You must be signed in to read the rest of this article.
Registration on CDEWorld is free. You may also login to CDEWorld with your DentalAegis.com account.
The contemporary practice of dental medicine is characterized by a growing geriatric and medically complex population. The clinical significance of these demographic trends is seen in the use of more drugs and an increased risk for adverse drug interactions.1 Both prescription and common over-the-counter (OTC) drugs could lead to interactions that cause significant toxicity. A major source of such interactions involves alterations in the metabolic disposition of drugs, typically involving the prehepatic and hepatic cytochrome P450 system.1,2 Many drugs function as substrates, inhibitors, or inducers of individual cytochrome isoenzymes, and these types of interactions have been discussed in detail previously.1 These types of interactions are often not discovered and scientifically verified until multiple case reports are first published.1,3
Other potential drug interactions occur at the level of the drug receptor or, at the very least, at their proposed site(s) of action. The additive central nervous system (CNS) depressant effects of narcotics combined with alcohol is an example of an interaction occurring at two distinct types of CNS receptors, and appropriate warning of avoiding these combinations is given to patients by both dentists and pharmacists.
Recent literature has suggested a potentially serious interaction involving nonsteroidal antiinflammatory agents (NSAIDs) and selective serotonin reuptake inhibitors (SSRIs). In children and adults, SSRIs are widely prescribed drugs used for the treatment of depression, obsessive compulsive disorder, and anxiety.4-10 This article systematically reviews the published evidence supporting this interaction and suggests how these results may influence the prescribing of NSAIDs in these patients.
Methods
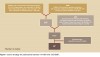
A structured literature search, limited to the years 1997-2007, is described in detail in Figure 1. Inclusion criteria were: population-based studies (studies including analysis of population-level data of a geographically defined sample), case-control, cohort, case-cohort, and randomized trials reporting outcome measures that included gastrointestinal (GI) bleeding. Acceptable exposure characteristics included recorded use of any type of SSRI and NSAID. Outcome characteristics included evidence (population, hospital, or national medical database) of hospitalization for GI bleeding or prescription of antiulcer drugs. Manuscripts with no specific outcome measure for gastric bleeding (eg, only general bleeding risk or cutaneous bleeding) were excluded. Case series, case reports, editorials, and letters were excluded from the final selection, as they offered only limited individual level data that was not able to be generalized. Data abstraction and manuscript selection was performed in duplicate (by two authors) and disagreement on inclusion was resolved by a third author. References from all selected manuscripts were reviewed by the authors to verify that no relevant manuscripts were missed (ie, papers with additional population-based data published between 1966 and 1997). A search was performed of the grey literature, and authors of four manuscripts included in the final selection were contacted successfully to inquire for any additional data or publications not identified in the search.
Results
Out of 15 studies that included the terms of interest, a total of nine studies (four case-control, one nested case-control, and four cohort) were identified for final review.7-15 One additional case-control study focusing on general bleeding events and SSRI use was excluded as it did not provide individual GI bleeding outcomes. Seven studies have been performed on adult European Caucasian populations, one in Canada, and one in the United States, with follow-up data collected ranging from 6 months to 13 years. A total of 30,334 cases of GI bleeding (or proxy for GI bleeding given by prescription of antiulcer drugs) were evaluated, and data on 446,427 patients were collected retrospectively in a cohort design. SSRI and NSAID exposure data varied among studies, ranging from 30 days to several years.
Seven out of nine manuscripts underscored the presence of an increased risk for adverse GI complications, mostly upper GI bleeding, in patients taking NSAID–SSRI combination therapy.7-9,11,12 One population-based study of 1,651 cases and 10,000 controls initially reported a 15-fold increase in risk of this event compared with the general population not taking either drug.9 Other studies performed stratified analyses of NSAID selectivity and SSRI potency, indicating a greater likelihood of a significant interaction with higher potency SSRIs (SSRIs with strongest inhibition of serotonin reuptake).8,9,11 The reported interaction is specific for NSAIDs and SSRIs even when controlling for other medications, and results were consistent across all studies, with the exception of Layton et al,15 in which sample selection (data were limited to four SSRIs, and the primary outcome was any bleeding secondary to SSRI use) and comparison groups (patients taking other psychiatric drugs with different demographic characteristics) could have attenuated this association. The strength of the association did vary depending on the population studied, but a dose–response relationship was seen in the increased risk linked to non-cyclooxygenase two (COX-2) selective NSAIDs and high potency SSRIs.9 These studies present epidemiologic differences, which are balanced by large sample sizes, similar outcome measures (with the exception of when a proxy for bleeding was used), and comparable effect sizes (strength of association) in three out of four manuscripts published in the past 5 years. To put these findings into clinical context, this article will first consider the data on NSAIDs and SSRIs separately and the mechanisms underlying their potential interaction.
NSAIDs
NSAIDs are among the most widely used medications in medical and dental practice because of their potent analgesic and antiinflammatory effects (Table 1). Four of these drugs (naproxen, celecoxib, ibuprofen, and diclofenac) are in the top 200 prescribed drugs in the United States, and these numbers do not take into account OTC sales of aspirin, ibuprofen, and naproxen.16,17 The indications for NSAIDs include the control of recurrent symptoms of many inflammatory diseases, including osteo- and rheumatoid arthritis, inflammatory degenerative disorders, and musculoskeletal pain.12,16-18 A major indication for aspirin is cardioprotection in at-risk individuals.12,19-26 NSAIDs are also highly effective in the treatment of postsurgical dental pain.16,19,20,27-31 NSAIDs act via inhibition of the cyclooxygenase one (COX-1) and COX-2 enzymes, which are important catalysts of the arachidonic acid pathway and the synthesis of prostaglandins.19-21 The high selectivity of certain NSAIDs, namely the “coxibs,” for blocking COX-2 greatly increases the gastrointestinal safety profile of these drugs, at least with chronic administration.16 Nevertheless, the widely publicized recall of two COX-2 inhibitors (rofecoxib and valdecoxib) because of increased incidence of cardiovascular events and, in the case of valdecoxib, an increased incidence of serious skin reactions, has left only celecoxib available for clinical use.16,19
NSAIDs are effective in reducing acute dental symptoms, and longer use (weeks to months) of NSAID therapy has been advocated for treatment of inflammatory temporomandibular joint (TMJ) arthropathies, synovitis, capsulitis, and other types of chronic orofacial pain.16,32 Their use is limited by side effects, particularly GI, the most severe being bleeding complications. Aspirin, in particular, is associated with reduced platelet function and nonoral bleeding risk.22,23,33,34 Every year, approximately 1% of patients taking long-term NSAIDs develop acute GI complications that require hospitalization.16 Moreover, mortality associated with NSAID-induced gastric complications as reported by the ARAMIS (Arthritis, Rheumatism, and Aging Medical Information System, a postmarketing surveillance of more than 36,000 patients from 12 centers in the United States and Canada) is comparable with the risk of significant morbidity from conditions such as AIDS.35-37
Upper GI complications induced by NSAIDs presumably occur secondary to systemic and topical effects on mucosal surfaces.38-40 Systemic activity derives from inhibition of the synthesis of endogenous prostaglandins, whereas acute mucosal insult results from depletion of mucosal prostaglandins and microcirculatory effects of interactions between leukocytes and endothelial cells. NSAIDs activate the expression of endothelial cell-adhesion molecules, which bind cellular receptors (mainly CD18) of polymorphonuclear leukocytes (PMNs), thereby increasing local inflammatory mediators and the potential for mucosal injury.37,38,40 Although the bleeding risk from chronic use of NSAIDs is known, there is scant information on the risk associated with number of prescriptions, time of use, or age at first prescription. It is thought that the prolonged use of NSAIDs by prescription carries a different safety profile than that of the same drugs used strictly by the reduced dose consistent with OTC guidelines, namely lower single and maximum daily dosages and no more than 10 consecutive days worth of use.24,33 A precise assessment of such risk is complicated by patients who purchase OTC drugs and self-administer them at doses and/or time intervals that are identical to prescription-use patterns.
Differences in the risk of developing serious GI bleeding or perforation also exist among individual NSAIDs, with selective COX-2 inhibitors (such as celecoxib) possessing the lowest risk.19 A comparison of rofecoxib vs naproxen in the rheumatoid arthritis model in the Vioxx Gastrointestinal Outcomes Research (VIGOR) study yielded an approximate 50% reduction in the risk of upper GI bleeding, ulcers, perforation, or obstruction, in the rofecoxib arm.19,40 A similar comparison between celecoxib and NSAIDs (ibuprofen and diclofenac) in patients treated for rheumatoid arthritis and osteoarthritis demonstrated fewer adverse gastric outcomes in the celecoxib arm (Celecoxib Long-term Arthritis Safety Study [CLASS]).19,41 Among noncoxibs, ibuprofen appears to have the lowest risk, followed by aspirin and naproxen with intermediate risk, and piroxicam and ketoprofen (with low-dose ketoprofen also being approved for OTC use) among the drugs with highest risk (9- to 10-fold greater risk than ibuprofen, respectively).24,36,38,39 Patients on chronic NSAID therapy also appear to have varying risk of developing serious GI complications. Patients with rheumatoid arthritis on NSAIDs carry a 2-fold greater risk of a serious complication than patients with osteoarthritis.34,38,39 This risk has been reported as high as 9-fold in a published report using a retrospective case-control design.42
These findings raise the question of whether there is any way to use premorbid symptoms to predict the risk for serious GI outcomes. Classic GI symptoms associated with NSAID intake have a low predictive value for forecasting serious GI outcomes, such ulcerations, perforations, and bleeds. Dyspepsia, for example, a common side effect of NSAID therapy, occurs in 15% of patients, and more than 50% of these do not have endoscopic findings consistent with mucosal injury.17,39,43,44 It seems that the severity of complications is associated with a previous history of gastric bleeding, chronicity of NSAID use, use of multiple NSAIDs, and interaction with other drugs, including anticoagulants and corticosteroids.43,44 The use of mucosal protective agents, such as misoprostol, or proton-pump inhibitors has been suggested in high-risk patients.39,43,44 Although NSAIDs commonly are prescribed in daily practice, it is imperative that clinicians recognize the importance of cautious and time-limited dosing of these analgesics, especially in patients with chronic pain.2,16,32
SSRIs
SSRIs are indicated for treatment of depression, obsessive compulsive disorder, acute anxiety, and panic disorders13,45-49 (Table 2). Among these, depression is a major cause of morbidity and is among the top 5 diagnoses requiring office visits in the United States.4,13,47 In 2006, escitalopram, paroxetine, fluoxetine, and sertraline were among the top 200 drugs of all categories prescribed in the United States.17 SSRIs target the serotonin reuptake pump, sparing other neurotransmitter transporters and the fast sodium channels. Prolonged exposure to serotonin at a dendritic level leads to down-regulation of postsynaptic receptors, which appears to correlate with clinical improvement, at least in regards to depression.50 Significant drug interactions are known to occur when prescribed in combination with other serotonergic agents, such as high doses of certain narcotics like propoxyphene (Darvon®, Xanodyne Pharmaceuticals Inc, Newport, KY), meperidine (Demerol®, Sanofi-Synthelabo Inc, New York, NY), and tramadol (Ultram®, Ortho-McNeill Pharmaceutical Inc, Raritan, NJ), leading to the development of a serotonergic syndrome, consisting of tachycardia, sweating, dilated pupils, hypertension, hyperthermia, and, in severe cases, seizures, metabolic acidosis, renal failure, and intravascular coagulation, and the development of extrapyramidal symptoms, including tardive dyskinesia or dystonia.48,49 In addition, several reports raise the potential for bleeding complications in patients on SSRIs.14,15,50,51 As such, the potential of these drugs to increase postoperative bleeding in patients after oral and periodontal surgery procedures needs to be explored.
The pathophysiology underlying the bleeding side effects of these drugs is related to decreased platelet serotonin uptake that inhibits aggregation.52,53 Many clinicians are aware of the importance of the cyclooxygenase product, thromboxane A2, in normal platelet aggregation; however, they may not know serotonin also plays a pivotal role. Being anucleated, platelets do not produce their own serotonin, but somewhat analogous to neurons in the central nervous system, “suck it up” from the plasma. This uptake blockage affects the platelets’ ability to capture and secrete serotonin, which down-regulates the expression of important platelet cell surface markers, responsible for the aggregative and migratory characteristics of these cells.52 Some evidence suggests an immediate prothrombotic effect of SSRIs, which is balanced by this platelet-inhibitory effect with prolonged therapy.8,52,54 Studies performed on patients with moderate-to-high cardiovascular risk demonstrated that SSRIs produce an increased antiplatelet effect in participants already taking aspirin.55 Reports of bleeding complications with use of SSRIs include the drugs paroxetine, fluvoxamine, sertraline, fluoxetine, and citalopram.45
Several authors have proposed a significant association between SSRIs and the prehepatic and hepatic cytochrome P450 system, in particular CYP2D6, as partially responsible for the onset of gastric complications.50,52 This interaction depends on the type of SSRI, as some (paroxetine and fluoxetine) are potent inhibitors of CYP2D6 and others (sertraline and fluvoxamine) are not.50,56 Certain individuals may be “slow metabolizers” of SSRIs because of genetically programmed low levels of CYP2D6 or CYP2C9 leading to SSRI accumulation and an increased risk of gastric bleeding.
Population-based studies, mostly done in Europe (United Kingdom and Denmark), have identified the use of SSRIs as an independent factor for bleeding risk, even without the effect of drug interactions.9,14,33,46 In addition, analysis of nonserotonergic antidepressant drugs did not yield any adverse effect on hemostasis.9 Several studies reviewed the risk related to the stratification of SSRIs according to serotonin reuptake potency for further analyses.8,9,11 As expected, drugs with higher potency carry the highest risk of gastric complications.
NSAIDs and SSRIs: Is There an Interaction?
In view of the described complications arising from both drugs individually, it is plausible that their combined use could lead to an increased effect with respect to GI bleeding risk7-9,13,14,46-48 (Figure 2). What is alarming is the magnitude of the risk associated with this interaction reported in several studies.
In the previously mentioned study on SSRIs and upper gastric bleeding,9 de Abajo reported a 15-fold increased risk of an upper GI bleed with the combination of SSRIs and NSAIDs, compared to the risk of 10,000 non-SSRI or non-NSAID user population controls. A Danish study in 2003 examined 26,005 patients in a limited geographic location and compared the incidence of upper GI bleeding with the combined use of SSRIs and NSAIDs with the general inhabitants of the county of North Jutland, Denmark. The authors identified a 12-fold increased risk associated with the combined use of both types of drugs (5.2-fold greater risk for concurrent aspirin use in patients consuming SSRIs) compared with population-expected risks of all the inhabitants of the county who were not prescribed either drug nor were at risk for gastric or esophageal bleeding between the years of 1977 and 1995.14 Non-SSRI antidepressants did not carry a significant risk, even when combined with NSAIDs. Time and duration of the antidepressants did not affect the risk for bleeding. A Canadian cohort study examined 317,824 people in a drug-monitoring database.8 It concluded that the combinations of NSAIDs and SSRIs increased the risk for gastric bleeding events 3-fold when compared with the use of NSAIDs or aspirin alone. This study failed to make comparisons with the general population not on either drug. An additional cohort study from England examined 15,445 new users of antidepressants, using a first prescription for peptic ulcer drugs as a proxy measure for gastric complications.15 A 12-fold increase in risk was reported with the concurrent use of both medications compared with unexposed controls not on either drug, in agreement with the Danish study reported 1 year later.
Providing a somewhat divergent perspective on the magnitude of the interaction, a case-control retrospective study from the United Kingdom, used a population database, The Health Improvement Network (THIN),10 to compare 11,261 cases with upper GI bleeding with 53,156 age- and gender-matched controls. The authors concluded that the incidence rates and odds ratios for NSAIDs and SSRIs used alone or in combination did not differ significantly, even among subsets of NSAIDs. The overall risk for each of the drugs used alone was 2-fold greater compared with nonusers, with only an approximate 3-fold (incidence ratio of 3.25) increase when used together (still representing a 50% increase in the event compared with NSAID or SSRI use alone). A recent case-control Finnish study evaluated cases of serious upper GI events (perforation, ulceration, or bleeding) drawn from the Hospital Discharge Registry (n = 9,191) and 41,780 controls from the population registry between 2000 and 2004.11 The authors reported a 60%, statistically significant increase in risk for GI complications with the combined use of NSAIDs and SSRIs vs NSAIDs alone (odds ratio of 1.57) and a 4.19-fold increased risk compared with nonuse of either drug. The increase, however, was far from the 12- to 15-fold increase in risk reported in 1999, 2002, and 2003. The same study reported a negligible reduction in gastric risk with semiselective COX-2-blocking NSAIDs (nimesulide, nabumetone, meloxicam, and etodolac) when taken with SSRIs (adjusted odds ratio reduced from 4.19 to 4.17 compared with nonuse of either drug class) in 30 patients, and a major reduction in risk (adjusted odds ratio reduced from 4.19 to 2.56) with concurrent highly selective COX-2 inhibitors and SSRIs, compared with the combination of nonselective NSAIDs and SSRIs. The vast majority of epidemiologic measures of association reported in these studies are higher when comparing the interaction of both classes (SSRIs and NSAIDs) with the GI risk of each drug alone.
Limitations
The studies identified by the authors’ search carry the robustness of population-based data and large sample sizes. However, there are discrepancies (information bias) in the classification of antidepressants as SSRIs between studies. Some have included combined types of antidepressants within their scheme, such as trazodone and venlafaxine, which are not really SSRIs.4 Although most studies considered a common outcome defined as gastric bleeding events, one study used a proxy measure (prescription of peptic ulcer drug).15 Patients could have received such prescriptions for other side effects such as dyspepsia, which is more common than and not as reliable an indicator of gastric bleeding. Only a few of these studies examined the role of the level of serotonergic activity of the SSRI vs the COX-2 selectivity of the NSAID in relationship to the gastric complication.
The potential for confounding by indication has to be considered when analyzing this interaction in an observational study. Because the use of antidepressants and the disease for which they are used, namely depression, cannot be separated easily, it is not known if it is the drug or the disease causing the increased risk. For example, patients taking antidepressants may have higher underlying anxiety levels that may lead to increased hydrochloric acid production and increased bleeding. It is also possible that because of the retrospective nature of many of these studies, the potential for misclassification bias is increased, and whether this bias is differential is open to discussion.
Physiologic evidence supports an independent effect of NSAIDs and SSRIs on platelets. The reviewed data suggests that there is a temporal association between intake of both drugs and GI bleeding when compared with controls. The risk associated with chronic vs acute combination of these drugs is unknown. The strength of the association did vary depending on the population studied, but a dose–response relationship was seen in the increased risk group linked to non-COX-2-selective NSAIDs and high-potency SSRIs.
The possibility of publication bias also could skew the literature toward reporting only positive results from population-based studies. Most likely, negative results exploring the association between these drugs done in population-based settings would have been considered important enough to be published in the past 10 years.
Clinical Considerations in Dental Practice
A major concern for the clinician is whether the risk associated with the combined intake of these two drug classes represents an effect that should modify dental practice. It is known that the risk of gastric bleeding with the use of NSAIDs alone increases approximately 2-fold, although there is variation among individual NSAIDS (compared with nonusers). For example, in comparing individual NSAIDs with each other concerning the risk of a serious GI event, ibuprofen has an estimated relative risk of 1, naproxen of 7, indomethacin of 8, and ketoprofen of 10.5,17,18,22,24,33-36,38,39,43 Based on the evidence presented in this article, the estimated risk of serious GI complications with the use of NSAIDs and SSRIs is at least 3-fold higher than the risk in the non-NSAID or non-SSRI consuming population and approximately 1.5 times the risk of consuming an NSAID alone. This increase in risk seen in published studies may represent an important finding, particularly in patients with other elevated risk factors. The dental clinician should be prepared to perform an adequate risk assessment on patients consuming SSRIs when NSAID use is being considered for antiinflammatory or pain-control purposes, especially in those individuals who would use NSAIDs on a chronic basis. The literature on NSAIDs is not consistent with the definition of time of drug intake vs risk of gastric bleeding. Unfortunately, major gastric side effects occur sometimes within the first week of therapy and mostly are related to predisposing risk factors and maximum dosing of NSAIDs. Although one study did not find duration of NSAID–SSRI use to correlate with the risk of bleeding, it is likely that the risk for gastric complications will rise with longer-term exposures to both classes of drugs.10
Conclusion
In spite of methodologic differences, the existing epidemiologic evidence supported by the antiplatelet action of both drugs suggests that the combination of SSRIs and NSAIDs may be associated with an increased risk of bleeding GI complications compared with either drug group alone. Although significant GI bleeding is rare in patients taking SSRIs and NSAIDs concomitantly, the clinical implications of these findings suggest the following recommendations:
1. Dentists should consult patients’ physicians before the initiation of chronic and/or high-dose NSAID therapy in patients already taking SSRIs.
2. Practitioners should be aware of other medical issues that may exacerbate the risk of a clinically significant interaction, including a previous history of gastric or esophageal hemorrhage, coagulopathy, patient age greater than 65 years, alcoholism, the presence of Helicobacter pylori in the GI tract, and the intake of other concomitant medications with antiplatelet activity (clopidogrel, ticlopidine, or methotrexate).
3. Based on the information presented in this article, it appears that short-term use of NSAIDs in patients taking SSRIs carries a low risk for GI bleeding.
Randomized controlled clinical trials and additional epidemiologic studies are needed to further characterize the true extent of this interaction and which patients are, in fact, most susceptible to significant GI bleeding from the combination. Because a relatively large number of patients are taking SSRIs and because NSAIDs are prescribed routinely in dental practice, the authors felt it important to inform practitioners of the possible untoward effects of this combination.
References
1. Hersh EV, Moore PA. Drug interactions in dentistry: the importance of knowing your CYPs. J Am Dent Assoc. 2004;135(3):298-311.
2. Moore PA, Hersh EV. Common medications prescribed for adolescent dental patients. Dent Clin North Am. 2006;50(1):139-149.
3. Bailey DG, Dresser GK. Interactions between grapefruit juice and cardiovascular drugs. Am J Cardiovasc Drugs. 2004;4(5):281-297.
4. Mort JR, Aparasu RR, Baer RK. Interaction between selective serotonin reuptake inhibitors and nonsteroidal antiinflammatory drugs: review of the literature. Pharmacotherapy. 2006;26(9):1307-1313.
5. García Rodriguez LA, Barreales Tolosa L. Risk of upper gastrointestinal complications among users of traditional NSAIDs and COXIBs in the general population. Gastroenterology. 2007;132(2):498-506.
6. Zullino DF, Khazaal Y. Increased risk of gastrointestinal adverse effects under SSRI/NSAID combination may be due to pharmacokinetic interactions. Br J Clin Pharmacol. 2005;59(1):118-119.
7. de Jong JC, van den Berg PB, Tobi H, et al. Combined use of SSRIs and NSAIDs increases the risk of gastrointestinal adverse effects. Br J Clin Pharmacol. 2003;55(6):591-595.
8. van Walraven C, Mamdani MM, Wells PS, et al. Inhibition of serotonin reuptake by antidepressants and upper gastrointestinal bleeding in elderly patients: retrospective cohort study. BMJ. 2001;323(7314):655-658.
9. de Abajo FJ, Rodríguez LA, Montero D. Association between selective serotonin reuptake inhibitors and upper gastrointestinal bleeding: population based case-control study. BMJ. 1999;319(7217):1106-1109.
10. Tata LJ, Fortun PJ, Hubbard RB, et al. Does concurrent prescription of selective serotonin reuptake inhibitors and non-steroidal anti-inflammatory drugs substantially increase the risk of upper gastrointestinal bleeding? Aliment Pharmacol Ther. 2005;22(3):175-181.
11. Helin-Salmivaara A, Huttunen T, Grönroos JM, et al. Risk of serious upper gastrointestinal events with concurrent use of NSAIDs and SSRIs: a case-control study in the general population. Eur J Clin Pharmacol. 2007;63(4):403-408.
12. Helin-Salmivaara A, Huupponen R, Virtanen A, et al. Frequent prescribing of drugs with potential gastrointestinal toxicity among continuous users of non-steroidal anti-inflammatory drugs. Eur J Clin Pharmacol. 2005;61(5-6):425-431.
13. Wessinger S, Kaplan M, Choi L, et al. Increased use of selective serotonin reuptake inhibitors in patients admitted with gastrointestinal haemorrhage: a multicentre retrospective analysis. Aliment Pharmacol Ther. 2006;23(7):937-944.
14. Dalton SO, Johansen C, Mellemkjaer L, et al. Use of selective serotonin reuptake inhibitors and risk of upper gastrointestinal tract bleeding: a population-based cohort study. Arch Intern Med. 2003;163(1):59-64.
15. Layton D, Clark DW, Pearce GL, et al. Is there an association between selective serotonin reuptake inhibitors and risk of abnormal bleeding? Results from a cohort study based on prescription event monitoring in England. Eur J Clin Pharmacol. 2001;57(2):167-176.
16. Klasser GD, Epstein J. Nonsteroidal anti-inflammatory drugs: confusion, controversy and dental implications. J Can Dent Assoc. 2005;71(8):575-580.
17. Lamb E. Top 200 prescription drugs of 2006. Pharm Times. 2007;73(5):34-37.
18. Lanas A, Hirschowitz BI. Toxicity of NSAIDs in the stomach and duodenum. Eur J Gastroenterol Hepatol. 1999;11(4):375-381.
19. Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in colorectal adenoma chemoprevention trial [published erratum appears in: N Engl J Med. 2006;13;355(2):221]. N Engl J Med. 2005;352(11):1092-1102.
20. Moore PA, Hersh EV. Celecoxib and rofecoxib. The role of COX-2 inhibitors in dental practice. J Am Dent Assoc. 2001;132(4):451-456.
21. Chan FK, Wong VW, Suen BY, et al. Combination of a cyclo-oxygenase-2 inhibitor and a proton-pump inhibitor for prevention of recurrent ulcer bleeding in patients at very high risk: a double-blind, randomised trial. Lancet. 2007;369(9573):1621-1626.
22. Vonkeman HE, Fernandes RW, van der Palen J, et al. Proton-pump inhibitors are associated with a reduced risk for bleeding and perforated gastroduodenal ulcers attributable to non-steroidal anti-inflammatory drugs: a nested case control study. Arthritis Res Ther. 2007;9(3):R52.
23. Sorensen HT, Mellemkjaer L, Blot WJ, et al. Risk of upper gastrointestinal bleeding associated with use of low-dose aspirin. Am J Gastroenterol. 2000;95(9):2218-2224.
24. Hersh EV, Moore PA, Ross GL. Over-the-counter analgesics and antipyretics: a critical assessment. Clin Ther. 2000;22(5):500-548.
25. Kimmey MB. Cardioprotective effects and gastrointestinal risks of aspirin: maintaining the delicate balance. Am J Med. 2004;117(Suppl 5A):72S-78S.
26. Gaziano JM, Gibson CM. Potential for drug-drug interactions in patients taking analgesics for mild-to-moderate pain and low-dose aspirin for cardioprotection. Am J Cardiol. 2006;97(9A):23-29.
27. Hersh EV, Levin LM, Adamson D, et al. Dose-ranging analgesic study of Prosorb diclofenac potassium in postsurgical dental pain. Clin Ther. 2004;26(8):1215-1227.
28. Hersh EV, Levin LM, Cooper SA, et al. Ibuprofen liquigel for oral surgery pain. Clin Ther. 2000;22(11):1306-1318.
29. Cooper SA, Schachtel BP, Goldman E, Gelb S, et al. Ibuprofen and acetaminophen in the relief of acute pain: a randomized, double-blind, placebo-controlled study. J Clin Pharmacol. 1989;29(11):1026-1030.
30. Dionne RA, Gordon SM. Nonsteroidal anti-inflammatory drugs for acute pain control. Dent Clin North Am. 1994;38(4):645-667.
31. Kiersch TA, Halladay SC, Hormel PC. A single-dose, double-blind comparison of naproxen sodium, acetaminophen, and placebo in postoperative dental pain. Clin Ther. 1994;16(3):394-404.
32. Dionne RA. Pharmacologic approaches. In: Laskin DM, Greene CS, Hylander WL, eds. Temporomandibular Disorders: An Evidence-Based Approach to Diagnosis and Treatment. 1st ed. Chicago, IL: Quintessence Publishing Co, Inc; 2006:347-357.
33. Mellemkjaer L, Blot WJ, Sorensen HT, et al. Upper gastrointestinal bleeding among users of NSAIDs: a population-based cohort study in Denmark. Br J Clin Pharmacol. 2002;53(2):173-181.
34. Singh G, Triadafilopoulos G. Epidemiology of NSAID induced gastrointestinal complications. J Rheumatol. 1999;56(Suppl):18-24.
35. Abramson SB, Weaver AL. Current state of therapy for pain and inflammation. Arthritis Res Ther. 2005;7(Suppl 4):S1-S6.
36. Peura DA, Goldkind L. Balancing the gastrointestinal benefits and risks of nonselective NSAIDs. Arthritis Res Ther. 2005;7(Suppl 4):S7-S13.
37. Singh G. Gastrointestinal complications of prescription and over-the-counter nonsteroidal anti-inflammatory drugs: a view from the ARAMIS database. Arthritis, Rheumatism, and Aging Medical Information System. Am J Ther. 2000;7(2):115-121.
38. Laine L, Bombardier C, Hawkey CJ, et al. Stratifying the risk of NSAID-related upper gastrointestinal clinical events: results of a double-blind outcomes study in patients with rheumatoid arthritis. Gastroenterology. 2002;123(4):1006-1012.
39. Singh G, Ramey DR, Morfeld D, et al. Gastrointestinal tract complications of nonsteroidal anti-inflammatory drug treatment in rheumatoid arthritis. A prospective observational cohort study. Arch Intern Med. 1996;156(4):1530-1536.
40. Bombardier C, Laine L, Reicin A, et al. VIGOR Study Group. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. VIGOR Study Group. N Engl J Med. 2000;343(21):1520-1528.
41. Silverstein FE, Faich G, Goldstein JL, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: A randomized controlled trial. Celecoxib Long-term Arthritis Safety Study. JAMA. 2000;284(10):1247-1255.
42. García Rodríguez LA, Jick H. Risk of upper gastrointestinal bleeding and perforation associated with individual non-steroidal anti-inflammatory drugs [published erratum appears in: Lancet. 1994;343(8904):1048]. Lancet. 1994;343(8900):769-772.
43. Silvani MC, Motola D, Poluzzi E, et al. Gastro-intestinal problems and concomitant medication in NSAID users: additional findings from a questionnaire-based survey in Italy. Eur J Clin Pharmacol. 2006;62(3):235-241.
44. Lanas A, García-Rodríguez LA, Arroyo MT, et al. Effect of antisecretory drugs and nitrates on the risk of ulcer bleeding associated with nonsteroidal anti-inflammatory drugs, antiplatelet agents, and anticoagulants. Am J Gastroenterol. 2007;102(3):507-515.
45. Yuan Y, Tsoi K, Hunt RH. Selective serotonin reuptake inhibitors and risk of upper GI bleeding: confusion or confounding? Am J Med. 2006;119(9):719-727.
46. Dalton SO, Sørensen HT, Johansen C. SSRIs and upper gastrointestinal bleeding: what is known and how should it influence prescribing? CNS Drugs. 2006;20(2):143-151.
47. Serebruany VL. Selective serotonin reuptake inhibitors and increased bleeding risk: are we missing something? Am J Med. 2006;119(2):113-116.
48. Weinrieb RM, Auriacombe M, Lynch KG, et al. Selective serotonin re-uptake inhibitors and the risk of bleeding. Expert Opin Drug Saf. 2005;4(2):337-344.
49. Mansour A, Pearce M, Johnson B, et al. Which patients taking SSRIs are at greatest risk of bleeding? J Fam Pract. 2006;55(3):206-208.
50. Lam YW, Gaedigk A, Ereshefsky L, et al. CYP2D6 inhibition by selective serotonin reuptake inhibitors: analysis of achievable steady-state plasma concentrations and the effect of ultrarapid metabolism at CYP2D6. Pharmacotherapy. 2002;22(8):1001-1006.
51. Brambilla P, Cipriani A, Hotopf M, et al. Side-effect profile of fluoxetine in comparison with other SSRIs, tricyclic and newer antidepressants: a meta-analysis of clinical trial data. Pharmacopsychiatry. 2005;38(2):69-77.
52. Horiuchi H. Recent advance in antiplatelet therapy: the mechanisms, evidence and approach to the problems. Ann Med. 2006;38(3):162-172.
53. Hersh EV, Moore PA. Adverse drug interactions in dentistry. Periodontology 2000. 2008;46:109-142.
54. de Abajo FJ, Jick H, Derby L, Jick S, et al. Intracranial haemorrhage and use of selective serotonin reuptake inhibitors. Br J Clin Pharmacol. 2000;50(1):43-47.
55. Serebruany VL, Glassman AH, Malinin AI, et al. Selective serotonin reuptake inhibitors yield additional antiplatelet protection in patients with congestive heart failure treated with antecedent aspirin. Eur J Heart Fail. 2003;5(4):517-521.
56. Liston HL, DeVane CL, Boulton DW, et al. Differential time course of cytochrome P450 2D6 enzyme inhibition by fluoxetine, sertraline, and paroxetine in healthy volunteers. J Clin Psychopharmacol. 2002;22(2):169-173.
About the Authors
Andres Pinto, DMD, MPH,
Assistant Professor of Oral Medicine
Director
Oral Medicine Clinical Center
Department of Oral Medicine
University of Pennsylvania
School of Dental Medicine
Philadelphia, Pennsylvania
John T. Farrar, MD, PhD
Academic Director
Educational Programs in Epidemiology
Center for Epidemiology and Biostatistics
University of Pennsylvania
School of Medicine
Philadelphia, Pennsylvania
Elliot V. Hersh, DMD, MS, PhD
Professor of Pharmacology
Director Division of Pharmacology
Department of Oral and Maxillofacial Surgery/Pharmacology
University of Pennsylvania
School of Dental Medicine
Philadelphia, Pennsylvania