Is Different
The FDA has specific warning labels for propofol listed in the package insert, which state: “For general anesthesia or monitored anesthesia care (MAC) sedation, DIPRIVAN Injectable Emulsion should be administered only by persons trained in the administration of general anesthesia and not involved in the conduct of the surgical/diagnostic procedure.”7,13 The American College of Gastroenterology petitioned for removal of this warning label, but in 2010 the FDA denied this request for several valid reasons. The FDA noted the failure to rescue a patient from oversedation was a prominent cause of poor outcomes that have been reported in the gastroenterology literature.7 In addition, a single dose can result in general anesthesia within 30 seconds. Individual responses to propofol vary widely in the levels of anesthetic depth achieved, and a 20-fold variation in the metabolism of the drug among individuals was observed. As noted earlier, no reversal agent for propofol is available. The FDA has found a significant lack of training in gasteroenterologists and other medical specialists for employing propofol sedation—some physicians have as little as 20 minutes of training in airway management.7 Gastroenterology’s core curriculum makes no mention that a specific number of anesthesia cases are needed for proficiency. It is only suggested that the physician should “master” moderate sedation and have a “familiarity” with drugs intended for deep sedation, such as propofol.22
As the need for sedation and anesthesia in dentistry rises, dental practitioners can learn several lessons from the FDA’s decision. While operator–sedationists trained in intravenous moderate sedation may desire to provide deep sedation, it would be unsafe to attempt sedation with propofol for the same reasons cited by the FDA with regards to operator–sedationist gasteroenterologists, namely the lack of formal education and training in anesthesiology coupled with the drug’s ability to induce profound respiratory depression and the loss of a patent airway. Anesthesia providers within dentistry, such as dentist–anesthesiologists and oral and maxillofacial surgeons, are different from gastroenterologists, as both of the former have had months to years of formal training in anesthesiology. In fact, dentist–anesthesiologists must complete training in at least 500 general anesthesia cases.23 With such adequately educated providers within the dental community, as well as nurse–anesthetists and physician–anesthetists, there is no valid reason to believe that the benefits of administering propofol by individuals trained in moderate intravenous sedation will exceed the many risks outlined in this article. Furthermore, the American Society of Anesthesiologists (ASA) has recognized the profound potential for respiratory depression and even death with any drug regimen.24 ASA has expanded its requirements for standard basic anesthetic monitoring to include mandatory exhaled end-tidal carbon dioxide monitoring for any moderate sedation, deep sedation, or general anesthetic administered to a patient.24 Only with formal training in anesthesia can one expect safe provision of deep sedation with this drug and the ability to recognize and mitigate any complications, such as maintaining airway patency, ventilation, and hemodynamic stability in a full spectrum of patients.
Conclusions
Although behavioral management is the safest and least physiologically disruptive means for relieving fear and anxiety in dental patients, some still require conscious sedation. These include very young children who are not only anxious but are uncooperative because they are unable to understand the benefit to the upcoming dental treatment, the extremely fearful adult, the medically compromised and those with special needs, severe gaggers, patients with allergies to local anesthesia, and otherwise healthy patients undergoing invasive procedures such as the extraction of impacted third molars.19 Conscious or moderate sedation without using drugs such as propofol is often effective for most patients in the hands of adequately trained providers. With the exception of patients with allergies to amide local anesthetics, profound local anesthesia is still requisite in any successful sedation regimen. Compensation for inadequate local anesthesia should not be done with deeper sedation or general anesthesia, which is accompanied by risks for cardiovascular and respiratory depression.
While drugs such as propofol provide deeper sedation with quicker onset and offset, the potential for the patient to achieve a deeper level of anesthesia with profound cardiovascular depression, airway obstruction, and apnea warrants that the use of this agent be reserved for those who have proficiency in the administration of general anesthesia. The FDA warning on propofol states that only providers trained in anesthesia and whose undivided focus is in administering the drug and monitoring the patient should perform sedation procedures employing propofol.7 The FDA maintained this warning in response to a petition by gastroenterologists requesting the warning label be removed. The FDA clearly stated that nonanesthesiologists can have the appropriate education and experience to administer propofol along with the ability to quickly identify and respond to the airway, respiratory, cardiovascular, and hemodynamic sequelae as these problems may arise precipitously.7,13 Unlike gastroenterologists, dentists have a low mortality rate among their patients in outpatient anesthesia: Gastroenterologists had 52 mortalities per 1,048 practitioners in a 2-year period; the mortality for general anesthesia provided by both oral surgeons or dentist–anesthesiologists ranged from 1 in 248,000 cases18 to 1 in 840,000 cases.18 Based on data collected between 1973 and 1995, dentist–anesthesiologists and oral and maxillofacial surgeons in Canada have demonstrated an excellent safety record of 1 mortality per 1.4 million cases of deep sedation or general anesthesia.18
Adequate anxiolysis and analgesia are essential elements in dentistry. This may consist of local anesthesia and behavioral management in patients or require mild-to-moderate sedation or perhaps deep sedation and general anesthesia. While propofol is a potent and valuable tool in the hands of an adequately trained anesthesia provider, there is a very real risk of cardiac and respiratory complications that preclude its use to only those formally trained in general anesthesia.
References
1. Propofol (Diprivan®). US Department of Justice. Drug Enforcement Administration. Office of Diversion Control. Drug & Chemical Evalaluation Section. http://www.deadiversion.usdoj.gov/drugs_concern/propofol.pdf. January 2013. Accessed February 20, 2013.
2. Wischmeyer PE, Johnson BR, Wilson JE, et al. A survey of propofol abuse in academic anesthesia programs. Anesth Analg. 2007;105(4):1066-1071.
3. Roussin A, Montastruc JL, Lapeyre-Mestre M. Pharmacological and clinical evidences on the potential for abuse and dependence of propofol: a review of the literature. Fundam Clin Pharmacol. 2007;21(5):459-465.
4. Kranioti EF, Mavroforou A, Mylonakis P, Michalodimitrakis M. Lethal self administration of propofol (Diprivan). A case report and review of the literature. Forensic Sci Int. 2007;167(1):56-58.
5. Wilson C, Canning P, Caravati EM. The abuse potential of propofol. Clin Toxicol (Phila). 2010;48(3):165-170.
6. Denisco RC, Kenna GA, O’Neil MG, et al. Prevention of prescription opioid abuse: the role of the dentist. J Am Dent Assoc. 2011;142(7):800-810.
7. Food and Drug Administration, US Department of Health and Human Services. Docket No. 2005P-0059. http://isobsurgery.org/wp-content/uploads/2010/08/Propofol-FDA-denial.pdf. Accessed February 20, 2013.
8. Barash PG, Cullen BF, Stoelting RK, et al. Clinical Anesthesia. 6th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2009.
9. Morgan GE, Mikhail MS, Murray MJ. Clinical Anesthesiology. 4th ed. New York, NY: McGraw-Hill Medical; 2005.
10. Becker DE. Pharmacodynamic considerations for moderate and deep sedation. Anesth Prog. 2012;59(1):28-42.
11. Hepner DL, Castells MC. Anaphylaxis during the perioperative period. Anesth Analg. 2003;97(5):1381-1395.
12. Murphy A, Campbell DE, Baines D, Mehr S. Allergic reactions to propofol in egg-allergic children. Anesth Analg. 2011;113(1):140-144.
13. Food and Drug Administration. Docket No. 2005P-0267. http://www.fda.gov/ohrms/dockets/dockets/05p0267/05P-0267-EC185.htm. January 11, 2006. Accessed February 20, 2013.
14. Rex DK. Effect of the Centers for Medicare & Medicaid Services policy about deep sedation on use of propofol. Ann Intern Med. 2011;154(9):622-626.
15. Cravero JP, Havidich JE. Pediatric sedation—evolution and revolution. Paediatr Anaesth. 2011;21(7):800-809.
16. Centers for Medicare and Medicaid Services. Clarifications of the Interpretive Guidelines for the Anesthesia Services Condition of Participation, and Revised Hospital Anesthesia Services Interpretive Guidelines-State Operations Manual (SOM) Appendix A, S&C-10-09-Hospital. CMS publication No. 100-07. Baltimore, MD.
17. Finder R, Moore P, Close J. Flumazenil reversal of conscious sedation induced with intravenous fentanyl and diazepam. Anesth Prog. 1995;42(1):11-16.
18. Dionne R, Yagiela A, Moore PA, et al. Comparing efficacy and safety of four intravenous sedation regimens in dental outpatients. J Am Dent Assoc. 2001;132(6):740-751.
19. Yagiela J. Making patients safe and comfortable for a lifetime of dentistry: frontiers in office-based sedation. J Dent Educ. 2001;65(12):1348-1356.
20. American Dental Association Guidelines for the Use of Conscious Sedation, Deep Sedation and General Anesthesia for Dentists. Chicago, IL: American Dental Association; 2000.
21. Andolfatto G, Abu-Laban RB, Zed PJ, et al. Ketamine-propofol combination (ketofol) versus propofol alone for emergency department procedural sedation and analgesia: a randomized double-blind trial. Ann Emerg Med. 2012;59(6):504-512.e1-e2.
22. A Journey Toward Excellence: Training Future Gastroenterologists. The Gasteroenterology Core Curriculum. 3rd ed. American Gasteroenterological Association. http://www.gastro.org/2007_Version_Core_Curriculum.pdf. May 2007. Accessed June 10, 2012.
23. Accreditation Standards for Advanced General Dentistry Education Programs in Dental Anesthesiology. Chicago, IL: Commission on Dental Accreditation, American Dental Association; 2007. http://www.ada.org/sections/educationAndCareers/pdfs/anes.pdf. 2007. Accessed June 10, 2012.
24. Weaver J. The latest ASA mandate: CO(2) monitoring for moderate and deep sedation. Anesth Prog. 2011;58(3):111-112.
Related Content:
A CE article, Prescribing Recommendations for the Treatment of Acute Pain in Dentistry, is available from CDEWorld at dentalaegis.com/go/cced365
About the Authors
Mana Saraghi, DMD
Instructor
Division of Pediatric Dentistry and Department of Periodontics
University of Pennsylvania School of Dental Medicine
Philadelphia, Pennsylvania
Former Chief Resident
Dental Anesthesiology
Jacobi Medical Center
Bronx, New York
Victor M. Badner, DMD, MPH
Chairman
Department of Dentistry
Jacobi Medical Center
Bronx, New York
Leonard R. Golden, MD
Chairman
Department of Anesthesiology
Jacobi Medical Center
Bronx, New York
Elliot V. Hersh, DMD, MS, PhD
Director
Division of Pharmacology and Therapeutics
Professor
Oral and Maxillofacial Surgery and Pharmacology
University of Pennsylvania School of Dental Medicine
Philadelphia, Pennsylvania

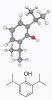 Figure 1 Propofol structure.">
Figure 1 Propofol structure.">